Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Facultad Nacional de Agronomía Medellín
Print version ISSN 0304-2847
Rev. Fac. Nac. Agron. Medellín vol.65 no.2 Medellín June/Dec. 2012
USE OF SOIL MICROORGANISMS AS A BIOTECHNOLOGICAL STRATEGY TO ENHANCE AVOCADO (Persea americana)-PLANT PHOSPHATE UPTAKE AND GROWTH
USO DE MICROORGANISMOS DEL SUELO COMO ESTRATEGIA BIOTECNOLÓGICA PARA MEJORAR LA ABSORCIÓN DE FÓSFORO Y EL CRECIMIENTO DE PLANTAS DE AGUACATE (Persea americana)
Nelson Walter Osorio Vega1; Sandra Luz Serna Gómez2 and Beatriz Elena Montoya Restrepo3
1 Associated Professor. Universidad Nacional de Colombia - Sede Medellín - Faculty of Sciences - School of Biosciences. A.A. 3840, Medellín, Colombia <nwosorio@unal.edu.co>
2 Agronomic Engineer, Student in Geomorphology and Soils. Universidad Nacional de Colombia - Sede Medellín -Faculty of Sciences-, A.A. 3840, Medellín, Colombia <sandraluz571@gmail.com>
3 Agronomic Engineer, M. Sc. in Geomorphology and Soils. Universidad Nacional de Colombia - Sede Medellín -Faculty of Sciences-, A.A. 3840, Medellín, Colombia <bemontoy@gmail.com>
Received: October 07, 2011; Accepted: October 18, 2012.
Abstract. In recent years the area cultivated with avocado has increased significantly in Colombia and this trend is expected to continue in the coming years. However, most of the soils in the highlands of the Colombian Andean, that have been used to grow avocado, are strongly acidic and nutrient deficient, particularly phosphorus (P). To alleviate these problems, high rates of lime and P fertilizers are frequently applied, in many cases the applied dose are excessive. This increases production costs, produces plant nutrient imbalance in the plant, and increases the risk of polluting surface waters. A biotechnological alternative is the combined use of phosphate solubilizing microorganisms (PSM) and arbuscular mycorhizal fungi (AMF). This work describes a series of experiments carried out to isolate, characterize, and evaluate the effectiveness of AMF and PSM in increasing plant growth and P uptake of avocado plants. The results indicate that the mycorrhizal association is an effective alternative that can easily be established in the crops; however, the excessive use of P fertilizer in these soils may limit the benefits of these fungi on avocado plants. Currently, effective PSM of avocado rhizosphere have not been detected. In some cases, these microorganisms are absent in the avocado rhizosphere and in other cases their low capacity to solubilize P discourage their use in the crop.
Key words: Arbuscular mycorrhiza, phosphate solubilizing microorgansims, Andisols, fixation of phosphorus.
Resumen. En los últimos años el área cultivada con aguacate en Colombia ha crecido notoriamente y se espera que esta tendencia continúe en los próximos años. Sin embargo, muchos de los suelos de la zona andina utilizados para su siembra exhiben fuerte acidez y niveles deficientes de varios nutrientes, particularmente fósforo (P). Para corregir este problema se hacen aplicaciones de cales y fertilizantes fosfóricos solubles, en mucho casos las dosis aplicadas son altas. Esto eleva innecesariamente los costos de producción, crea desbalance nutricional en la planta y genera riesgos de contaminación ambiental. Una alternativa biotecnológica es el uso de microorganismos solubilizadores de fosfato (PSM) y hongos formadores de micorriza-arbuscular (AMF). En este trabajo se describe el aislamiento, caracterización y evaluación de la efectividad de AMF y PSM para mejorar la absorción de P por las plantas de aguacate. La asociación micorrizal ha probado ser una alternativa factible de implementar; sin embargo, el excesivo uso de fertilizantes fosfóricos puede limitar los beneficios de estos hongos en las plantas de aguacate. Hasta ahora no se han detectado PSM efectivos en la rizosfera de aguacate. En algunos casos, estos microorganismos están ausentes en la rizosfera del aguacate, y en otras circunstancias la baja capacidad para solubilizar P desestimula su uso en el cultivo.
Palabras clave: Micorriza arbuscular, microorganismos solubilizadores de fosfato, Andisoles, fijación de fósforo.
Currently, the cultivation of avocado (Persea americana) in Colombia has received much attention due to the high demand for this fruit in national and international markets (Bernal y Díaz 2008). The Colombian government has planned to increase the area with avocado plantations from the current 16,220 ha to 29,600 ha, in the year 2020. Also, the mean yield is expected to increase from 11.2 t ha-1 to 13.54 t ha-1, although the potential yield estimated is 30 t ha-1 (Bernal and Díaz, 2008). In order to reach international markets, good agricultural practices must be adopted (FONTAGRO, 2006). However, the intensive use of agrochemicals is a serious limit to satisfy this objective (ASOHOFRUCOL-FNFH, 2005; INFOAGRO, 2006).
A constraint for agricultural production in the Andean region, where avocado is intensively cultivated, is the low soil phosphate (P) availability (IGAC, 1988; González and Osorio, 2008). This occurs due to the high P sorption capacity of these soils (mostly Andisols) that reduce the amount of P available for plant roots (IGAC, 1988). The ion phosphate (H2PO4-) is strongly adsorbed onto the surface of soil minerals (allophane, goethite, kaolinite) in such a way that most of the applied P fertilizers (>90%) became non-available (Havlin et al., 1999; Miyasaka and Habte, 2001). Besides, free ions of aluminum and iron can also precipitate phosphate into insoluble compounds (Barber, 1995). As a consequence, high doses of soluble P fertilizers are regularly applied in avocado plantations (Serna et al., 2012); however, this increases the production costs, causes plant nutrient imbalances (micronutrient deficiencies), and increases the risk of pollution of surface water (Miyasaka and Habte, 2001; Montoya, 2007). In addition, the variation of the international price of soluble P fertilizer (in Colombia all P fertilizer is imported) is a challenge to the food production, as occurred in 2007 and 2008 when these increased by 355% (Hylton, 2008). For all these reasons, it is desirable increase the efficiency of soluble P fertilizers by using soil microorganisms (Osorio, 2012).
Given the role of arbuscular mycorrhizal fungi (AMF) and phosphate solubilizing microorganisms (PSM) (Thies and Grossman, 2006), these can be used as an attractive biotechnological strategy in avocado plantations. In some countries, such as Mexico, Chile, Brazil and United States (California), the use of AMF during the nursery stage of avocado has been successful in enhancing water and nutrient uptake (particularly P), and reduce the pos-transplant stress (Menge et al., 1978; Hernández, 2001). In Colombia, recently some authors has been investigating the effect of AMF inoculation in avocado cv. Villagorgona (Montoya, 2007), cv. Hass (Montañez, 2009), and cvs. Santana, Lorena and Común (Melo, 2011), whose findings indicate that mycorrhizal inoculation (with different AMF in each case) significantly promoted plant growth at the nursery. On the other hand, Bolaños and Saenz (2010) found that, in addition to the plant growth promotion, the mycorrhizal inoculation also reduced significantly the avocado root rot caused by some phytopathogens (Fusarium sp. and Phytohphtora cinnamomi); the reduction was higher when some biocontrol agents, such as Trichoderma, Paecilomyces or Pseudomonas fluorescent, were co-inoculated with the AMF.
The use of PSM can enhance the soil P availability by dissolving native or applied P compounds, particularly rock phosphates (RP) (Omar, 1998; Osorio and Habte, 2009). The advantage of using RP is that they are locally available in many countries (including Colombia) and are cheaper than P soluble fertilizers, unfortunately their low solubility discourage their use, but this can corrected by effective PSM (Msolla et al., 2005; Ojo et al., 2007). This strategy has been successfully proved by several authors in many plant species and soils (Sreenivasa and Krishnaraj, 1992; Whitelaw, 2000; Osorio and Habte, 2001; Barea et al., 2005a, 2005b); but in Andisols this may have a limited efficiency, because the P released can be retained by soil minerals (Bolan, 1991). To correct this limitation, the co-inoculation with PSM and AMF seems to be desirable because the mycorrhizal hyphae may absorb the released P by PSM and in this way improve plant P uptake (Osorio and Habte, 2009). However, the excessive use of P fertilizers, and their residual effects on soil P availability (Serna et al., 2012), may impair the benefits of AMF as reported in other crops (e.g., coffee (Coffee arabica), lulo (Solanum quitoense), tomato (Solanum lycopersicum)) (González, 2006; Montoya, 2007; Jaramillo and Osorio, 2009; González and Osorio, 2008). Currently, the presence of effective PSM in the rhizosphere of avocado plants has not been confirmed.
The aim of this study was to evaluate the use of AMF and PSM in avocado plants at the Northern and Eastern Antioquia, Colombia and to detect potential soil and crop factors that limit their effectiveness in enhancing plant P uptake and growth.
MATERIALS AND METHODS
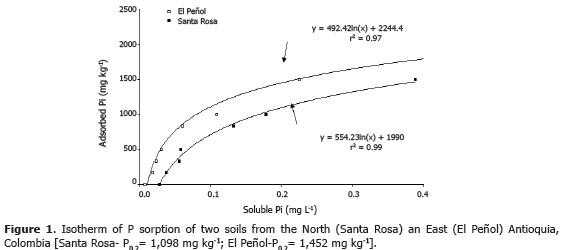
Soil phosphate sorption. Soil surface samples (A horizon, 0-20 cm) were collected from two places (Santa Rosa and El Peñol), which are representative of the two productive areas of avocado at Northern and Eastern Antioquia, respectively. To quantify the soil P fixation capacity, the method developed by Fox and Kamprath (1970) was used. Briefly, this consisted of the use of 3 g-soil (dry basis, sieved at<2 mm) that were transferred into plastic centrifuge tubes (volume of 50 mL), which received grading amounts (0-2,000 mg kg-1) of H3PO4 (SOLUFOS®) dissolved in 30 mL of 0.01 M CaCl2. Two drops of toluene were added into each tube to impair microbial activity. Then, the tubes were shaken in a reciprocal shaker at 100 rpm (30 min each 12 h) during six days at room temperature (25-27 ºC). After the incubation period, the tube and their contents were centrifuged for 15 min at 2,500 x g and the uppernatant was passed through a Whatman No. 1- filter paper. Soluble P concentration was measured by the molybdate-phosphate method (Murphy and Riley, 1962) in a spectrophotometer Genesys®-20 at 890 nm.
Monitoring soil microorganisms in avocado plantations. Seventy rhizosphere samples of three-to four-years-old avocado plants were collected from commercial plantations located at the towns of San Pedro, Entrerrios and Santa Rosa (Northern Antioquia) and Rionegro, El Retiro, La Ceja, El Carmen de Viboral and San Vicente (Eastern Antioquia). In each farm, fine roots and rhizosphere soil (200 g) of avocado plants were collected at 0-10 cm of depth, packed into plastic bags and later transported in polystyrene boxes to the Laboratory of Ecology and Environmental Conservation (LECA) at the Universidad Nacional de Colombia. Soil pH (1:2, water) and soluble P in 0.01 M CaCl2 (Olsen and Sommers, 1982) were also measured in these samples and already reported by Serna et al. (2012).
Mycorrhizal colonization. Samples of fine roots were cut in 1-cm fragments, rinsed with tap water and transferred into 50-mL plastic vials. 20-mL of KOH (10%) solution were added to clear root contents (Phillips and Hayman, 1970) and removed 24 h later. After that, root fragments were washed with tap water and then maintained in H2O2 (35%) solution for 30 min and washed again with tap water. Then, 20 mL of HCl (10%) solution were added and 5 min later removed (without water rinsing). 20-mL of trypan blue (0.15%, dissolved in glycerol and lactic acid) were added and the roots were left at room temperature for 48 h for staining (Brundrett et al., 1996). Next, the excess of trypan blue was removed by glycerol and lactic acid for 48 h. Root colonization was measured by the grid-line intercept method (Giovanneti and Mosse, 1980).
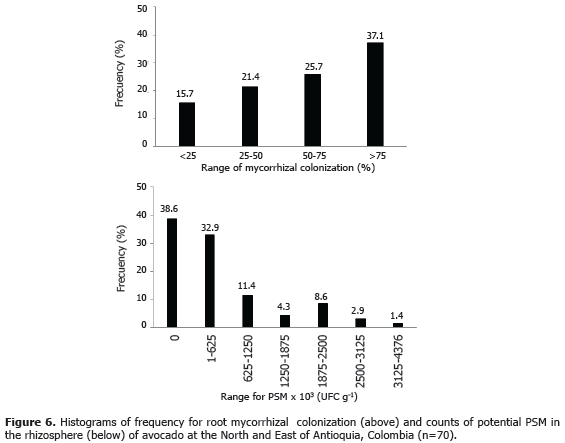
Rhizosphere PSM. Once in the laboratory, rhizosphere samples were hand-dispersed in order to destroy soil aggregates. Serial dilutions (10-1 - 10-6) were prepared with a 0.01 M CaCl2 solution. Aliquots of 100 mL were aseptically transferred into Petri dishes that contained a sterile (120 ºC, 0.1 MPa, 40 min) selective medium for PSM (Osorio and Habte, 2001). The medium contained per liter 1.0 g of NaCl, 0.2 g of CaCl2.2H2O, 0.4 g of MgSO4.7H2O, 1.0 g of NH4NO3, 10 g of glucose, 15 g of Agar, 3.5 g of Huila rock phosphate and 5 mL of bromothymol blue as a pH indicator. Petri dishes were incubated at 28 ºC for three to five days. Colony forming units were counted and some morphotypes were preliminary selected for further studies based on the presence of yellow halo around the colony, abundance and growth. After that, an in vitro test (Osorio, 2011) was conducted to quantify the capacity of the isolates to dissolve Huila RP as explain above. Selected microorganisms were transferred to Petri dishes with the same medium for purification and conservation at 4 ºC until their use. Frequency histograms were prepared for both variables: mycorrhizal colonization and rhizosphere PSM.
Identification of AMF spores. Spores of AMF were isolated from the 20 rhizosphere samples following the method described by Habte and Osorio (2001). Briefly, this consisted of wet-sieving aliquots (5 g, dry basis) of rhizosphere samples in nested series of sieves (500, 250, 100 and 50 mm of aperture pore). The AMF spores were collected from the last two sieves and observed under the microscopy for their morphological identification following the guides of INVAM (www.invam.caf.wvu.edu).
Test for the effectiveness of AMF. Crude inocula of these AMF species were taken from the collection of the LECA and multiplied separately during four months in a sterile substrate (soil: sand, 2:1 proportion) (autoclaved 120 ºC, 0.1 MPa, 2 h) with corn (Zea mays) and brachiaria grass (Brachiaria decumbens) as host plants. To evaluate the effectiveness of the inocula, the method developed by Habte and Manjunath (1987) was used; in this case, the host plant (Leucaena leucocephala) was used as indicator plant, given its very high mycorrhizal dependency and fast growth. These plants were grown in plastic pots (900 mL) that contained soil (pH 6.0 and soluble P concentration of 0.02 mg L-1), and were separately inoculated with each crude inoculum (spores, hyphae, infected roots in a soil-sand substrate) with 25 g/pot; each gram had 250-270 spores of the respective fungus.
Plants were grown during 45 days under greenhouse conditions and watered (if required) to maintain 50-60% of the maximal water hold capacity. Each week, 25 mL of the P-free Hoagland solution were added. The experimental design was completely randomized; treatments consisted of the three inoculations and an uninoculated control. Each treatment had three replicates. Response variables were shoot dry weight (oven-dried for 96 h at 60 ºC); shoot P content, measured by the molybdate-phosphate method developed by Murphy and Riley (1962), after obtain ashes from the shoot tissues (500 ºC, 3 h); and mycorrhizal colonization as explained above, but in this case H2O2 was not used and fucsin acid (0.15%) was used for staining roots.
Data were subjected to analysis of variance and mean separation through the multiple range test of Duncan; in both cases a significant P≤0.05 was used. Statistical analyses were conducted in the software Statgraphics Centurion version XV.
Effect of mycorrhizal inoculation on avocado plants. To this experiment an Andisol (volcanic ash soil) from the Experimental Forestry Station "Piedras Blancas" (Santa Elena, Medellín) was used. The soil was air-dried, sieved (<4 mm), autoclaved (120 ºC, 0.1 MPa, 2 h) and then transferred into plastic bags,
5.6 kg per bag. Soil pH was adjusted to 6.0 with calcium oxide (CaO); additionally, the following compounds were added: ammonium nitrate (173 mg kg-1), potassium nitrate (380 mg kg-1) and magnesium sulfate (3,280 mg kg-1). Then, KH2PO4 was applied in order to have three concentrations of soluble P in the soil solution (0.002, 0.02 y 0.2 mg L-1). The amount of P added was determined through a soil P sorption isotherm (Fox and Kamprath, 1970). Afterward, the soil was inoculated with 75 g of the crude inoculun of G. fasciculatum, which contained 43 infective propagules (most probable number; Porter, 1979). The uninoculated soil received 75 g of autoclaved inoculum and 20 mL of washings of the crude inoculum after removing AMF propagules by filtration with Whatman No. 1 filter paper.
Two months old seedlings of avocado (cv. "Villagorgona") were obtained from Profrutales (a commercial nursery at Palmira, Colombia) and transplanted into the treated soils according to treatments. Seedlings were grown for 150 days under greenhouse conditions and watered (if required) to maintain 50-60% of the maximal water hold capacity. Every two weeks, 25 mL of the P-free Hoagland solution were added per pot.
The experimental design was completely randomized; treatments had a factorial arrangement 3x2 (three levels of soil soluble P and two level of mycorrhizal inoculation-inoculated and uninoculated-). Each treatment had five replicates. Response variables were shoot dry weight (oven-dried for 96 h at 60 ºC), foliar P concentration measured in the foliar ashes (500 ºC, 3 h) by the molybdate-phosphate method (Murphy and Riley 1962) and mycorrhizal colonization measured as explained above.
Data were subjected to analysis of variance and mean separation through the LSD test; in both cases a significant P≤0.05 was used. Statistical analyses were conducted in the software Statgraphics Centurion version XV.
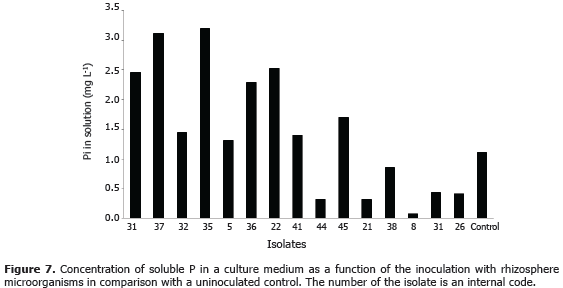
In vitro test for PSM efficiency. Selected colonies of PSM were purified with a sterile loop and aseptically transferred by duplicates into a 250-mL Erlenmeyer flask that contained the medium for PSM agar-free (Osorio and Habte, 2001). The flasks were continuously shaken in a reciprocal shaker at 150 rpm, 25 °C for 7 days; for comparison an uninoculated control was included. At the end of the incubation period, the medium pH was measured by introducing directly a H+- selective electrode. Also, the concentration of P in the medium was measured by the molybdate-phosphate method; for this purpose, the solution was removed by centrifugation for 15 min at 2,500 x g and the uppernatant was filtered by a Whatman® No. 43 filter paper (2 mm of aperture size).
RESULTS
Soil P sorption isotherm. The value of P0.2, amount of P (mg kg-1) required to achieve soil solution P concentration of 0.2 mg L-1 (Hue and Fox, 2010), was 1,098 and 1,452 mg kg-1 for Santa Rosa and El Peñol soils, respectively (Figure 1). These results allow classify the soil P sorption capacity as very-high (P0.2 > 1000 mg kg-1) (Juo and Fox, 1977). These levels of soil P sorption are associated with volcanic ash soils (Andisols) and are comparable with other studies conducted in this area (Osorio, 2008). This very high P fixation is responsible for the high requirements of P fertilizers in these soils, which is desirable to reduce by introducing other practices; for instance, (i) moderate liming to increase soil pH up to a range 5.5-6.0, (ii) use of organic amendments to reduce P sorption, (iii) use of AMF and PSM to reduce the soil soluble P requirement, (iv) improve placement of P fertilizers and (v) use of rock phosphates (Havlin et al., 1999).
Effectiveness of AMF associated to avocado plants. The AMF population in the rhizosphere had low diversity because this was dominated by three species Glomus fasciculatum (present in 35-45% of the samples), G. fistulosum (20-30%) and E. colombiana (40-50%), others morphotypes likely Glomus were less frequent and were not identified.
The origin of these AMF native or introduced was unclear; however, their presence has been registered in other soils and crops of Colombia (Guerrero, 1996; Guerrero et al., 1996; Cano, 1996; Cancio et al., 2005; Salamanca and Cano, 2005; Jaramillo and Osorio, 2009; Cabrales and Campo 2010; Daza and Osorio, 2011; Díaz et al., 2011). Furthermore, these AMF species are commonly used in commercial mycorrhizal inocula frequently applied at the Northern and Eastern Antioquia regions for avocado plantations.
On the other hand, the evaluation of these AMF from inocula allows us to separate them according to their effectiveness for enhancing plant performance (Figure 2). The results showed that the inoculum of G. fasciculatum was more effective in enhancing Leucaena plant P uptake and growth than the other AMF inocula studied. The increase in shoot dry weight with G. fasciculatum inoculation respect to uninoculated plants was of 75%, while the shoot P concentration was 3.75-fold higher and the shoot P content was 6.8-fold higher. Although the benefits on plant performance with G. fistulosum inoculation were lower than with G. fasciculatum, the shoot dry weight, P concentration and P content were 72%, 3-fold and 5.32-fold significantly higher than in the uninoculated plants. By contrast, the inoculum of E. colombiana was ineffective to enhance plant performance.
The technique used was very simple and useful to evaluate inoculum quality, as illustrated before by González and Osorio (2005), as well as to determine the mycorrhizal effectiveness of native AMF in soils (Jaramillo et al., 2004; Sierra, 2006). However, the results should be associate to the quality of a particular inoculum (AMF spores number and viability and other infective propagules) and not to the AMF species. As a conclusion, the crude inoculum of G. fasciculatum was the most effective and therefore used in further studies.
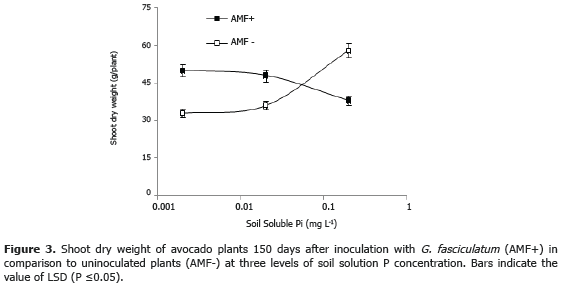
Effectiveness of G. fasciculatum on avocado plants. The results showed that the inoculation with this fungus significantly (P≤0.05) increased avocado plant growth (Figures 3 and 4). However, this effect depended on the concentration of P in the soil solution. For instance, at the level 0.002 mg L-1 the increased on plant growth was by 48%, at 0.02 mg L-1 this was by 35%. By contrast, at the level of 0.2 mg L-1 the plant response was negative to the inoculation because the shoot dry weight was significantly reduced by 26%.
These results probed that AMF inoculation is highly promissory to enhance plant performance on avocado plantations in soils with high P fixation, as those considered in this study (e.g., Santa Rosa and El Peñol). The use of these fungi may help to reduce the amount of P fertilizers and in this way reduce the production costs. The combination of AMF and RP application seems to be very attractive to evaluate in avocado plants since positive results have been observed in other soils and crops (Sreenivasa and Krishnaraj, 1992; Omar, 1998; Barea et al., 2005a 2005b; Osorio, 2008). Although AMF do not dissolve RP, they can absorb more efficiently the P release from the RP than the non-mycorrhizal roots (Bolan, 1991; Smith et al., 2003; Smith et al., 2011). When AMF take P, up reduce the concentration of Pi in the soil solution, favors the RP dissolution. In addition, the mycorrhizal association may promote the activity of the rhizosphere microorganisms capable of P dissolution (PSM) (Barea et al., 2005a, 2005b) and enhance plant P uptake by plant roots and/or mycorrhizal hypha (Whitelaw, 2000; Osorio and Habte, 2009).
Since the soil P availability can control the effects (from positive to negative) of AMF inoculation (Figure 3), it is worrisome that the avocado growers apply high rates of P fertilizers with the consequent effect on soil soluble P levels. Serna et al. (2012), found, in the same soil samples used here, that the soil soluble P ranged from 0.01 to 1.78 mg L-1; 47% of the rhizospheric soil samples exhibited levels of soluble P excessively high (0.25-1.69 mg L-1). According to our results, it is logic to expect that native or applied AMF would reduce plant growth under these conditions. In order to avoid these problems, it is recommendable to monitor the level of soluble P and mycorrhizal colonization of plant roots before making decisions on AMF inoculation. It must be clarified that the consideration made here are based only on the effects on plant nutrition and growth; it is possible that other benefits on water uptake and reduction on some plant diseases incidence (Allen, 1996; Smith and Read, 1997), must be considered in relation to the convenience of AMF inoculation under these conditions.
On the other hand, consistently with the treatments only those plants grown in inoculated soils developed the mycorrhizal association. The value of this variable fluctuated between 57 to 77%, and it was not significantly affected by the level of soluble P. However, at the two lowest levels (0.002 and 0.2 mg L-1) there was abundance of vesicles and arbuscules in the roots, while at the highest level of soil soluble P (0.2 mg L-1) the roots were exclusively colonized by intraradical hyphae without vesicles and arbuscules. This should be a matter for further research.
Monitoring soil microorganisms in avocado plantations.
Mycorrhizal colonization of avocado plant roots. In avocado plantations the values of mycorrhizal colonization were variable, these fluctuated between 0 to 90% (Figures 5a and 6). The significance of this should be considered in relation to the level of soil P available. For instance, at low soil soluble P concentration (<0.05 mg L-1) the mycorrhizal association promotes plant growth; but at higher concentrations it decreases (Montoya, 2007). Recently, in the same samples studied here, Serna et al. (2012), found that more than 60% of the soil samples had soil soluble P>0.2 mg L-1. Under such conditions, the presence of AMF in the roots may have negative effects on plants. This indicates that to determine the convenience of the mycorrhizal inoculation in avocado plantations, it is necessary to monitor the levels of soil P availability as well as the presence of infective mycorrhizal propagules in the soil (Sierra, 2006).
PSM in the avocado rhizosphere. The interpretation of PSM plate-counts from the rhizosphere has some limitations. First, there is no certainty about which is the adequate value of PSM-population at the rhizosphere. Second, the presence of these microorganisms is not a guarantee to improve soil P availability since either competition/antagonism among microbes and/or the soil P sorption capacity may limit the effectiveness of them to enhance plant P uptake (Osorio, 2011). Then, their presence in the rhizosphere only indicates that their potential role in enhancing soil P availability may occur; on the other hand, their absence certainly indicates that the microbial solubilization of P will not happen. Even so, we should not forget that the counts conducted only detect those rhizospheric PSM cultivable, and all discussion just made here is irrelevant. Thus, in 39% of the soil samples studied, where PSM were not detected, the mechanism of microbial P solubilization perhaps does not occur. In the rest of the samples (61%) PSM were found and likely they contribute to enhance soil P availability to plant roots. Some other factors might help to PSM effectiveness such as the mycorrhizal association because the fungal hyphae would absorb the P released by PSM. In addition, the presence of NH4+ favors the dissolution of RP, while NO3- is inhibitory (Osorio, 2008).
In vitro test for PSM efficiency. The in vitro test allow us to detect that some microorganisms produced acidity in the medium and thus reducing pH down to 3.1 (Figure 7), which is required to dissolve RP (Welch et al., 2002; Oberson et al., 2006; Yusdar et al., 2007; Hamdali et al., 2010; Bashan et al., 2012). Despite that the concentration of P in solution was relatively low, if compared with other effective PSM's (Aspergillus niger and Mortierella sp.) used with the same test (Osorio and Habte, 2009, Zaidi et al., 2009; Bashan et al., 2012), at pH of 3.1 a value of soluble P around 15 mg L-1 is expected (Osorio, 2012); however, the value found was only 3.2 mg L-1, although this level was significantly higher than that in the uninoculated control, clearly it suggests a low effectiveness (Osorio, 2008). Likely, the microbes did solubilize RP but most of it was immobilize in their cells. This transformation of P, from insoluble RP toward microbial-P, is not necessarily unfavorable for plants because this pool is later subjected to decomposition by other microorganisms, leaving P available for root uptake (Joergensen and Wichern, 2008; Posada et al., 2012).
The lack of effective PSM's in the rhizosphere of avocado plants lead us to consider the use of effective PSM obtained from other plant rhizosphere. This approach is valid because the microbial solubilization of P-compunds does not require a specific relationship between plant-microbe as other interactions (e.g., legume-rhizobio). In this way, the relationship plant-PSM is quite similar to that observed between plant-PGPR (Bashan and Holguin, 1998). It has been proposed that an effective PSM must be (i) a good competitor for resources in the rhizosphere (especially carbonaceous compounds), (ii) efficient colonizer of the rhizosphere and/or the rhizoplane, and (iii) an active producer of organic acids (di and tri-carboxylic acids such as oxalic acid and citric acid) (Osorio, 2011; Bashan et al., 2012). Also, the PSM effectiveness will be higher in the presence of mycorrhizal plants, particularly if they grow in soils with high P sorption capacity as those considered in this study.
CONCLUSIONS
In general, the soils from the North and East Antioquia exhibited a very high P sorption capacity (P0.2>1.000 mg kg-1), which is responsible of the low soil P availability observed in these soils under natural conditions (soil soluble P<0.05 mg L-1), limiting the plant productivity. To prevent plant P deficiency, avocado growers apply high rates of soluble P fertilizers; in many cases the amount applied is excessive and thus soil soluble P increase at levels unnecessarily high (>0.2 mg L-1). This increases in production costs, raises environmental concerns, and creates plant nutrient imbalance. A valid strategy is the use of arbuscular mycorrizal fungi (AMF); in this study the fungus Glomus fasciculatum was capable of increasing avocado plant growth at low levels of soil soluble P (0.002 y 0.02 mg L-1), but the effect became negative at high levels (0.2 mg L-1). The use of phosphate solubilizing microorganisms (PSM) is quite attractive given its potential to dissolve rock phosphate and native soil P compounds. However, the excessive levels of soil soluble P detected in avocado plantations threaten the proper functioning of the mycorrhizal association, in many cases this association was absent of the plant system. The counts of PSM in the rhizosphere of avocado were quite variable; in some cases, they were not detected and those isolated were ineffective in dissolving RP.
ACKNOWLEDGEMENT
This work was financially supported by the Direction of Research of the Universidad Nacional de Colombia. We want to thank the criticism of the reviewers whose contributions improved the document. We also thank Claudia Alvarez for reading the final version.
BIBLIOGRAPHY
Allen, M. 1996. The ecology of arbuscular mycorrhizas: a look back into the 20 th century and a peek into the 21st. Mycological Research 100(7): 769-782. [ Links ]
ASOHOFRUCOL-FNFH. 2005. Convocatoria para el proyecto "Programa de desarrollo de proveedores: Implementación y certificación de buenas prácticas agrícolas en fincas productoras de frutales de clima frío exportables en el departamento de Cundinamarca". Bogotá. Mayo 2005. [ Links ]
Barber, S. 1995. Soil nutrient bioavailability: a mechanistic approach. Willey Interscience, New York. 398 p. [ Links ]
Barea, J.M., R. Azcón and C. Azcón. 2005a. Interaction between mycorrhizal fungi and bacteria to improve plant nutrient cycling and soil structure. pp. 195-212. In: Buscot, F. and A. Varma (eds.). Microorganisms in soils: roles in genesis and functions. Springer-Verlag, Heidelberg. 419 p. [ Links ]
Barea, J.M., D. Werner, C. Azcón and R. Azcón. 2005b. Interaction of arbuscular mycorrhiza and nitrogen fixing simbiosis in sustainable agricultura. In: Werner, D. and W.E. Newton (eds.). Nitrogen Fixation in Agriculture forestry, Ecology and the Environment. Kluwer Academic Publishers, Netherlands. 349 p. [ Links ]
Bashan, Y. and G. Holguin. 1998. Proposal for the division of plant growth - promoting rhizobacteria into two classifications: biocontrol - PGPB (plant growth - promoting bacteria) and PGPB. Soil Biology and Biochemistry 30(9): 1225 - 1228. [ Links ]
Bashan, Y., A.A. Kamnev, and de-Bashan, L.E. 2012. Tricalcium phosphate is inappropriate as a universal selection factor for isolating and testing phosphate-solubilizing bacteria that enhance plant growth: a proposal for an alternative procedure. Biology and Fertility of Soils. In: http://link.springer.com/article/10.1007%2Fs00374-012-0737-7. 15 p.: consulta: noviembre 2012. [ Links ]
Bernal, J. y C. Díaz. 2008. Tecnología para el cultivo del aguacate. Manual Técnico No. 5. CORPOICA, Rionegro. 241 p. [ Links ]
Bolan, N. 1991. A critical review on the role of mycorrhizal fungi in the uptake of phosphorus by plants. Plant and Soil 134: 189-293. [ Links ]
Bolaños, M.M. y E.P. Sáenz. 2010. Efecto de hongos benéficos sobre la nutrición y sanidad de aguacate. http://www.corpoica.org.co. http://www.corpoica.org.co/sitioweb/Archivos/oferta/ArticuloEfectoHongosbenficosnutaguacate.pdf. 10 p; consulta: noviembre 2012. [ Links ]
Brundett, M., N. Bougher, B. Dell, T. Grove, and N. Malajczuk. 1996. Working with mycorrhizas in forestry and agriculture. Australian Centre for International Agricultural Research. Monograph No. 32. 374 p. [ Links ]
Cabrales, E.M. y R. Campo. 2010. Hongos micorrizo arbusculares asociados a los principales cultivos de los suelos sulfatados ácidos de Córdoba, Colombia. Suelos Ecuatoriales 40(1): 57-61. [ Links ]
Cancio, M.A., G.L. Madrid, R.O. Campo y E.M. Combatt. 2005. Efecto del encalamiento en la diversidad de micorrizas vesiculo-arbusculares y nemátodos asociados al maíz en suelos sulfatados ácidos en el departamento de Córdoba, Colombia. Suelos Ecuatoriales 35(2): 24-27. [ Links ]
Cano, C.A. 1996. Manejo de un banco de germoplasma de hongos formadores de micorriza arbuscular (MA). pp. 125-143. En: Guerrero E. (ed.). Micorrizas. Recurso Biológico del Suelo, Fondo FEN, Bogotá. 208 p. [ Links ]
Daza, P.C. y N.W. Osorio. 2011. Promoción de crecimiento y absorción de fósforo de plántulas de Leucaena por un hongo micorrizal en un suelo degradado por minería de aluvión. Suelos Ecuatoriales 41(2): 144-149. [ Links ]
Díaz, D.C., O.L. Hernández, L.M. Zárate y J.F. García. 2011. Efecto de la aplicación de diferentes fertilizantes sobre la actividad micorrizal en gulupa. Suelos Ecuatoriales 41(2): 115-121. [ Links ]
FONTAGRO. 2006. Métodos no destructivos y marcadores moleculares para la determinación de fechas apropiadas de cosecha de cinco variedades de aguacate. Fondo Regional de Tecnología Agropecuaria. Proyecto FTG 24/01. En: http://www.fontagro.org/en/convocatorias/convocatoria-2001/propuestas-formales/m%C3%A9todos-no-destructivos-y-marcadores-moleculares. Consulta: abril 2010. [ Links ]
Fox, R. and E. Kamprath. 1970. Phosphate sorption isotherms for evaluating the phosphate requirements of soils. Soil Science Society of America Journal 34(6): 902-907. [ Links ]
Giovannetti, M. and B. Mosse. 1980. An evaluation of techniques for measuring vesicular-arbuscular mycorrhizal infection in roots. New Phytologist 84: 489-500. [ Links ]
González, O. 2006. Determinación de la dependencia micorrizal de frutales de clima frio. Tesis Magister en Geomorfología y Suelos, Universidad Nacional de Colombia, Medellín. 158 p. [ Links ]
González, O. y N.W. Osorio. 2005. Evaluación de la efectividad de inóculos micorrizales. Suelos Ecuatoriales 35(2): 19-23. [ Links ]
González, O.A. y N.W. Osorio. 2008. Determinación de la dependencia micorrizal del lulo. Acta Biológica Colombiana 13(2): 63-174. [ Links ]
Guerrero, E. 1996. Micorriza: Fundamentos y estado de arte. pp. 3-46. En: Guerrero E. (ed.). Micorrizas. Recurso Biológico del Suelo, Fondo FEN, Bogotá. 208 p. [ Links ]
Guerrero, E., C. Rivillas y E.L. Rivera. 1996. Perspectivas de manejo de la micorriza arbuscular en ecosistemas tropicales. pp. 181-206. En: Guerrero E. (ed.). Micorrizas. Recurso Biológico del Suelo, Fondo FEN, Bogotá. 208 p. [ Links ]
Habte, M. and A. Manjunath. 1987. Soil solution phosphorus status and mycorrhizal dependency in Leucaena leucocephala. Applied and Environmental Microbiology 53: 797-801. [ Links ]
Habte, M. and N.W. Osorio. 2001. Arbuscular Mycorrhizas: Producing and applying Arbuscular mycorrhizal inoculum. University of Hawaii, Honolulu, 47 p. [ Links ]
Hamdali, H., A. Smirnov, C. Esnault, Y. Ouhdouch and M.J. Virolle. 2010. Physiological studies and comparative analysis of rock phosphate solubilization abilities of actinomycetales originating from Moroccan phosphate mines and of Streptomyces lividans. Applied Soil Ecology 44(1): 24-31. [ Links ]
Havlin, J., J. Beaton, S.L. Tisdale, and W. Nelson. 1999. Soil fertility and fertilizers. An introduction to nutrient management. Prentice Hall, Upper Saddle River, NJ. 499 p. [ Links ]
Hernandez C. 2001. Efecto del hongo micorrizal (Glomus intraradices Schenk and Smith) en el crecimiento del portainjerto Mexícola (Persea americana Mill) cultivado bajo 5 tratamientos de fertilización. Facultad de Agronomía, Universidad Católica de Valparaíso, Chile, 95 p. [ Links ]
Hue, N.V. and Fox R. 2010. Predicting plant phosphorus requirements for Hawaii soils using a combination of phosphorus sorption isotherms and chemical extraction methods. Communications in Soil Science and Plant Analysis 41: 133-143. [ Links ]
Hylton, K. 2008. Visión global del mercado de fertilizantes. pp. 7-18. En: Jiménez, F. (ed.) Actualización en fertilización de cultivos y uso de fertilizantes. Sociedad Colombiana de la Ciencia del Suelo, Bogotá 236 p. [ Links ]
INFOAGRO. 2006. El cultivo del aguacate. En: http://www.infoagro.cons/frutas/frutas.tropicales/aguacate.htm. 11 p.; consulta: septiembre 2007. [ Links ]
Instituto Geográfico Agustín Codazzi (IGAC). 1988. Suelos y bosques de Colombia. Subdirección Agrológica, Bogotá. 127 p. [ Links ]
Jaramillo, S. and N.W. Osorio. 2009. Mycorrhizal dependency of coffee seedlings at three levels of soil solution phosphorus. Suelos Ecuatoriales 39(1): 143-147. [ Links ]
Jaramillo, S.P., J.M. Silva y N.W. Osorio. 2004. Potencial simbiótico y efectividad de hongos micorrizo arbusculares de tres suelos sometidos a diferentes usos. Revista Facultad Nacional de Agronomía Medellín 57(1): 2203-2214. [ Links ]
Joergensen, R.G. and F. Wichern. 2008. Quantitative assessment of the fungal contribution to microbial tissue in soil. Soil Biology and Biochemistry 40(12): 2977-2991. [ Links ]
Juo, A.S.R. and R.L. Fox. 1977. Phosphate sorption characteristics of some bench-mark soils of West Africa. Soil Science 124(6): 370-376. [ Links ]
Melo, Y.P. 2011. Respuesta de la inoculación de micorrizas en plántulas de aguacate Persea americana Mill. variedad Hass en diferentes sustratos. Tesis Maestría. Universidad Nacional de Colombia, Palmira. 136 p. [ Links ]
Menge J., R. Davis, E. Johnson, and G. Zentmyer. 1978. Mycorrhizal fungi increase growth and reduce transplant injury in avocado. California Agriculture 32(4): 6-7. [ Links ]
Miyasaka, S. and M. Habte. 2001. Plant mechanisms and mycorrhizal symbioses to increase phosphorus uptake efficiency. Communications in Soil Science and Plant Analysis 32(7-8): 1101-1147. [ Links ]
Montañez, B.I. 2009. Efecto de la micorrización en plantas de aguacate (Persea americana L.) durante la fase de vivero en suelos provenientes de los Llanos Orientales. Tesis Maestría. Universidad Nacional de Colombia, Bogotá. 145 p. [ Links ]
Montoya, B. 2007. Determinación de la dependencia micorrizal del aguacate. Tesis Magister en Geomorfología y Suelos. Universidad Nacional de Colombia, Medellín. [ Links ]
Msolla, M.M., J.M.R. Semoka, and O.K. Borggaard. 2005. Hard Minjingu phosphate rock. An alternative P source for maize production on acid soils in Tanzania. Nutrient Cycling in Agroecosystems 72: 299-308. [ Links ]
Murphy, J. and J.P. Riley. 1962. A modified single solution method for the determination of phosphate in natural waters. Analytica Chimica Acta 27: 31-36. [ Links ]
Oberson, A., E.K. Bunemann, D.K. Friesen, I.M. Rao and P.C. Smithson. 2006. Improving phosphorus fertility in tropical soils through biological interventions. pp. 531-546. In: Uphoff N. (ed.). Biological approaches to sustainable soil systems. CRC Press, Boca Raton, Fl. 764 p. [ Links ]
Ojo, O.D., A.A. Kintomo, E.A. Akinride, and M.O. Akoroda. 2007. Comparative effect of phosphorus sources for grain amaranth production. Communications in Soil Science and Plant Analysis 38:35-55. [ Links ]
Olsen, S.R. and L.E. Sommers. 1982. Phosphorus. pp. 403-430. In: A.L. Page and D.R. Keeney (eds.) Methods of soil analysis. Part 2. Chemical and Microbiological Properties. American Society of Agronomy and Soil Science Society of America, Madison, WI. 1159 p. [ Links ]
Omar, S.A. 1998. The role of rock-phosphate-solubilizing fungi and vesicular-arbuscular-mycorrhiza (VAM) in growth of wheat plants fertilized with rock phosphate. World Journal of Microbiology and Biotechnology 14:211-218. [ Links ]
Osorio, N.W. and M. Habte. 2009. Strategies for utilizing arbuscular mycorrhizal fungi and phosphate-solubilizing microorganisms for enhanced phosphate uptake and growth of plants in the soils of the tropics. pp. 325-351. In: Khan, M.S., A. Zaidi and J. Musarrat (ed.). Microbial strategies for crop improvement, Springer-Verlag, Berlin. 358 p. [ Links ]
Osorio, N.W. 2008. Effectiveness of microbial solubilization of phosphate in enhancing plant phosphate uptake in tropical soils and assessment of the mechanisms of solubilization. Thesis Ph.D. University of Hawaii. Manoa. 391 p. [ Links ]
Osorio, N.W. 2011. Effectiveness of phosphate solubilizing microorganism in increasing plant phosphate uptake and growth in tropical soils. pp. 65-80. In: Maheshwari, D.K. (ed.). Bacteria in agrobiology: Plant nutrient management. Springer-Verlag, Berlin. 345 p. [ Links ]
Osorio, N.W. 2012. Manejo de Nutrientes en Suelos del Trópico. Universidad Nacional de Colombia, Medellín. 339 p. [ Links ]
Osorio, N.W. and M. Habte. 2001. Synergistic influence of an arbuscular mycorrhizal fungus and P solubilizing fungus on growth and plant P uptake of Leucaena leucocephala in an Oxisol. Arid Land Research and Management 15: 263-274. [ Links ]
Posada, R.H., M. Sánchez, E. Sieverding, K. Aguilar y G.P. Heredia. 2012. Relaciones entre los hongos filamentosos y solubilizadores de fosfatos con algunas variables edáficas y el manejo de cafetales. Revista de Biología Tropical 60(3): 1075-1096. [ Links ]
Salamanca, C.R. y C.A. Cano. 2005. Efecto de las micorrizas y el sustrato en el crecimiento vegetativo y nutrición de cuatro especies frutales y una forestal en fase de vivero. Suelos Ecuatoriales 35:5-11. [ Links ]
Serna, S.L., B. Montoya y N.W. Osorio. 2012. Monitoreo del pH y fósforo soluble en la rizosfera de árboles de aguacate en el Norte y Oriente Antioqueño. Suelos Ecuatoriales 42(2): 93-97. [ Links ]
Sierra, J.A. 2006. Dependencia micorrizal de cuatro especies forestales y efectividad micorrizal de Andisoles en función de su uso y manejo. Tesis Magister en Bosques y Conservación Ambiental. Universidad Nacional de Colombia, Medellín. 84 p. [ Links ]
Phillips, J.M. and D.S. Hayman. 1970. Improved procedures for clearing and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Transactions of the British Mycological Society 55: 158-161. [ Links ]
Porter, W. 1979. The "Most Probable Number" method for ennumerating infective propagules of vesicular arbuscular micorrizal fungi in soil. Australian Journal of Soil Research 17: 515-519. [ Links ]
Smith, S. and D. Read. 1997. Mycorrhizal Symbiosis. Academic Press, London, UK. 605 p. [ Links ]
Smith, S.E., F.A. Smith and I. Jakobsen. 2003. Mycorrhizal fungi can dominate phosphate supply to plants irrespective of growth responses. Plant Physiology 133(1): 16-20. [ Links ]
Smith, S.E., I. Jakobsen, M. Grønlund, and F. A. Smith. 2011. Roles of arbuscular mycorrhizas in plant phosphorus nutrition: interactions between pathways of phosphorus uptake in arbuscular mycorrhizal roots have important implications for understanding and manipulating plant phosphorus acquisition. Plant Physiology 156(3): 1050-1057. [ Links ]
Sreenivasa, M. and P.U. Krishnaraj. 1992. Synergistic interaction between VA mycorrhizal fungi and a phosphate solubilizing bacterium in chili (Capsicum annuum). Zentralblatt für Mikrobiologie 147(1-2): 126-130. [ Links ]
Thies, J. and J.M. Grossman. 2006. The soil habitat and soil ecology. pp. 59-78. In: Uphoff N. (ed.). Biological approaches to sustainable soil systems. CRC Press, Boca Raton, FL. 764 p. [ Links ]
Welch, S., A.E. Taunton and J.F. Banfiled. 2002. Effect of microorganisms and microbial metabolites on apatite dissolution. Geomicrobiology Journal 19: 343-367. [ Links ]
Whitelaw, M.A. 2000. Growth promotion of plants inoculated with phosphate-solubilizing fungi. Advances in Agronomy 69: 99-151. [ Links ]
Yusdar, H., A.R. Anuar, M.M. Hanafi and H. Azizah. 2007. Analysis of phosphate rock dissolution determining factors using principal component analysis in some acid Indonesian soils. Communication in Soil Science and Plant Analysis 38(1-2): 273-282. [ Links ]
Zaidi, A., M.S. Khan, M. Ahemad, M. Oves and P.A. Wani. 2009. Recent advances in plant growth promotion by phosphate-solubilizing microbes. pp. 23-50. In: Khan M.S., A. Zaidi and J. Musarrat (ed.). Microbial strategies for crop improvement. Springer-Verlag, Berlin. 358 p. [ Links ]