Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Facultad Nacional de Agronomía Medellín
Print version ISSN 0304-2847
Rev. Fac. Nac. Agron. Medellín vol.65 no.2 Medellín June/Dec. 2012
PROTEIN AND MINERAL METABOLITES FOR DAIRY COWS DURING THE TRANSITION PERIOD UNDER TROPICAL CONDITIONS
METABOLITOS PROTEICOS Y MINERALES EN VACAS LECHERAS EN PERÍODO DE TRANSICIÓN BAJO CONDICIONES TROPICALES
Rómulo Campos Gaona1; Katherine García Alegría2; Erika Andrea Hernández3 and Leónidas Giraldo Patiño4
1 Associate Professor. Universidad Nacional de Colombia - Sede Palmira - Faculty of Agricultural Sciences. Carrera 32 No 12 - 00 Chapinero, Vía Candelaria Palmira - Valle del Cauca - Colombia. <rcamposg@unal.edu.co>
2 Zootechnical. Universidad Nacional de Colombia - Sede Palmira - Faculty of Agricultural Sciences. Carrera 32 No 12 - 00 Chapinero, Vía Candelaria Palmira - Valle del Cauca - Colombia.<kgarciaa@unal.edu.co>
3 Zootechnical, Magíster Student in Animal Production. Universidad Nacional de Colombia - Sede Palmira - Faculty of Agricultural Sciences. Carrera 32 No 12 - 00 Chapinero, Vía Candelaria Palmira - Valle del Cauca - Colombia. <eahernandez@unaledu.co>
4 Assistant Professor. Universidad Nacional de Colombia - Sede Palmira - Faculty of Agricultural Sciences. Carrera 32 No 12 - 00 Chapinero, Vía Candelaria Palmira - Valle del Cauca - Colombia. <lgiraldopa@unal.edu.co>
Received: May 9, 2012; Accepted: November 6, 2012.
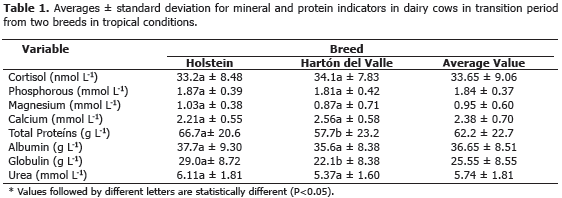
Abstract. In bovines, the transition period goes from the last three prepartum weeks to the first three postpartum weeks. This period is characterized by the metabolic adaptation of lipids, carbohydrates, minerals and proteins that try to balance the low intake of dry matter and the beginning of the secretion of milk. These changes are regulated by corticosteroids that are released at the time of birth. Some metabolites show noticeable changes in their blood concentration during this stage, which can lead to sanitary, reproductive and productive problems. The objective of the present work was to analyse the homeostasis in the transitional period, by determining the concentration of protein indicators, and some macrominerals, in dairy cows from two breeds under low-tropic conditions. The work was carried out in two commercial farms with similar management conditions in the Colombian south eastern region, in a agro-ecological zone of tropical dry forest according to Holdridge (1987), situated at 1,050 masl, with an average temperature of 23 ºC and an annual rainfall of 1200 mm bimodally distributed. The feed consisted of Bermuda grass (Cynodon nlemfuensis). The present work used seven Holstein cows and seven Hartón del Valle cows, from which blood samples were taken by coccygeal venepuncture in vacuum tubes, with heparin as anticoagulant and without anticoagulant, during the transition period, and up to the 60th day after delivery. A total of 156 samples were analysed. The following values were determined: total protein, albumin, globulin and urea; macro-minerals such as Ca, P and Mg and the cortisol hormone. The average values found for the Holstein breed were: total protein 66.7 g L-1, albumin 37.6 g L-1, globulins 27.7 g L-1, urea 6.11 mmol L-1, Ca 2.21 mmol L-1, P 1.87 mmol L-1, Mg 1.03 mmol L-1 and cortisol 33.2 nmol L-1. For the Hartón del Valle breed the following average values were found: total protein 57.7 g L-1, albumin 35.6 g L-1, globulins 20.3 g L-1, urea 5.37 mmol L-1, Ca 2.56 mmol L-1, P 1.81 mmol L-1, Mg 0.81 mmol L-1 and cortisol 34.1 nmol L-1. The only significant differences between the Hartón del Valle and the Holstein breeds were total protein, globulin and the serum cortisol levels.
Key words: Proteins, Holstein, Hartón. homeostasis, protein indicators.
Resumen. En bovinos, el período de transición comprende las tres últimas semanas preparto y las primeras tres posparto. Es caracterizado por adaptaciones en el metabolismo de lípidos, carbohidratos, minerales y proteínas buscando compensar el bajo consumo de materia seca y el inicio de la secreción láctea; estos cambios son regulados por corticosteroides liberados al parto. Algunos metabolitos presentan en esta etapa cambios manifiestos en su concentración sanguínea que puede llevar a problemas sanitarios, reproductivos y productivos. El objetivo fue analizar la homeostasis en el período de transición a través de la determinación de la concentración de indicadores proteicos, y de algunos macrominerales, en vacas lecheras de dos grupos raciales en condiciones de trópico bajo. El trabajo se realizó en dos fincas comerciales con similares condiciones de manejo, en la región suroriental de Colombia, en zona agroecológica de bosque seco tropical según Holdridge (1987), a 1.050 msnm., temperatura promedio de 23 ºC y precipitación anual de 1.200 mm distribuida en forma bimodal; la alimentación se basó en pasto Estrella (Cynodon nlemfuensis). Se utilizaron siete vacas Holstein y siete vacas Hartón del Valle, de las cuales se obtuvieron muestras de sangre recolectadas por venipunción coccígea en tubos a vacío, con heparina como anticoagulante y sin anticoagulante, durante el período de transición y hasta el día 60 del posparto; se analizaron un total de 156 muestras. Se determinó: proteína total, albúmina, globulinas y urea; los macrominerales Ca, P y Mg, y la hormona cortisol. Los valores promedios encontrados fueron: proteínas totales 66,7 g L-1, albúmina 37,6 g L-1, globulinas 27,7 g L-1, urea 6,11 mmol L-1, Ca 2,21 mmol L-1, P 1,87 mmol L-1, Mg 1,03 mmol L-1 y cortisol 33,2 nmolL-1, para la raza Holstein. Para el grupo Hartón se encontró: proteínas totales 57,7 g L-1, albúmina 35,6 g L-1, globulinas 20,3 g L-1, urea 5,37 mmol L-1, Ca 2,56 mmol L-1, P 1,81 mmol L-1, Mg 0,81 mmol L-1 y cortisol 34,1 nmol L-1. Entre las razas Hartón del valle y Holstein sólo se presentaron diferencias significativas para proteínas totales, globulinas y en los niveles séricos de cortisol.
Palabras clave: Proteínas, Holstein, Hartón, homeostasis, indicadores proteicos.
The transition period for dairy cattle goes from the last three prepartum weeks to the first three post partum weeks. This period is characterised by a reduction in the intake of dry matter and an increase in energy requirements of the cow for milk production (Huzzey et al., 2005).
The metabolic adaptation processes during the transition period include a complex interaction between the energy supply, the protein indicators and the necessary minerals to guarantee structural functions, the restart of reproductive activity and the dynamism of the immune system. This generates homeostasis, which allows production and preservation of health (Leblanc, 2010).
Peripartum cows with inadequate calcium (Ca), phosphorus (P) and magnesium (Mg) concentrations in their blood can experience loss of muscular and nerve function, decrease in food intake, low ruminal activity and decline of intestinal motility (Goff, 2006). Besides the physiological stress inherent to this period, the immune system is affected which means that lactating cows are liable to infectious diseases, such as mastitis and metritis, that subsequently alters productive and reproductive functions (Andrieu, 2008). There is little known about the role played by minerals in these metabolic processes (Wilde, 2006) and there is a need to quantify these requirements according to the productive stage where the animal is at (Goff, 2006).
The Ca, P and Mg requirements, depend on the weight of the animal, the production and composition of milk, and the pregnancy stage. In general terms, it is considered that cows during the transition period require 0.45% of Ca, 0.3-0.4% of P and 0.35-0.40% of Mg from the dry matter in the diet (NRC, 2001). Klimiene et al. (2005), consider that these requirements may vary according to breed, weather conditions, the digestive process and other factors that can support or inhibit the incorporation of these elements, they also think, that the quantity of minerals present in the food eaten by the animal affect the serum concentration of Ca, P and Mg.
Metabolic disorders linked to the transition period occur mainly when there is an alteration in the balance between consumption and excretion. In most cases, a mineral deficiency is manifested in a sub-clinical way; therefore, laboratory testing is carried out in order to determine the minerals profile of animals (Goff, 2006). Blood plasma proteins are a structural part of cells, they maintain the osmotic pressure, are involved as catalysers in chemical reactions helping to maintain the acid-base balance, take part in blood clotting and in the organism's defence systems as antibodies. Furthermore, they work as transport compounds for plasma constituents (Kaneko et al., 1997). Physiological factors such as age, growth, hormonal profile, sex, gestation, lactation, nutritional status, stress or loss of liquids can cause alterations in the plasmatic concentration of proteins (Jordán et al., 2006).
The concentration of plasmatic proteins (albumin and globulin fractions) is used as a diagnosis of pathologies, such as severe protein deficiency, low nourishment, metabolic alterations or liver and kidney diseases among others (Campos et al., 2007).
Cortisol is the physiological trigger of labour; it induces metabolic changes due to its activity on minerals and partially activates gluconeogenesis. This is why its evaluation during the transition period is a valuable tool for the understanding of the homeostatic mechanism and its maladjustments in the peri-partum period (Forslund et al., 2010; Campos et al., 2008).
Milk production in tropical regions takes place mainly within grazing systems using grain-based supplements at different percentages, according to the genetic merit of the animals and the productive phase during the lactation period (Alvés et al., 2009; Sairanen et al., 2006). Under similar management conditions, high individual genetic variation will be responsible for the homeostatic regulation during the transition period (Álvarez, 2001); however, this variation has not yet been completely elucidated.
The main objective of the present work was to analyse the variations in protein and minerals indicators of homeostasis during the transition period of dairy cows of two Bos taurus breeds, under tropical lowland conditions.
MATERIALS AND METHODS
The present work was carried out in a period of weather transition (from rains for dry period). It was done simultaneously with two commercial systems of milk production, under similar management conditions. One of them was done with the Hartón del Valle creole breed and the other one was done with the Holstein breed. The first breed is located in the central region of the Valle del Cauca department (Colombia) 4°27'N, 76°20' W, within a tropical dry forest ecosystem (Holdridge, 1987), situated at 950 masl, with an average temperature of 24 ºC, with a relative humidity of 75% and an annual rainfall of 1,050 mm bi-modally distributed. The basis of animal feed consisted of Bermuda grass (Cynodon nlemfuensis) managed with a rotational system and supplemented with a mixture of cracked corn plus soy cakes, with a total digestive nutrients (TDN) of 78%; they also had permanent access to mineralised salt at 6% and unrestricted access to water.
The second group of cows (Holstein) was located in a semi-intensive system at 3º27' N and 76º32' W, in a similar ecosystem to the previous group, situated at 1,050 masl, with an average temperature of 23 ºC and an annual rainfall of 1.200 mm bi-modally distributed. The feed consisted of Bermuda grass (Cynodon nlemfuensis) plus a supply of concentrate feed (TDN 85%) supplemented according to production; they also received mineralised salt (8% of P) and unrestricted access to water. Bromatological analysis was carried out both in the fodder offered and in the supplements provided to the animals. The Spartam® dairy ration programme (Michigan State University, 2010) was employed to calculate the dietary requirements of the animals.
The bromatological composition of Bermuda grass for the two groups showed the following average values: 61.5% (NDF), 36.8% (ADF), 3,800 cal/g of gross energy and 9.2% of protein. The estimated load capacity was of 4 mature animals per hectare in rotations of 32 days. The animals grazed in similar conditions during the prenatal and postpartum stages; in the postpartum stage, they were supplemented with the type of feed chosen by each farm.
Seven adult cows of the specialized Holstein breed were used. Their average production was of 15 L/day, their average weight was 550 kg and they had an average of 4 births. The same number of cows from the Hartón del Valle breed was used. Their average production was of 9 L/day, their average weight was 450 kg and they had an average of 5 births. The basic grazing diet did not have significant changes before and after the birth. All animals were chosen because they were in the last stages of gestation. The prepartum samples, taken 30 and 15 days before the birth, were done with blood tests. Samples were also taken on the actual day of birth and in the 3rd, 4th, 5th, 8th, 10th and 15th day of the early post partum stage; this was periodically done until the 60th day, with samples being taken every 9 days. In total there were 156 samples taken. They were collected after the morning milking, between 07:00 and 09:00 am, by coccygeal venepuncture using a vacutainer system in tubes with anticoagulant heparin and without it. Later on, they were centrifuged for 15 min at 800 g to obtain plasma and serum. These were identified and fractionated before freezing them at -20 ºC up to the moment of analysis. The body score condition of the animals and the average milk production were evaluated the day of the sampling.
Four indicators of the protein metabolism and three mineral indicators present in serum were evaluated. Cortisol was also determined. Total protein was determined through the Biuret method; albumin was determined by the bromocresol green method; globulins by the difference between total protein and albumin; and urea through the enzymatic colorimetric test. Calcium was determined through the methylthymol blue method. Phosphorous was determined by molybdate and magnesium by calmagite. For cortisol, radioimmunoassay (RIA) was used for the solid phase. For metabolites, specific reagents from Randox® (Crumlin, UK) were used for each test, except for total proteins that used reagents from IHR Diagnóstica and for the hormone cortisol for which the commercial reagent Siemens-DPC® (Los Ángeles, Ca) was used.
The results were analysed statistically in a 2x14 factorial model (two breeds, 14 collection sampling periods) in a completely randomized design. For each individual case, analysis of variance for principal effects was carried out using the statistical software package SAS 9.1 (Cary, NC). The accepted probability was less than 0.05 (P<0.05). When there were significant differences, the Tukey test was applied for comparison.
RESULTS AND DISCUSSION
Dairy cows experience big changes at the final stage of gestation and at the beginning of lactation. Such changes have a significant impact on their reproductive efficiency and on lactation, specially on the lactation curve and on the chemical composition of milk (Álves et al., 2009) The Bos taurus dairy cattle have productive characteristics with substantial differences regarding the volume of milk produced and the compositional quality of it (Elzo, 2001). Metabolism has a clear impact in the homeostatic physiological response during the transition period (Drackley, 1999).
The serum values of the analysed metabolites are useful for establishing a comparison with dairy cows, with moderate milk production, under similar conditions of nutritional management and sanitation. When compared, the changes predicted for highly productive cows, -those yielding 20 litres of milk per kilogram of body weight throughout the lactation period (Mühlbach, 2010)-, do not appear in cows with smaller production (Overton y Waldron, 2004).
In order to reach the greatest production efficiency, animals should be kept under appropriate sanitary conditions, especially at the beginning of lactation when the postpartum ovarian resumption occurs (Crowe y Williams, 2011). Metabolic decompensate (Goff, 2008) is a critical factor that affects the animal's health during this period. It is known that the transition period entails nutritional and metabolic problems (Drackley, 1999). Generally, nutritional primary healthcare in dairy cows is centred in the energy requirements of the animal; however, cattle also need minerals and protein for their development (Leblanc, 2010).
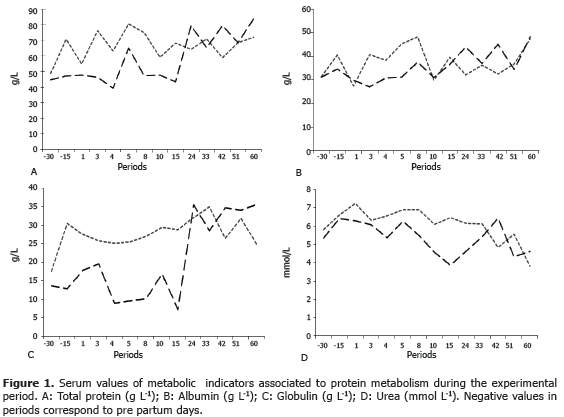
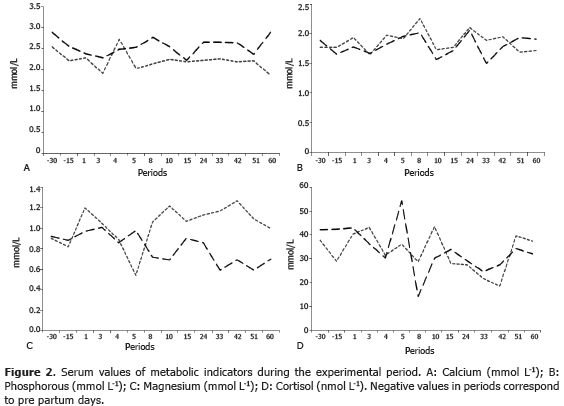
The present work compared the concentrations of indicators of protein metabolism and the minerals Ca, P and Mg, in two breeds located in semi-intensive grazing systems. The effect of the racial group, the transition period and the period up to the 60th day of lactation were studied (Figures 1 and 2). Differences were not found for racial group variable (P>0.05), neither for the sampling period. Given that there were no differences found, the average values for the two racial groups during the experimental period are shown (Table 1).
There is evidence to support the claim that the patterns of behaviour of the metabolism of dairy cows under tropical conditions is different to that of highly productive cows exploited in a temperate zone (Campos et al., 2007), given the complexity of the transition period, it should be analysed integrally, defining the parameters accomplished by the animal in order to guarantee physiological homeostasis (Drackley, 1999); it is important that the racial differences that constitute a source of variation are analysed in relation to the levels of production and adaptation (Álves et al., 2009; Campos et al., 2004). In the present work, the analysed metabolites show an important action of metabolic adjustment, but there was no variation between the racial groups, perhaps because the physiological mechanisms do not present significant challenges due to the production level.
Calcium. The homeostasis of calcium is fundamental for neuromuscular excitability, for blood clotting and for hormonal secretion (Wu et al., 2008). In the present work, the homeostasis of Ca was evident since differences between races (P>0.05), or between sampling periods, were not found. The average value was 2.38±0.7 mmol L-1, which is within the 2.33-3.10 mmol L-1 ranges found by Kaneko et al. (1997), and it is also similar to 2.5-3.0 mmol L-1 found by Klimiene et al. (2005). According to Ceballos et al. (2004), the transition period in cows is characterised by endocrine and homeostatic changes aimed at keeping calcemia at a minimal range of variation. The decline in the concentration of blood calcium in the postpartum period is due to the redirection of this mineral towards the mammary gland for colostrum synthesis (Wu et al., 2008); therefore, in this stage, the animal's ability to resume the homeostasis of Ca is put to the test. However, if the mechanisms to keep the normal concentration of calcium in the blood fail, the post partum cow runs a high risk of suffering hypocalcemia or, in less severe cases, a reduction of food intake, ruminal and intestinal hipomotility, reduced milk production and increased susceptibility to infectious and metabolic disease (Goff, 2008). The reduction of the dry matter intake, due to problems with the homeostasis of Ca, is an aggravating factor of the negative energetic balance (Leblanc, 2010). This can cause fat mobilization causing fatty liver disease and ketosis, which may lead to an interruption of the production and reproduction of the animal (Wilde, 2006). Hypocalcemia is caused by failures in the mobilization mechanism of calcium, which is controlled by parathormone, its imbalance causes a severe metabolic mismatch, responsible for high morbidity (Overton and Waldron, 2004). The present work did not find evidence of subnormal calcemia during the analysed periods.
Phosphorus. P carries out functions related to the secretion of milk, the energetic metabolism, the transportation of amino acids and the synthesis of phospholipids and proteins. It is also involved in cellular metabolism. About 80% to 86% of P present in the body of the animal is found in bones, teeth, and the rest of it is found in soft tissue (Álvarez, 2001). In the present study, the concentration of P in blood did not show differences (P>0.05) between breeds, the average value was of 1.84±0.37 mmol L-1, which lies between the ranges found by Kaneko et al. (1997) (1.81- 2.10 mmol L-1), Klimiene et al.(2005) (1.4-2.0 mmol L-1) and Ceballos et al. (2004) in high production cows (1.92±0.44 mmol L-1) and in low production cows (2.22±0.65 mmol L-1) in the Colombian Andean region. Similarly, the present study found that cows from the Hartón del Valle breed, with a lower milk production and a lower body weight, showed higher P values in comparison to Holstein cows without statistical differences between them.
The lowest levels of P were observed at the beginning of lactation, on the 3rd day after the birth (1.69± 0.35 mmol L-1) and then on the 10th and 15th day with 1.77±0.22 and 1.78±0.25 values, respectively, which indicates that at the beginning of this phase there is a redirection of the P present in blood towards the mammary gland. The deficiency of this mineral in the organism is part of the multifactor complex associated to the low food intake and as such there is a deficiency in nutrients that is reflected in the low production of milk. According to Ekelund et al. (2006), the risk of P deficiencies increases at the beginning of lactation when Ca and P are mobilized through bone resorption to meet the demand for those nutrients. Reports to Karn (2001) notified the excess P is mainly excreted in feces, but there are also homeostatic processes for this mineral in which bone resorption, salivary recirculation and urinary excretion play a part. The levels of P at blood level are closely linked to its concentration in fodder and supplements. The serum concentration of P found in this study is within the reference values (Kaneko, 1997; Russell and Roussel, 2007) and reflects the contribution of nutrients supplied in the diet, especially in the mineral supplementation.
Magnesium. This mineral is a cation, at intracellular level works as an enzymatic catalyzer in metabolic reactions and, at extracellular level, is involved in nerve conduction, muscular function and in the mineral formation of bones (Goff, 2006). The requirement for Mg depends on factors such as age, production and biological availability of the mineral in the diet, but also on the levels of P and Ca in the provided feed, if these levels increase, then the need for Mg also increases (Álvarez, 2001). The present study did not find any differences (P>0.05) between races in terms of levels of Mg in blood; the average value was of 0.95±0.6 mmol L-1 this is within the value (1.01±0.19 mmol L-1) proposed by Campos et al. (2007) for cows at the beginning of lactation and in the range 0.74-0.95 mmol L-1 found by Kaneko et al. (1997). Within periods, differences (P<0.05) were only found on the 42nd postpartum day with a value of 2.06±1.24 mmol L-1, being this, the higher value found. This high concentration of Mg in blood may be due to the mobilization of this mineral from the skeletal system to milk; which, is at its ascent phase in the productive curve (Alvarez, 2001). This does not mean that the increase is due to dietary contributions since the mineral supply was not modified during the experimental period. Maintaining the plasma concentration of Mg depends more on the constant flux of this mineral present in the diet offered to the animal than on the mobilization from the skeletal system. Despite the fact that about a 70% of organic Mg is found in bones, these do not constitute an important mineral source, since the homeostatic mechanisms produced are the result of problems in the levels of Ca not in the levels of Mg. The excess of Mg is quickly released through kidney filtration (Goff, 2008). Low levels of Mg in plasma affect the metabolism of Ca by reducing the secretion of parathormone (PTH) in response to hypocalcemia or diminishing tissue sensitivity to it.
Taken into consideration the calculations carried out for the diet according to the levels of production of the animals and the type of pasture and supplement provided for the cows, the present study considers that the Mg requirements were adequately covered. This was verified both by the serum values found and by the absence of clinical reports of Mg deficiency as a metabolic disease in both examples. Likewise the Mg serum values found did not show a deficit that could have pointed towards subclinical hypomagnesemia.
Total serum protein. The sum of albumin and globulin constitute the total serum protein. Proteins are involved in the maintenance of the osmotic pressure and are a source of amino acids. Moreover, they fix and transport lipids, fatty acids, copper, iron and hemoglobin (Rusell and Roussel, 2007). The found average value for total protein was 62.2±22.7 g L-1, being the only metabolite that showed differences (P<0.05) among breeds (Table 1). In this case, the value found for the Hartón del Valle breed (57.7±23.2 g L-1) agrees with the findings (75.1±6.9 g L-1) of Campos et al. (2004) for the same breed. In cows of the Holstein breed, the value found (66.7±20.6 g L-1) is markedly lower than the values found for this breed by Ceballos et al. (2002) (75±1 g L-1) and similar to the value found by Campos et al. (2007) (66.2±2.7 g L-1). If it is accepted that the nutritional requirements of the animals used in this study were provided by the diet, then the type of protein supplied may have generated significant alterations for its rumen metabolism, which could have happened to the Holstein cows whose serum value showed lower direct synthesis (Kaneko et al., 1997).
Differences were found between periods (P<0.05) in total protein on the 4th day (50.1±24.1) and on the 60th day (78.7±6.0 g L-1). This could be explained by the transition in the adaptation process to the new physiological state of the animal. In so far as lactation progresses, requirements decrease and so the animal can reach homeostasis (Ceballos et al., 2002). In general, low values of total protein can show a relative deficit in the input of protein in the diet of the animals, which reduces the availability of amino acids for the synthesis of microbial protein. Lower synthesis of bacterial protein generates lower quantities of precursors for the synthesis of albumin in the liver.
Depending on the moment of measurement, it is possible to see a reduction in milk production or a delay in fetal development (Klimiene et al., 2005). The higher values of total protein for the Hartón del Valle breed can be explained by the fact that these cows have lower requirements given their adaptation to the ecological environment where they live, which guarantees a rational productivity with the available nutrients under adverse conditions.
Albumin. Albumin synthesized in the liver and catalyzed by all the metabolically active tissue is the main protein taking part in plasma osmosis and in the transport of its constituents (Russell and Roussel, 2007). In the present study, the average value for albumin obtained was 36.65±8.5 g L-1 without significant differences between breeds, this value coincides with that informed by Kaneko et al. (1997) in a range between 30.3-35.5 g L-1 and the value determined by Ceballos (2002) (32±7 g L-1). The concentration of albumin did not show differences between the sampling periods. Albumin in healthy animals does not show significant modifications given that its values are altered only in instances associated with its synthesis or use, which did not happen in the present study. Given that the protein metabolism is difficult to assess accurately, and given that the data collected in urea from the blood only provided partial information, an alternative is to determine the protein consumption based on the serum levels of albumin, like it was done in the present study (Campos et al., 2004).
Globulins. Serum globulins comprise alpha, beta and gamma globulins. Their values depend on the different situations associated to the response to external challenges such as ectoparasites (Campos et al., 2007). Equally, values respond to humoral defense mechanisms activated by the animal under specific circumstances (Meza et al., 2010; Jordán et al., 2006); the general values reported in literature show a wide variation and the physiological state and health of the animal have a significant impact in these values (Goff and Horst, 1997). The present study found a 27.7 g L-1 value for the Holstein breed and a 20.3 g L-1 for the Hartón del Valle breed, this shows a lower activation of immune response in the last breed likely associated to being well adapted to the environment. The values found are slightly lower than others reported for these racial groups (Ceballos et al., 2002; Campos et al., 2004).
Urea. The ruminal metabolism of nitrogen compounds produces ammonia, which is not used completely by microflora, a part of it goes to the liver where is transformed in urea and it is excreted in the urine and partially in milk too (Biswajit et al., 2011). The average value of blood urea (5.74±1.81 mmol L-1) did not vary by breed or period (P>0.05), and was lower than that found by Noro et al. (2011) (6.29±1.02 mmol L-1). Non-protein nitrogen circulating in blood is important because it has effects on the integrity of the liver and mamary tissue, it can also alter the reproductive behaviour with decreases in the pregnancy rates and an elevation of the uterine pH after oestrus (Biswajit et al., 2011).
Cortisol. Cortisol in mammals is released routinely and it increases as a response to stressful situations; for this reason, concentrations of cortisol in plasma is used as a physiological indicator of adaptation to the environment or to new management conditions for the animal. The average value found in this study for this hormone was 33.65±9.06 nmol L-1, without differences (P>0.05) between breeds. This value coincides with that found by Hernández et al. (2011) (32.42 nmol L-1) for Hartón del Valle cows under tropical conditions and it ranges between 36-81 nmol L-1 proposed by Kaneko et al. (1997). High cortisol levels around the transition period can be explained by the challenge that milk production entails for the cows in order to find a homeostatic state. This hormone, due to its gluconeogenic activity, guarantees milk production despite the temporary insulin resistance and it guarantees increased lactose synthesis to ensure milk production (Leblanc, 2010; Campos et al., 2008).
CONCLUSIONS
The present study did not find differences between the two racial groups for most metabolites and minerals analyzed. This is due to the homeostatic adjustments that cows make according to milk production and it is independent of management or racial group.
BIBLIOGRAPHY
Álvarez, J. 2001. Bioquímica nutricional y metabólica del bovino en el trópico. Primera edición. Ciencia y Tecnología, Universidad de Antioquia, Medellín. 73 p. [ Links ]
Alvés, N.G., M.N. Pereira e R.M. Coelho. 2009. Nutrição e reprodução em vacas leiteiras. Revista Brasileira de Reprodução Animal Suplemento Belo Horizonte 6: 118-124 [ Links ]
Andrieu, S. 2008. Is there a role for organic trace element supplements in transition cow health? Veterinary Journal 176(1): 77-83. [ Links ]
Biswajit, R., B. Brahma, S. Ghosh, P.K. Pankaj and G. Mandal. 2011. Evaluation of milk urea concentration as useful indicator for dairy herd management: A review. Asian Journal Animal and Veterinary Advances 6(1): 1-19. [ Links ]
Campos, R., E.S. Carreño y F.D. González. 2004. Perfil metabólico en vacas nativas. Revista Orinoquia 8(2): 32-41. [ Links ]
Campos, R., C. Cubillos y A. Rodas. 2007. Indicadores metabólicos en razas especializadas en condiciones tropicales en Colombia. Acta Agronómica 56(2): 85-92. [ Links ]
Campos, R., L.A. Lacerda, S.R. Terra y F.H. González. 2008. Parâmetros hematológicos e níveis de cortisol plasmático em vacas leiteiras de alta produção no Sul do Brasil. Brazilian Journal Veterinary Research and Animal Science 45(5): 354-361. [ Links ]
Ceballos, A., N.A. Villa, A. Bohórquez, J. Quiceno, M. Jaramillo y G. Giraldo. 2002. Análisis de los resultados de perfiles metabólicos en lecherías del trópico alto del eje cafetero colombiano. Revista Colombiana de Ciencias Pecuarias 15(1): 26-35. [ Links ]
Ceballos, A., N.A. Villa, T.E. Betancourth y D.V. Roncancio. 2004. Determinación de la concentración de calcio, fósforo, y magnesio en el periparto de vacas lecheras en Manizales, Colombia. Revista Colombiana de Ciencias Pecuarias 17(2): 125-133. [ Links ]
Crowe, M.A. and E.J. Williams. 2011. Triennial lactation symposium: effects of stress on postpartum reproduction in dairy cows. Journal of Animal Science 90(5): 1722-1727. [ Links ]
Drackley, J.K. 1999. Biology of dairy cows during the transition period: the final frontier? Journal of Dairy Science 82(11): 2259-2273 [ Links ]
Elzo, M.A. 2001. Evaluación genética de animales en poblaciones multirraciales de bovinos utilizando modelos lineales. Archivos Latinoamericanos de Producción Animal 14(4): 154-160. [ Links ]
Ekelund, A., R. Spörndly and K. Holtenius. 2006. Influence of low phosphorus intake during early lactation on apparent digestibility of phosphorus and bone metabolism in dairy cows. Livestock Science 99(2-3): 227-236. [ Links ]
Forslund, K.B., O.A. Ljungvall and B.V. Jones. 2010. Low cortortisol levels in blood from dairy cows with ketosis: a field study. Acta Veterinaria Scandinavica 52:31. [ Links ]
Hernández, E., R. Campos y L. Patiño. 2011. Comportamiento metabólico en el periparto de vacas Hartón del Valle bajo condiciones de trópico bajo. Acta Agronómica 60(1): 13-26. [ Links ]
Holdridge, L. 1987. Ecología basada en zonas de vida. Instituto Interamericano de Cooperación para la Agricultura (IICA), San José, Costa Rica. 9 p. [ Links ]
Huzzey, J.M., M.A. Von Keyserlingk and D.M. Weary. 2005. Changes in feeding, drinking, and standing behavior of dairy cows furing the transition period. Journal of Dairy Science 88(7): 2454-2461 [ Links ]
Goff, J.P. 2008. The monitoring, prevention and treatment of milk fever and subclinical hypocalcemia in dairy cows. Veterinary Journal 176(1): 50-57. [ Links ]
Goff, J.P. 2006. Macromineral physiology and application to the feeding of the dairy cow for prevention of milk fever and other periparturient mineral disorders. Animal Feed Science and Technology 126: 237-257. [ Links ]
Goff, J.P. and R.L. Horst. 1997. Physiological changes at parturition and their relationship to metabolic disorders. Journal of Dairy Science 80(7): 1260-1268. [ Links ]
Jordán, D.; Villa, N.; Gutiérrez, M.; Gallego, A.; Ochoa, G.; y Ceballos, A. 2006. Indicadores bioquímicos sanguíneos en ganado de lidia mantenido en pastoreo en la cordillera central colombiana. Revista Colombiana de Ciencias Pecuarias 19(1): 18-26. [ Links ]
Kaneko, J.J., J.H. Harvey and M.L. Bruss. 1997. Clinical biochemistry of domestic animals. Fifth edition. Academic Press, San Diego, CA. 932 p. [ Links ]
Karn, J.F. 2001. Phosphorus nutrition of grazing cattle: a review. Animal Feed Science and Technology 89: 133-153. [ Links ]
Klimiene, I., V. Spakauskas and A. Matusevicius. 2005. Correlation of different biochemical parameters in blood sera of healthy and sick cows. Veterinary Research Communications 29(2): 95-102. [ Links ]
Leblanc, S. 2010. Monitoring metabolic health of dairy cattle in the transition period. The Journal of Reproduction and Development 56:S29-35. [ Links ]
Michigan State University, 2010. Spartam dairy ration evaluator/Balancer versión 3.0. User guide. Animal Science Deparment, Dairy Nutrition Group. 150 p. [ Links ]
Meza, M.A., A.F. González, C.M. Becerril, F.J. Ruiz, P. Diaz and B. Vallejo. 2010. Polimorfismo genético de la b-Lactoglobulina en la leche de vacas Holstein y Criollo lechero tropical. Agrociencia 44(5):531-539 [ Links ]
Mühlbach, P.R. 2010. Considerações sobre a otimização do consumo da vaca leiteira In: Consumo e preferência alimentar dos animais domésticos, Ed. Vieira, S.L., Londrina, Phytobiotics Brasil. 315 p. [ Links ]
NRC (National Research Council). 2001. Nutrient requirements of dairy cattle. National Academy Press, Washington, D.C. 381 p. [ Links ]
Noro, M., J. Borkert, G.A. Hinostroza, R. Pulido y F. Witter. 2011. Variaciones diarias de metabolitos sanguíneos y su relación con el comportamiento alimenticio en vacas lecheras a pastoreo primaveral. Revista Científica FCV-LUZ 21(2): 125-130. [ Links ]
Overton, T.R. and M.R. Waldron. 2004. Nutritional management of transition dairy cows: strategies to optimize metabolic health. Journal of Dairy Science 87: 105-119. [ Links ]
Russell, K.E. and A.J. Roussel. 2007. Evaluation of the ruminant serum chemistry profile. The Veterinary Clinics of North America, Food Animal Practice 23(3): 403-426. [ Links ]
Sairanen, A., H. Khalili and P. Virkajärvi. 2006. Concentrate supplementation response of the pasture-fed dairy cows. Livestock Science 104(3): 292-302 [ Links ]
Wilde, D. 2006. Influence of macro and micro minerals in the peri-parturient period on fertility in dairy cattle. Animal Reproduction Science 96(3-4): 240-249. [ Links ]
Wu, W., J. Liu, G. Xu and J. Ye. 2008. Calcium homeostasis, acid-base balance, and health status in periparturient Holstein cows fed diets with low catión-anión difference. Livestock Science 117(1): 7-14. [ Links ]