Introduction
A multifunctional coating is defined as a material that has an excellent combination of mechanical, electrical, biological, chemical, magnetic, thermal, or optical properties which make it indispensable for improving the durability and applicability of conventional materials. One of them is nanocomposite coatings, which are formed by mixing at least two different materials or combining a crystalline and an amorphous phase. This union allows obtaining the best properties of each phase, combined in a material with unique characteristics (Vanegas et al., 2019).
The component with the highest percentage is called the matrix, and the one with the lowest percentage is called reinforcement, which must be nanometer-sized (Prakash Sharma et al., 2018). Now, the nanocomposite coating is formed by at least two phases, i.e., a nanocrystalline phase and an amorphous phase, or two nanocrystalline phases. These coatings can exhibit a hardness significantly higher than that given by each of the parts of the mixture; they can be hard, super-hard, or even ultra-hard, depending on the coating design and application. Hard materials generally refer to materials with a hardness greater than 20 GPa, those with a hardness greater than 40 GPa are classified as super-hard, and those with a hardness greater than 80 GPa are often referred to as ultra-hard materials.
In recent years, TiN, CrN, TiAlN, and TiCrN thin films have been used as hard coatings due to their high hardness, chemical stability, corrosion resistance, and wear resistance. As for the oxidation of these films, play an important role in cutting tool applications because of their frequent exposure to oxidative atmospheres at high temperatures during machining processes where the use of synthetic coolant is not very common.
Theoretical and experimental studies have been carried out to synthesize and study these nanocomposite coatings with super-hardness and high toughness (Ardila-Téllez et al., 2014). Nanocomposite films with high mechanical properties are generally characterized by their low signal intensity in an X-ray diffraction pattern. These films form in transition regions, where their crystalline structure undergoes significant changes. There are three types of changes. The first is related to a transition from a crystalline to an amorphous structure, the second involves a change between the crystalline phases of the material, and the third implies changes in the preferential orientation of the grains (Popescu et al., 2009).
The transition from a crystalline phase to an amorphous phase occurs when an element B (e.g., Si) is added to the MeN compound (Me corresponds to a transition metal or an alloy composed of two or three transition metals, and N is nitrogen) in order to form an amorphous phase with ByNz and MeN nanoparticles in the MeBN grown film. At low Si contents, i.e., not higher than 7 at%, the compounds are found in the crystalline region, close to the amorphous transformation zone, and may even be substitutional atoms within the crystalline metal nitride (MeN) (Macías et al., 2019).
In general terms, this article aims to present a review of the main properties and applications of multifunctional coatings of the MeSiN form produced with PVD processes. As an example, the TiAlCrSiN system is presented, which has achieved an adequate combination of mechanical, tribological, and anticorrosive properties. These properties are important for applications in the metal-mechanical, naval, biomedical, and aeronautical industries. To this effect, the evolution of these materials is systematically presented, staring with the main binary stoichiometries, continuing to the ternary ones, until finally achieving quaternary coatings.
Methodology
Deposition Methods
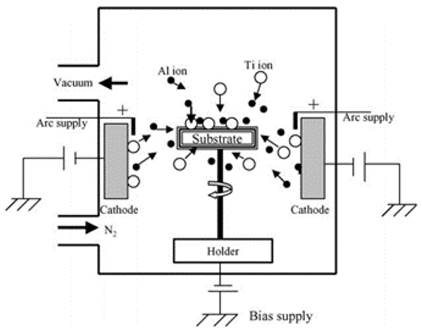
Multifunctional nitride coatings can be successfully produced by employing physical vapor deposition techniques such as sputtering and cathodic arc techniques. Coatings based on the magnetron sputtering technique are carried out in vacuum chambers and with the use of DC or RF sources. Inside the chamber, the substrate is connected to a negative potential, or left floating, and the target is used as a cathode (Figure 1). The process starts with the evacuation of the air in the chamber at pressures ranging from 0.75 to 7.5e-4 mTorr. The chamber is usually filled with argon gas (with p.d = 0.3 torr·cm) due to the argon’s low reactivity and appreciable mass. Then, a potential difference of 300 to 5000 V is applied between the anode and the cathode, which produces an electrical discharge due to the emission of electrons from the cathode and argon gas ionization (Baptista et al., 2018; Borra, 2006).
The positive ions are accelerated against the target (negatively charged), thus producing sufficient momentum transfer to sputter their atoms, which then condense on the substrate surface. The advantage of this process lies in the wide variety of materials that can be used as target. The disadvantages are low deposition rates and the contamination of the deposited films, mainly due to particles generated by the heat of the plasma on the chamber walls and the components of the vacuum system.
The basic sputtering process has been used for several years, and many materials have been deposited using this system. To improve the efficiency of the sputtering process, a magnetron is included. Magnetrons are configured in such a way that one pole is positioned at the central axis of the target, and the second pole is formed by a ring of magnets around the outer edge of the target (Mattox, 2010). In this way, the electrons are confined, and the probability of collision with atoms is increased. The increased ionization efficiency of magnetron results in a dense plasma in the target region, which is associated with the ion bombardment of the target, this yielding higher sputtering and deposition rates, as well as and a decrease in the working pressures. In fact, in normal magnetron conditions, the discharge could be maintained at lower operating pressures (typically, 10~3 mbar) and lower operating voltages, typically -500V (Kelly and Arnell, 2000).
In recent years, the density and reactivity of this process have significantly increased through the inclusion of high-power pulsed magnetron sputtering (HIPIMS). This process can employ a DC generator source to charge the capacitor bank of a pulsed unit, which is connected to the magnetron.
The charging voltage of the capacitor bank typically ranges from several hundred V to several kV. The stored energy is released in pulses of defined frequency and width, using transistors with a switching capacity in the μs range, which are located between the capacitors and the cathode. The pulse width (also referred to as pulse-in-time) varies, typically from 5 to 5000 μs, while the pulse repetition frequency ranges from 10 to 10 kHz. Under these conditions, the target peak current density can reach values of 3-5 A/cm2, which are up to three orders of magnitude higher than conventional sputtering densities. Furthermore, the ionization levels in the pulsed process are very high, reaching values close to 90% of the eroded particles on the target. This is due to the appearance of power density zones on the target, whose behavior obeys a stochastic process that cannot be predicted.
The second process (Figure 2) is the cathodic arc, which has considerable potential in the present and future of the tool coating industry, mainly because of its simplicity, easy implementation, low operating cost under industrial production conditions, and especially because of its high ionization rate, which accelerates the deposition process and also generates excellent adhesion to substrates (Curtins, 1995; Randhawa and Johnson, 1987).
Arc deposition is a PVD (physical vapor deposition) technique that involves the vaporization of an electrode by an electric arc as a plasma source (Lindfors et al., 1986; PalDey et al., 2004; Vetter and Perry, 1993). The arc conditions require high currents (˷̴ 100 A) and voltages close to the ionization potential of the gas or vapor (> 25 volts) (Mattox, 1998).
The ion bombards the cathode, and the electron bombards the anode, thus heating both electrodes. Most of the evaporated material becomes part of the plasma. However, a small portion of the material is ejected in the form of molten droplets. These small droplets (not vaporized) on the surface cause a substantial increase in roughness. This roughness is key in the case of cutting tools, as it increases the coefficient of friction with the chip and, therefore, the cutting forces. For a stable arc to form, a minimum current must be present. The current value varies from 50 to 100 A for low melting point materials such as copper and titanium. Meanwhile, for refractory materials such as tungsten, it reaches 300-400 A. The arc voltage can be between 15 and 100 volts, depending on the ease of electron movement from the cathode to the anode (Mattox, 1998).
Nanostructured and functional MeBN coatings deposited by PVD seek to protect the material from degradation, and their effects positively impact a nation's economy. From an environmental perspective, the degradation of a material by corrosion and wear phenomena becomes a source of pollution, as it reduces its useful life. For example, deterioration due to oxidation or pitting prevents the recycling of a product and causes the emission of substances that affect the quality of the air or water, thus affecting the health conditions of species. A second factor is related to the consumption of energy and resources needed to replace or recover a degraded material. In addition, the manufacture of a new product produces gases and liquid waste that can pollute the environment.
On the other hand, in the metalworking industry, where the machining of a product for an aeronautical, automotive, or biomedical application is based on the detachment of material due to wear with a cutting tool, it is important to note that machining processes constitute approximately 15% of the economic cost of the product in industrialized countries (Markopoulos, 2013). In the same way, tool wear has additional environmental effects, such as increased energy consumption, wear in other parts of the machine, increased emission of toxic gases due to evaporation of coolants or lubricants, and increased noise production. This means that, if functional coatings such as PVD techniques are developed, more durable materials can be produced, and better use will be made of the available resources, resulting in reduced energy consumption and less damage to the environment.
Binary Coatings
Transition metal nitrides have a considerable content of metal bonds, although nitrogen is covalently bonded to metals. In a densely packed in FCC cubic structure, there are two interstitial sites: the tetrahedral and octahedral sites. Nitrogen occupies only the octahedral sites because the tetrahedral sites are too small to accommodate nitrogen. Nitrides form, as groups 4 and 5 have refractory characteristics, with melting points above 1800 °C; while the refractory properties of group 6 nitrides, such as CrN, MoN, and WN, are reduced because they have low melting points and dissociate rapidly to N2 and MT at temperatures near 1000 °C (Capote-Rodríguez et al., 2015).
Metal nitrides have been applied in the industry due to their excellent response when deposited on metallic surfaces. Metal nitrides from groups 4 to 6 are the most widely applied because they exhibit the following properties:
Low friction coefficients (kinetic)
Good surface finish
High surface hardness, good adhesion, and abrasion resistance
Increased corrosion resistance
They are chemically stable and inert
Titanium Nitride (TiN) Coatings
Titanium nitride (TiN) is classified as a gold-colored ceramic material with 1800-2100 HV (Table 1). It is one of the first thin films obtained by using the PVD technique (Xi et al. 2017) to improve the surface properties of substrates such as steels, titanium alloys, hard metals, aluminum, and carbides. It is generally applied in cutting tools, elements for medical applications, parts for the food industry, and mold components, among others (Domínguez-Crespo et al., 2018).
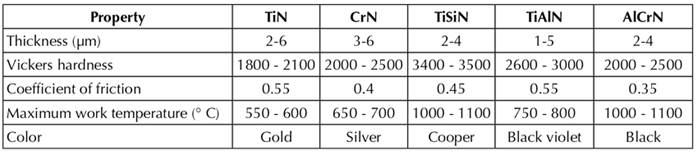
Table 1 Comparison of nitride-based nanostructured coatings properties (MatWeb - Material Property Data, 2020).
Titanium nitride (TiN) has a friction coefficient between 0.4 and 0. 9 without lubrication, depending on the surface finish and substrate material. Its melting point is 2930 °C, and it oxidizes at a temperature of approximately 600 °C in a normal atmosphere. Its typical formation has a crystalline NaCl-type structure with a determined stoichiometric ratio of 1:1. TiNx compounds are thermodynamically stable in the x-range of 0.6 to 1.2 (C. F. Wang et al., 2014).
It has been reported that the bonding of these two materials is a combination of three types of bonds (Barshilia et al., 2010): (i) the ionic bond resulting from the transfer of electrons from titanium to nitrogen, since the latter has a higher electronegativity (the electronegativity of N is 3.04 and that of Ti is 1.54); (ii) metallic bonding, with a finite density of states at the Fermi energy level, (iii) covalent bonding, which occurs in a higher proportion and is generated by the superposition between the d states of titanium and the p states of nitrogen, with some metal-metal interaction.
Chromium Nitride (CrN)
This coating, obtained via the PVD process, exhibits hardness in the range of 2000 to 2500 Vickers (Table 1), has a higher temperature resistance than TiN, and is mainly applied in precision metal parts (Kabir et al., 2016). CrN thin films are ideal in industrial applications where corrosion protection and high adhesive wear resistance are required (Olaya and Cifuentes, 2018; T. Wang et al., 2016).
It is generally used in dies for metal forming, plastic injection molds, and cutting tools for machining titanium alloys, as well as in non-ferrous alloys such as aluminum (Sui et al., 2018). The coating forms a metal bond with the substrate which does not form chips, flakes, or delamination. In several nanostructured coatings, CrN or pure chromium is deposited on the substrate as a thin adhesion film to prevent the delamination caused by residual stresses typical of PVD techniques or the mismatch of the thermal expansion coefficient between substrate and coating (Ou et al., 2016).
Ternary Coatings
Titanium-Silicon Nitride (TiSiN) Coating
Although TiN is widely used in the cutting tool sector, the need to improve the cutting speed and increase mechanized parts production led to research aimed at enhancing properties. In this sense, the addition of Si arises as an alternative to increase hardness and/or reduce its friction coefficient. Therefore, in the last 20 years, there has been an increase in the studies on this TiSiN material (An et al., 2008; Chawla et al., 2010; Ding et al., 2004; Diserens et al., 1998, 1999; Jiang et al., 2004; Liu et al., 2018a; Mei et al., 2005). With X-ray diffraction, the incorporation of silicon atoms was observed to cause a decrease in the peak high of TiN, which suggests crystallite size reduction or a tendency towards amorphous phase formation (Mei et al., 2005). This result proves the existence of Si3N4 in contrast to the multiple studies carried out by other authors who, based on X-ray analysis, did not find the presence of this nitride. The chemical composition of TiSiN, identified via the X-ray photoelectron spectroscopy technique (XPS), exhibits Ti 2p, Si 2p, and N 1s. In these results, it was observed that, for the Si 2p peak, the binding energy 101.7 eV demonstrates the existence of Si3N4 (Jiang et al., 2004). The N 1s peak exhibited a binding energy of 397.1 eV and showed the presence of TiN and, at 398 eV, the existence of Si3N4 (Jiang et al., 2004).
Regarding mechanical properties, Jiang et al. (2004) found a hardness of 3400-3500 Vickers at a 9% Si concentration. Mei et al. (2005) found a maximum hardness value of 3420 HV at a 7% Si concentration. Diserens et al. (1999), like the previous authors, evaluated the hardness, finding a maximum value of 3800 HV at a Si concentration of 5%. These values vary with a small silicon modification, which shows the remarkable effect of silicon addition on mechanical properties.
TiSiN is applied in cutting tools and is designed to achieve high-speed machining operations on hard materials such as nickel- and titanium-based alloys with no or minimal lubrication conditions. TiSiN-coated cutting tools are extremely wear-resistant. The coating could operate at temperatures of up to 1100 °C (Arab Pour Yazdi et al., 2014). The TiSiN layers are deposited on the cutting tool with the help of the PVD arc evaporation process or by reactive magnetron sputtering.
Titanium-Aluminium Nitride (TiAlN) Coatings
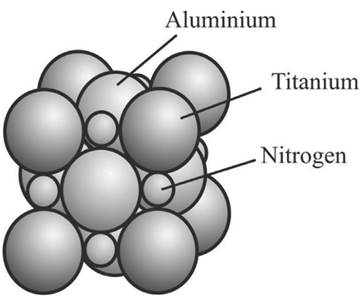
Titanium aluminum nitride has a complex structure consisting of TiN and AlN bonds in a crystalline matrix (Figure 3). Cutting tools with this violet-black coating are high-performance and ideal for machining abrasive and difficult-to-machine materials such as tool steels, aluminum alloys, cast iron, and nickel alloys (Keunecke et al., 2010).
AlTiN thin films exhibit higher temperature resistance than TiN (Barshilia et al., 2008) and slightly higher hardness, especially in hot work (Table 1). AlTiN coatings were developed from the tribological needs and characteristics of titanium nitride TiN (Liu et al., 2018b). The presence of high percentages of aluminum makes AlTiN harder and more thermally stable, even at high temperatures, where the formation of alumina favors heat dissipation, thus allowing for the reduced use of synthetic refrigerants (W. Zhou et al., 2014).
Nanostructured TiAlN coatings with a cubic NaCl-type structure, where aluminum replaces titanium in the TiN base structure (Ti1-XAlXN) by up to 65%, have been employed in metalworking due to their better oxidation resistance at elevated temperatures, higher hardness, and thermal stability in comparison with TiN. However, for aluminum contents exceeding the maximum solubility limit of x = 0.65, a mixture between a B1-NaCl-type cubic structure and a Wurtzite-type structure is created, which improves the oxidation resistance but degrades the mechanical and thermal properties of the TiAlN coating. Figure 4 shows a schematic of the structure of these coatings with increasing aluminum.
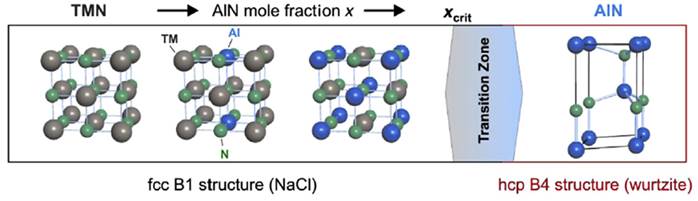
Figure 4 Transformation of the cubic B1 structure into the hexagonal B4 structure as an aluminum content (Mayrhofer et al., 2014).
It is also known that the substitution of Ti atoms by smaller atoms such as Al in TiN not only affects the size of the structure, but also its density, leading to a decrease in the crystal lattice. From this, the number of defects increases, thus leading to an increase of up to 65% in the hardness of the coatings, i.e., to a value close to 3300 HV. In addition, several authors have reported an improvement in oxidation resistance at elevated temperatures due to the formation of protective products such as aluminum oxide (Al2O3) (Oliveira et al., 2008; Stueber et al., 2005). Studies on industrial TiAlN coatings in machining processes at high cutting speeds show the formation of an aluminum oxide layer that helps to prevent high-temperature degradation of the nitride and acts as a diffusion barrier that reduces the chip, making it an excellent coating for machining abrasive materials. Thus, the use of TiAlN has been limited to applications for machining materials at medium to low speeds, as its performance depends on the formation of aluminum oxide.
TiAlN coatings are used to avoid extemporaneous consumption of wear-exposed cutting tools due to their excellent tribological properties, high-temperature oxidation resistance at a temperature range of 600-800 °C, and high chemical stability (Sarin, 2014), but the requirements of high-speed and dry cutting applications, for which a high hardness and a high-temperature oxidation resistance above 1000 °C are of great importance, place high demands on coatings that must possess high wear resistance. The improvement of these properties can be obtained by incorporating Si into TiAlN coatings during processing (Park et al., 2004; Veprek et al., 2004).
Miletic et al. (2014a) alternate TiAlN and TiSiN coating layers, expecting them to exhibit nanocoatings with a high hardness (approximately 4200 HV), an oxidation resistance of up to 900 °C, and a thermal stability around 600 °C under working conditions. This type of coating would be useful in the high-speed dry milling of materials that have been hardened.
Chromium Aluminium Nitride Coatings (CrAlN)
CrAlN, like TiAlN, can be synthesized in a cubic B1-NaCl-type structure, in which Al replaces Cr up to a maximum solubility of 75% (x = 0.75). After reaching these values, its structure changes to a B4-hexagonal configuration of the Wurtzite type, as detailed in Figure 5.
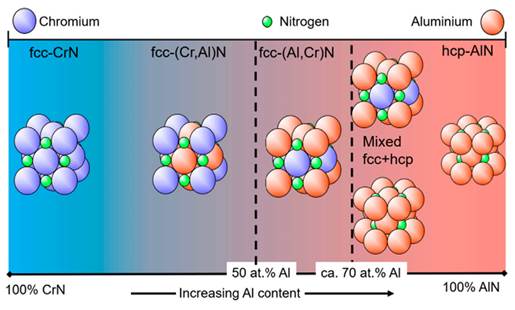
Figure 5 Different crystal structures of (CrAl)N and (AlCr)N coatings as a function of the Al content (Vetter et al., 2021).
This behavior is similar to that of AlTiN coatings, which implies that this type of cubic structure will exhibit similar behaviors despite the number of substitutional atoms in the crystal structure. However, is important to highlight that introducing atoms produces changes in lattice parameters, micro-deformations, residual stresses, and so on, modifying, among others, the mechanical properties, tribological performance, and corrosion resistance.
In this way, aluminum-chromium-nitride (AlCrN) coatings allow more aluminum particles to be added to its network without entailing structural deformations, which helps the formation of alumina (Al2O3), whose main function is heat dissipation. This results in a coating with excellent performance under extreme temperature conditions, which represents machining with higher cutting speeds, even with little or no lubrication (Wu et al., 2015).
Among its main applications are high-speed milling, the machining of special steel alloys, the coating of end mills and hobs for the manufacture of pinions, punches for cutting and punching sheets, and coating of injection molds, which increases oxidation resistance, all as a result of optimized process parameters and an optimum coating microstructure (Gong et al., 2017).
Results
As a summary, Table 1 shows the main properties of the above-described nitride coatings.
It seems that atoms incorporated in binary coatings, i.e., CrN or TiN, produce strengthening mechanisms of alloying processes including solid solution and dispersion strengthening. In the same way, tribological performance could be improved for aluminum and silicon additions through the already discussed mechanisms. Figure 6 depicts the relationship between hardness, friction coefficient, and oxidation resistance.
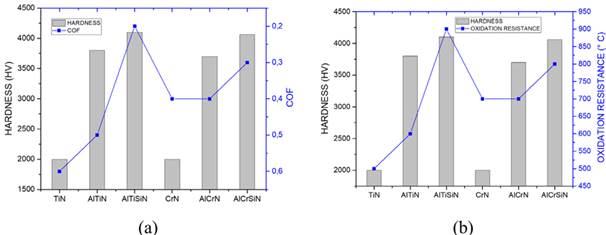
Figure 6 Comparison of hardness and COF (a) and hardness and oxidation resistance (b) of nitride-based PVD coatings.
In the case of TiN-based coatings, the addition of Al and Si elements could not only significantly improve the hardness and COF, but also yield a good oxidation resistance, as can be seen in Figure 6. On the other hand, CrN-based coatings exhibit a quite different behavior: the hardness and COF are not affected by adding aluminum, which evidences the close relationship between these mechanical properties. However, this behavior changes when silicon is added to the FCC crystal lattice; the hardness, COF, and oxidation resistance increase by around 9, 33, and 12.5%, respectively. In general, titanium-based coatings exhibit better properties when Al and silicon are incorporated as solid solution elements in the B1 NaCl lattice. Regarding quaternary coatings, the AlTiSiN coating exhibits a better high-temperature oxidation resistance than AlCrSiN.
Multicomponent coatings based on different metallic and non-metallic elements provide the benefit of individual components leading to further improvement of the coating properties. As mentioned before, these coatings show a unique B1-NaCl-type structure (under certain deposition conditions) and an increase in oxidation temperature to approximately 900 °C.
Some researchers (Ardila-Téllez et al., 2014; Z. F. Zhou et al., 2009), have compared these multicomponent coatings with well-known and traditional ones such as TiN and TiAlN. Z. F. Zhou et al. (2009) synthesized this type of hard coating through the reactive sputtering technique with an unbalanced magnetron on AISI M42 steel substrates, using Ti, Cr, and Al targets in a reactive sputtering system. The oxidation behavior at high temperatures was studied by static oxidation tests in the air at different temperatures (500-900 °C). The results showed that the (Ti, Cr, Al)N coating outperformed the other two conventional coatings in terms of oxidation resistance at high temperatures by forming a dense protective oxide layer of Cr2O3 and Al2O3. After oxidation at 900 °C, the (Ti, Cr, Al)N coating retained its crystalline structure, morphology, wear resistance, and hardness.
Yamamoto et al. (2005) and Z. F. Zhou et al. (2009) found that the microstructure with high Al contents (above 67 at% and up to 73 at%) remain unchanged after annealing at temperatures of 900 °C in air. Above this temperature, mixed cubic and hexagonal phases are formed. This high structural stability and excellent oxidation resistance are due to the high solubility of aluminum in the FCC-NaCl-type solid solution. Likewise, TiN and TiAlN, CrSiN, and CrAlSiN coatings have aroused great interest for their high hardness, oxidation resistance, and good performance at high temperatures (Chang and Hsiao, 2009; Chang and Lai, 2014; Wo et al., 2012). Furthermore, the addition of silicon to TiAlN has been shown to further improve hardness, withstand higher temperatures, and improve oxidation and wear resistance on substrates. Therefore, TiAlSiN is frequently employed as a coating for mainly dry machining applications.(Chang and Lai, 2014; Hwang et al., 2018; Siwawut et al., 2018).
The addition of new elements in the FCC crystal lattice to further improve the properties of coatings is still a challenge for researchers. An improvement in the tribological properties of CrTiAlSiN coatings has been recently found. These coatings were deposited by using a co-sputtering system. The deposition of the coating was conducted with 270 W pulsed-DC power applied to a TiAl target and RF power ranging between 70 and 170 W applied to a metallic Cr target. The results showed that the addition of Si promotes an increased hardness and elastic modulus. Moreover, mass loss in wear tests decreases as Si increases, which is due to the increase in hardness related to the microstructural refinement produced by the blocking of sliding bands by the grain boundaries (Estupiñán et al., 2021).
CrTiAlSiN, exhibits promising tribological and oxidation behavior that has not been properly studied to date. Cathodic arc evaporation (CAE) is a PVD technique that has been used to deposit CrTiAlSiN coatings involving AlSi and TiCr alloy targets achieving hardness above 40 GPa in these coatings (Kim et al., 2008; Luis Carlos Ardila-Tellez et al., 2014).
According to what has been discussed so far, it is clear that the increase in hardness is related to other properties of the coatings. Therefore, it is important to understand the mechanism and concepts involved in said property. The theory concerning an increase in hardness as a result of the microstructural refinement produced by the blocking of the sliding bands by the grain boundaries thus becomes relevant, as seen in Figure 7.
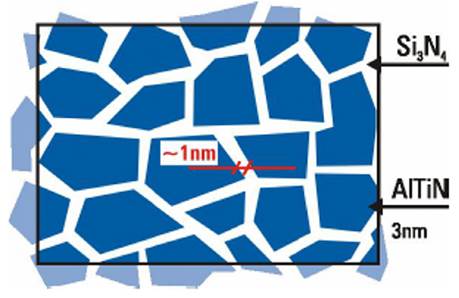
This high hardness is attributed to the existence of two phases: an nc-TiN or nc-AlTiN phase, and an amorphous Si3N4 matrix. In this system, the larger grains belonging to the nc-TiN or nc-AlTiN phases are embedded by an amorphous Si3N4 phase2 (Miletić et al., 2014b). The basic idea to explain the increase in hardness in these new coatings is based on Koehler's theory (Chu and Barnett, 1995), with some modifications. This theory is based on the fact that grains of a size between 10-20 nm could increase hardness, following the Hall-Petch law, when it is considered that the matrix phase is sufficiently hard and that the interfaces are fine and do not cause softening in the system.
Likewise, a remarkable increase in hardness (up to 4000 HV) was observed in the TiAlN coating, with titanium contents between 5 and 10 % atomic, which was obtained by employing the HiPIMs technique. These results were attributed to the fact that the solid solution exhibited greater strengthening due to the substitution of Cr atoms by Ti, as well as to the existence of a preferential orientation (111) and a denser microstructure. The layer with the best results was composed of Cr0.61Ti0.10Al0.29N (Lin et al., 2015).
Conclusions
Coatings based on MeSiN nanocomposites produced with plasma-assisted PVD processes have higher hardness and wear resistance, as well as lower friction and high corrosion resistance. The PVD technique is environmentally friendly and allows manufacturing multifunctional coatings with complex stoichiometry, high density, and adequate adhesion on metallic alloys. These achievements have resulted in the manufacturing of nanometer-scale materials that have allowed developing smaller and more efficient devices for several applications. This has created new industrial, technological, and scientific challenges, which implies acquiring state-of-the-art technology equipment that will allow for the generation of knowledge, new industry, and the training of human resources. This includes the development of green processes, new materials, and components at the nanometer scale.
Finally, the production of coatings of quaternary or quaternary metal nitrides allows the mechanical, thermal, and chemical properties of these materials to be adequately combined, thus allowing them to extend the durability of the materials and reducing energy consumption while protecting the environment. The degradation of a material due to corrosion and wear phenomena becomes a source of contamination, as it reduces its useful life. For example, a product of oxidation or pitting is the emission of substances that affect the quality of the air or water. Thus, the manufacture of MeSiN coatings is highly associated with reducing the consumption of energy and resources needed to replace or recover a degraded material. On the other hand, these coatings become fundamental in the metalworking industry, where the machining of a product for an aeronautical, automotive, or biomedical applications is based on material elimination with a cutting tool. It is important to highlight that the machining processes constitutes a significant economic cost of the product in industrialized countries. In the same way, reducing the wear and friction coefficient of a tool with MeSiN coatings has positive environmental effects due to the increased machining speed with minimal use of lubrication, which results in the reduced emission of toxic gases related to the evaporation of lubricating liquids.