Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Facultad Nacional de Agronomía Medellín
Print version ISSN 0304-2847
Rev. Fac. Nac. Agron. Medellín vol.66 no.1 Medellín Jan./June 2013
Chlorophyll Fluorescence in Partially Defoliated Grape Plants (Vitis vinifera L. cv. Chardonnay)
Fluorescencia de la Clorofila en Plantas de Uva (Vitis vinifera L. cv. Chardonnay) Defoliadas Parcialmente
Jaime Ernesto Peña-Olmos1 y Fánor Casierra-Posada2
1 Professional Professor. Universidad Pedagógica y Tecnológica de Colombia - Faculty of Agricultural Sciences. Research Group in Plant Ecophysiology. P.O. Box 661, Boyacá, Colombia. <jaime.pena@uptc.edu.co>
2 Associate Professor. Universidad Pedagógica y Tecnológica de Colombia -Faculty of Agricultural Sciences. Research Group in Plant Ecophysiology. P.O. Box 661, Boyacá, Colombia. <fanor.casierra@uptc.edu.co>
Received: May 12, 2012; acepted: January 05, 2013.
Abstract
The chlorophyll content and fluorescence were determined in five-year-old grape plants (Vitis vinifera L. cv. Chardonnay) that were subjected to early partial defoliation, in Villa de Leyva, Colombia. The experimental design was completely randomized, consisting of two treatments (50% defoliation and control), each with four replications of 35 plants. Every two weeks, one of every two recently-emerged leaves was removed from the non-control plants. The determination of total chlorophyll content was carried out on six leaves per plant using a CCM-200 Plus chlorophyll meter, while chlorophyll fluorescence measurements were taken with one darkadapted leaf per plant using a Junior-PAM fluorometer. Initial fluorescence (Fo), maximum fluorescence (Fm), terminal fluorescence (Ft), variable fluorescence (Fv), electron transport rate (ETR), maximum photochemical quantum yield of PSII (Fv/ Fm), effective photochemical quantum yield of photosystem II (Y(II)), photochemical fluorescence quenching coefficient (qP), two non-photochemical quenching coefficients (qN and NPQ), quantum yield of light-induced non-photochemical fluorescence quenching (Y(NPQ)), and quantum yield of non-light-induced non-photochemical quenching (Y(NO)) were measured. The chlorophyll concentration index showed higher values in the defoliated plants. There were no significant differences for the values of Fm, Ft and Fv. Fo was higher in the defoliated plants, while ETR, Fv/Fm and Y(II) showed higher values in the control plants. It is evident that a reduction in leaf area modifies the partitioning of excitation energy destined for photochemical and non-photochemical processes, thus directly influencing the photosynthetic process of the plants evaluated.
Key words: Maximum photochemical quantum yield, Y(II), stress, PSII.
Resumen
Utilizando un diseño completamente aleatorizado, con dos tratamientos (defoliación al 50% y control) y cuatro repeticiones de 35 plantas cada una, se determinó el contenido y la fluorescencia de la clorofila en plantas de uva, sometidas a defoliación parcial temprana, en Villa de Leyva, Colombia. Como material vegetal se utilizaron plantas de Vitis vinifera L. cv. Chardonnay de cinco años de edad. Quincenalmente, se realizó la defoliación parcial a las plantas, la cual consistió en retirar una de cada dos hojas recién emergidas en cada una de las plantas sometidas al tratamiento de defoliación. La determinación de los contenidos de clorofila total se realizó en seis hojas por planta con un analizador CCM-200 plus. Las mediciones de fluorescencia de la clorofila se realizaron en una hoja por planta, previamente adaptada a la oscuridad, utilizando un fluorómetro Junior-PAM. Se determinó la fluorescencia inicial (Fo), fluorescencia máxima (Fm), fluorescencia terminal (Ft), fluorescencia variable (Fv), tasa de transporte de electrones (ETR), eficiencia cuántica máxima del fotosistema II (Fv/Fm), eficiencia cuántica efectiva del fotosistema II o Y(II), el coeficiente qP de disipación fotoquímica, los coeficientes qN y NPQ de disipación no fotoquímica, el rendimiento cuántico, Y(NPQ), de disipación no fotoquímica inducida por la luz y el rendimiento cuántico, Y(NO), de disipación no fotoquímica no inducida por luz. El índice de concentración de clorofila mostró un mayor valor en las plantas defoliadas. No se encontraron diferencias significativas para los valores de Fm, Ft y Fv. Fo fue mayor en las plantas defoliadas, mientras que la ETR, Fv/Fm y Y(II) presentaron mayores valores en las plantas control. Con lo anterior, es evidente que la reducción del área foliar modifica la fracción de energía de excitación destinada para los procesos fotoquímicos y no fotoquímicos, incidiendo de forma directa en el proceso fotosintético de las plantas evaluadas.
Palabras clave: Eficiencia cuántica máxima, Y(II), estrés, PSII.
Viticulture is a recent development in Colombia as compared to other agricultural sectors such as coffee or bananas. Nevertheless, in the past two decades, this crop has been expanding and, in particular, improving in quality. Total production of wine grapes in Colombia is as yet relatively low, but it is clear that the way to compete with producers such as Argentina or Chile, which supply a large part of the domestic and international markets, is by gaining recognition locally and abroad in terms of wine quality.
Colombia has 2,578 ha of vineyards, producing close to 38,120 t of grapes per year, of which only 0.1% is used for wine (Agronet, 2009). The area harvested nationally is less than 2% of the total area harvested in countries such as Chile or Argentina, which have close to 190,000 and 220,000 ha, respectively. Accordingly, total production in Colombia is well below that of these countries. Chile produces close to 2,500,000 t of grapes, while Argentina leads production on the continent with 2,900,000 t (Faostat, 2009). In the highland tropical conditions of Colombia, the principal wine-growing zones are located in the department of Boyacá, between 2,200 and 2,560 masl. In this part of the country, it is possible, with good crop management, to obtain two harvests per year, thanks to the local agroclimatic conditions (Almanza et al., 2010).
Final production of a crop is affected by the conditions in which it was grown. Plants subjected to any type of stress show morpho-physiological responses that affect quantity and quality of the final product (Prins and Verkaar, 1992). The type of wine, its quality, its smell, its color, and its flavor depend directly on the content of secondary metabolites in the berry, which are in turn the result of the combination of microclimate, grape variety, soil properties, and nutritional status of the plant. These four factors frame the potential yield of the grapevine in terms of quantity and quality. Nevertheless, human actions partially or completely modify the behavior of each factor, thus determining the terroir of the vine's development (Vaudour, 2002; Quijano, 2006). Furthermore, it must be noted that secondary metabolites do not only determine wine quality, but they also play an important role as antioxidants that benefit human health by protecting against oxidative stress caused by highly-reactive free radicals in the body (Quijano, 2003).
One of the most influential human modifications of fruit composition in grapevines is early partial defoliation. This procedure is utilized in the majority of wine-producing countries to increase the concentration of various compounds in the fruits, such as phenols, thus giving new characteristics of flavor and aroma to the wine (Kemp et al., 2010). Plants present different reactions to defoliation. Leaf removal creates a negative effect by reducing growth rate, but plants possess a tolerance threshold to defoliation (McNaughton, 1983). Schneiter et al. (1987); Schneiter and Johnson (1994) confirmed that partial defoliation affected the yield of sunflower crops regardless of the developmental stage of the plant at the moment of defoliation. But, it is possible that some plants initiate a recovery period after defoliation so that the end effect is negligible (Prins et al., 1989).
The quantification of plant stress (in this case caused by defoliation) is a useful tool in plant ecophysiology, with which potential photosynthetic behavior and dissipation of absorbed energy can be determined.
Chlorophyll fluorescence is a widely-used technique for tracking this phenomenon, both in cultivated and wild plants. Photosystem II (PSII) is perhaps the most important complex in a plant's photosynthetic process (Ruban et al., 2003), and it is affected by various stress factors, especially abiotic factors. Hence, the characterization of PSII behavior is an important tool for researchers to evaluate photosynthetic activity in plants.
Part of the light energy absorbed by a plant is transferred as excitation energy and trapped by the PSII reaction center, where it is utilized for useful chemical work, and the other part is dissipated mainly as heat and to a lesser degree as fluorescence (González-Moreno et al., 2008). The distribution of energy in these three processes occurs simultaneously, so that an increase in efficiency of one process results in the reduction of the other two. Hence, by measuring chlorophyll fluorescence, it is possible to obtain information about the photochemical efficiency and thermal dissipation of absorbed energy (Maxwell and Johnson, 2000).
Given the optimal conditions and high observed yields for grape growing in Boyacá, Colombia, research is necessary to improve the physico-chemical fruit properties, in the spirit of raising quality standards for Colombian wine and competing at an international level with the major South American wine growing regions. The present study therefore aimed to evaluate the influence of early partial defoliation on chlorophyll fluorescence in grape plants (Vitis vinifera L. cv. Chardonnay) grown in the highland tropics.
Materials and methods
The experiment was carried out in Villa de Leyva, Colombia (5°38' N, 73°32' W) at an altitude of 2,143 masl, with an average temperature of 16.9 °C and average annual rainfall of 716.9 mm (Instituto Humboldt, 2010). The trial was performed on 280 five-year-old grape plants of the Chardonnay variety, planted in a trellis system with distances of 2.1 m x 1 m. The experiment followed a completely randomized design with two treatments (50% defoliation and a control with no defoliation), each with 4 repetitions of 35 plants. Every 15 days, between the initial pruning and 121 days after said pruning, half of the new leaves were removed from each plant in the defoliation treatment group.
Chlorophyll a fluorescence was determined 107 days after pruning (DAP), using a Junior-PAM modulated fluorometer (Walz® GmbH, Effeltrich, Germany) on 20 completely-expanded leaves taken from plants in each treatment and dark-adapted in aluminum foil bags at ambient temperature. An actinic pulse of light at 1,150 µmol m-2 s-1 was used. Measurements taken included: minimum fluorescence (Fo), maximum fluorescence (Fm), stable fluorescence (Ft), effective photochemical quantum yield of photosystem II (Y(II)), quantum yield of light-induced nonphotochemical quenching (Y(NPQ)), quantum yield of non-light-induced non-photochemical quenching (Y(NO)), electron transport rate (ETR), coefficient of photochemical fluorescence quenching (qP), and two non-photochemical quenching coefficients (qN and NPQ). Using these parameters, variable fluorescence (Fv) and maximum quantum efficiency of PSII (the ratio of Fv/Fm) were calculated.
The effective photochemical quantum yield of PSII (Y(II)) estimates the fraction of excitation energy used photochemically in light-adapted plants (Heinz Walz®, 2007). On the other hand, Y(NPQ) determines the fraction of excitation energy dissipated as heat through photo-protection mechanisms, while the Y(NO) factor shows the total non-photochemical losses, other than heat. The sum of Y(II), Y(NPQ) and Y(NO) should be equal to 1 (Kramer et al., 2004).
Chlorophyll content was measured at 125 days after initial pruning on six leaves per plant, using a CCM 200 plus chorophyll meter (Opti-sciences®, Hudson, USA). The chlorophyll concentration index (CCI) was determined. Photosynthetically-active radiation at the moment of the chlorophyll content measurement was calculated using measurements of light intensity expressed in lux, using a Testo-Term® 0500 light meter (Barcelona, Spain).
The data obtained in the experiment were subjected to classical variance analysis (P<0.05) using the PASW (Predictive Analytics Software) program, version 18.0.0 (30 July 2009, IBM Corporation, Somers, USA).
Results and discussion
The variance analysis showed significant differences (P<0.05) for minimum fluorescence (Fo), while for Fm and Fv there existed no statistical differences between treatments. The value of minimum fluorescence increased by 33.77% in the defoliated plants as compared to the control plants.
Partial defoliation induces changes in leaf arrangement and thus light exposure. When leaf area is reduced, photon flow increases towards the plant's remaining leaves, which can increase leaf temperature and saturate reaction centers due to an excess of light. Hence, the value of minimum fluorescence encountered in the defoliated plants in the present study support the findings of Gonçalves et al. (2001), who studied the behavior of Swietenia macrophylla King under conditions of high and low light and found higher values of Fo in plants exposed to direct light.
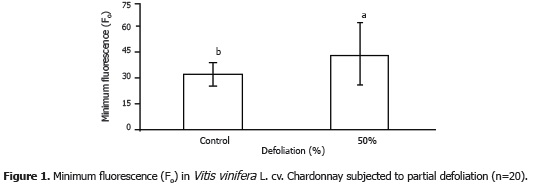
It is possible for Fo values to increase when there is a slowdown in excitation energy transfer from the light collection system to the reaction center (Baker and Rosenqvist, 2004), or when there is some type of damage in the PSII reaction centers themselves (Vieira et al., 2010). With regards to defoliation, in studies undertaken by Retuerto et al. (2006) on Ilex aquifolium plants, initial fluorescence was reduced when defoliation was caused by herbivory, but increased when defoliation was carried out manually.
This was perhaps due to the fact that herbivorous defoliation induces a quicker response from the plant as a result of the action of insect mandibles on the leaves, thus creating a compensatory response. On the other hand, Layne and Flore (1993) reported a reduction in minimum fluorescence values measured seven days after defoliation of Prunus cerasus plants exposed to 14 h of daily light. This was directly attributed to this species' tolerance to high radiation, as non-defoliated plants of the same species only showed photoinhibition when they were exposed to 24 h of continuous illumination.
Variance analysis revealed statistically significant differences (P<0.05) for maximum quantum yield of PSII (Fv/Fm) in dark-adapted leaves. This ratio decreased 7.75% in the defoliated plants as compared to the control plants (Figure 2).
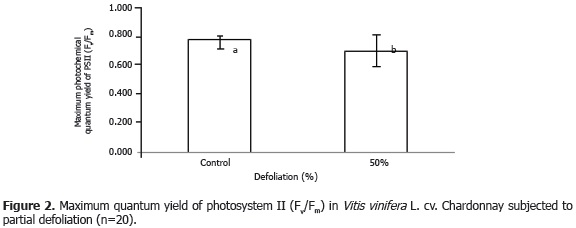
With regards to this result, Otronen and Rosenlund (2001) found no significant difference in maximum photochemical quantum yield of PSII when they subjected Pinus sylvestris plants to different levels of defoliation. In the same respect, maximum photochemical efficiency of PSII was not affected by either natural or experimental defoliation in Triticum aestivum plants (Macedo et al., 2007). Nevertheless, in the present study, a reduction was observed in this value, which indicates that defoliation induces a higher degree of stress in Chardonnay grape plants, perhaps due to higher exposure of the remaining leaves to direct radiation, which elevates leaf temperature and affects photosynthesis mechanisms. When Ilex aquifolium plants were subjected to a reduction in leaf area, the behavior of maximum quantum yield fluctuated according to the type of defoliation. Hence, Fv/Fm increased when defoliation was from herbivory, but decreased when the reduction in leaf area was manual (Retuerto et al., 2006), as herbivory damage induced a faster rate of recovery than manual defoliation did. A different result was obtained in Prunus cerasus, in which the maximum quantum efficiency of PSII, measured seven days after the defoliation event, increased slightly with respect to non-defoliated plants (Layne and Flore, 1993), possibly due to compensatory photosynthesis, which does not occur immediately but rather has a slow rate of recovery.
Karukstis (1991) stated that reductions in maximum quantum efficiency of PSII occur simultaneously with a reduction in net assimilation of CO2, and if this reduction is shown in continuously illuminated leaves, it is possible that the effect is due to photoinhibition. This may explain the results of the present study, because a reduction in leaf area leads to higher illumination of remaining leaves, which, combined with the high radiation typical of the study area (between 1,000 and 1,500 µmol m-2 s-1), could have caused a slight photo-inhibitory phenomenon in the leaves of the grape plants being studied. In the same way, Gamon and Pearcy (1990) confirmed that the ratio of variable fluorescence to maximum fluorescence in grape leaves decreases when plants are grown under high-light conditions, but not when they are grown under reduced light intensity.
The values of the non-photochemical quenching coefficients (qN and NPQ) showed no statistically significant differences (P>0.05) between the defoliated plants and the control plants. Nevertheless, the photochemical quenching coefficient qP was reduced by 17.89% in the defoliated plants, showing significant differences in comparison to the control plants (P<0.05) (Table 1).
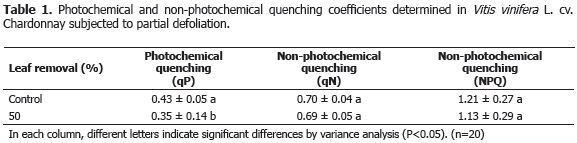
The above results support the values found for Fv/Fm, because, if the photochemical quenching coefficient is directly related to photosynthesis, as Karukstis (1991) claims, excitation energy destined for photosynthesis (qP) must follow the same trend as Fv/Fm, which is to say, it should be less in defoliated plants than in control plants, as was found in the present study.
Furthermore, Weis and Berry (1987) found that the photochemical quenching coefficient (qP) in different species was close to 1 in conditions of low light intensity, but diminished to 0.7 when light intensity was strongly increased, which indicates a low number of electron transporters in the reduced state, and thus, an oversaturation of the system. This explains the behavior of the defoliated plants in the present study, since a higher exposure to direct radiation after a reduction in leaf number increases photon flow and leaf temperature and finally triggers a failure of the photosynthetic system.
Figure 3 shows the partitioning of excitation energy in light-adapted plants. Effective quantum efficiency of PSII (Y(II)) showed a statistically significant reduction of 23.41% in the defoliated plants in relation to the control plants (P<0.05). Energy dissipation as heat (Y(NPQ)) showed no significant difference between treatments. However, nonheat non-photochemical losses (Y(NO)) did show significant differences (P<0.05) with an average value 16.61% higher in the defoliated plants than in the control plants.
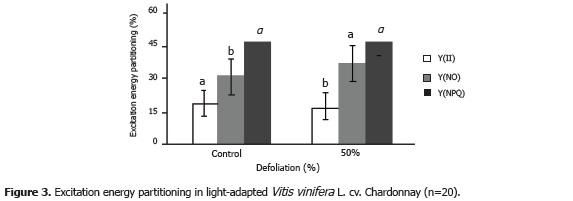
Genty et al. (1989) claimed that effective PSII photochemical efficiency is directly proportional to the product of the photochemical quenching coefficient qP and the efficiency of excitation energy capture by PSII reaction centers (maximum PSII quantum efficiency (Fv/Fm)). This agrees with the results of the present study, in which the value of Y(II) decreased in the defoliated plants at the same time as photochemical quenching and maximum quantum yield of PSII. On the other hand, the differences in Y(NO) could be related to the photoinhibition of PSII, because this parameter is related to non-photochemical losses other than heat (Y(NPQ)), which showed no significant differences. Y(NO) is related to the loss of energy throughout the electron transport chain (ΔpH) and to the destruction of the D1 protein of PSII.
In the same way, Weis and Berry (1987) confirmed that effective quantum yield in different plant species diminished as light intensity increased, until reaching a 40% reduction under conditions of excessive light as compared to the maximum value observed in lowlight conditions. Light intensity was a fundamental factor in the grape plants evaluated in the present study, as reduction in leaf area caused a higher exposure of the plants' remaining leaves, which also contributed to a rise in temperature.
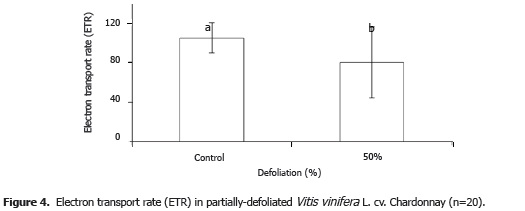
The average value for electron transport rate decreased 23.42% in the grape plants subjected to defoliation as compared to the intact control plants (P<0.05) (Figure 4). The amount of UV radiation hitting the leaves of the defoliated plants would have been higher than in the control plants. Many factors involved in photosynthetic efficiency decrease due to UV-B radiation, because said radiation inhibits PSII electron transport (Wang et al., 2010). UV radiation, and in particular UV-B radiation, causes damage to DNA (Britt, 1996) and the photosynthetic apparatus (Jansen et al., 1998). UV-B also causes respiration to increase, as well as the demand for resources to protect and repair photosynthesis mechanisms, both of which rob efficiency from photosynthetic electron transport (Gwynn-Jones, 2001). The low electron transport rate exhibited by the defoliated grape plants in the present study can thus be explained by the high incidence of ultraviolet radiation typical of the high-altitude tropical study area (Bohórquez- Ballén and Pérez-Mogollón, 2007).
Total chlorophyll concentration in the evaluated plants, expressed as CCI (chlorophyll content index), is shown in Figure 5. The average value registered for the defoliated plants was 9.1% higher than in the control plants (P<0.05). Leaves of defoliated plants are generally exposed to higher light intensity and, frequently, a modification of light quality, both of which influence the development of photosynthetic capacity (Richards, 1993). In this sense, it is possible that the grape plants of the present study modified their photosynthetic capacity in response to defoliation, raising total chlorophyll content (Richards, 1993) or diminishing the senescence rate of the remaining The average value for electron transport rate decreased 23.42% in the grape plants subjected to defoliation as compared to the intact control plants (P<0.05) (Figure 4). The amount of UV radiation hitting the leaves of the defoliated plants would have been higher than in the control plants. Many factors involved in photosynthetic efficiency decrease due to UV-B radiation, because said radiation inhibits PSII electron transport (Wang et al., 2010). UV radiation, and in particular UV-B radiation, causes damage to DNA (Britt, 1996) and the photosynthetic apparatus (Jansen et al., 1998). UV-B also causes respiration to increase, as well as the demand for resources to protect and repair photosynthesis mechanisms, both of which rob efficiency from photosynthetic electron transport (Gwynn-Jones, 2001). The low electron transport rate exhibited by the defoliated grape plants in the present study can thus be explained by the high incidence of ultraviolet radiation typical of the high-altitude tropical study area (Bohórquez-Ballén and Pérez-Mogollón, 2007).
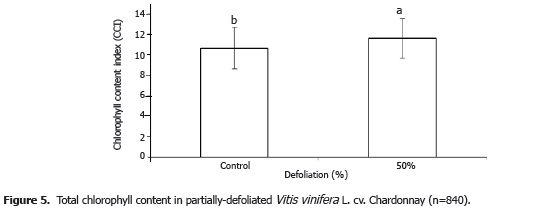
Total chlorophyll concentration in the evaluated plants, expressed as CCI (chlorophyll content index), is shown in Figure 5. The average value registered for the defoliated plants was 9.1% higher than in the control plants (P<0.05). Leaves of defoliated plants are generally exposed to higher light intensity and, frequently, a modification of light quality, both of which influence the development of photosynthetic capacity (Richards, 1993). In this sense, it is possible that the grape plants of the present study modified their photosynthetic capacity in response to defoliation, raising total chlorophyll content (Richards, 1993) or diminishing the senescence rate of the remaining leaves (Hunter and Visser, 1989). Though changes in chlorophyll content are related to photosynthesis, chlorophyll concentration is not always directly proportional to a plant's photosynthesis rate. This is because the remaining leaves in defoliated plants lengthen their useful life to compensate for lost leaf area, which modifies the chemical composition of these leaves in the form of increased sugar and lower concentrations of amino acids and organic acids (Hunter and Visser, 1989), which affect the leaf photosynthetic capacity. This is in agreement with the data obtained in the present study, because the ETR in leaves of the defoliated plants was lower than in the control plants, though chlorophyll content was lower in the latter. Thus, there was no positive relation found between chlorophyll content of leaves and their
photosynthetic efficiency.
Conclusions
Early partial defoliation induced an increase in stress in the grape plants evaluated. This reduced potential and effective photochemical efficiency for PSII (Fv/Fm and Y(II), respectively) as well as the photochemical quenching coefficient qP and electron transport rate. Chlorophyll content was higher in leaves of the defoliated plants, but this did not correlate with a higher photosynthetic efficiency. It is evident that a reduction in leaf area modifies the partitioning of excitation energy destined for photochemical and non-photochemical processes; and thus, directly influenced the photosynthetic process of the plants evaluated. The authors presume that the reduction in these indicators of photochemical function was related to oversaturation of light, leading to photoinhibition, and perhaps damage from high UV radiation in the remaining leaves on the defoliated plants.
Bibliography
Agronet, 2009. Principales departamentos productores. En: http://www.agronet.gov.co; consulta: marzo 2011. [ Links ]
Almanza, P.J., M.A. Quijano-Rico, G. Fischer, B. Chaves and H.E. Balaguera-López. 2010. Physicochemical characterization of 'Pinot Noir' grapevine (Vitis vinifera L.) fruit during its growth and development under high altitude tropical conditions. Agronomía Colombiana 28(2):173-180. [ Links ]
Baker, N.R. and E. Rosenqvist. 2004. Applications of chlorophyll fluorescence can improve crop production strategies: an examination of future possibilities. Journal Experimental Botany 55(403): 1607-1621. [ Links ]
Bohórquez-Ballén, J. y J.F. Pérez. 2007. Radiación ultravioleta. Ciencia y Tecnología para la Salud Visual y Ocular 9:97-104. [ Links ]
Britt, A.B. 1996. DNA damage and repair in plants. Annual Review of Plant Physiology and Plant Molecular Biology 47:75-100. [ Links ]
Dias, D.P. e R.A. Marenco. 2007. Fotossíntese e fotoinibição em mogno e acariquara em função da luminosidade e temperatura foliar. Pesquisa Agropecuária Brasileira 42(3):305-311. [ Links ]
Food and Agriculture Organization of the United Nations. 2009. Faostat. http://faostat.fao.org/site/567/default.aspx#ancor. accessed: march 2011. [ Links ]
Gamon, J.A. and R.W. Pearcy. 1990. Photoinhibition in Vitis californica: interactive effects of sunlight, temperature and water status. Plant Cell Environment 13(3): 267-275. [ Links ]
Genty, B., J.M. Briantais and N.R. Baker. 1989. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochimica et Biophysica Acta. General Subjects 990(1):87-92. [ Links ]
Gonçalves, J.F. de C., R.A. Marenco and G. Vieira. 2001. Concentration of photosynthetic pigments and chlorophyll fluorescence of Mahogany and Tonka bean under two light environments. Revista Brasileira de Fisiologia Vegetal 13(2):149-157. [ Links ]
González-Moreno, S., H. Perales-Vela y M.O. Salcedo-Álvarez. 2008. La fluorescencia de la clorofila a como herramienta en la investigación de efectos tóxicos en el aparato fotosintético de plantas y algas. Revista de Educación Bioquímica 27(4):119-129. [ Links ]
Gwynn-Jones, D. 2001. Short-term impacts of enhanced UV-B radiation on photo-assimilate allocation and metabolism: a possible interpretation for time-dependent inhibition of growth. Plant Ecology 154:67-73. [ Links ]
Heinz Walz GmbH. 2007. Junior-Pam Chlorophyll Fluorometer, operator's guide. Erhard Pfündel (ed). Effeltrich, Germany. 58 p. [ Links ]
Hunter, J.J. and J.H. Visser. 1989. The effect of partial defoliation, leaf position and developmental stage of the vine on leaf chlorophyll concentration in relation to the photosynthetic activity and light intensity in the canopy of Vitis vinifera L. cv. Cabernet Sauvignon. South African Journal of Enology and Viticulture 10(2): 67-73. [ Links ]
Instituto Humboldt. 2010. http://www.humboldt.org.co. Consulta: marzo 2011. [ Links ]
Jansen, M.A.K., V. Gaba and B.M. Greenberg. 1998. Higher plants and UV-B radiation: balancing damage, repair and acclimation. Trends in Plant Science 3(4): 131-135. [ Links ]
Karukstis, K.K. 1991. Chlorophyll fluorescence as a physiological probe to the photosynthetic apparatus. pp. 769-795. In: Sheer, H. (ed). Chlorophylls. CRC Press Inc., Boca Raton, FL. [ Links ]
Kemp, B.S., R. Harrison and G.L. Creasy. 2010. The Effect of the timing of leaf removal on berry ripening, flavour and aroma compounds in pinot noir wine. Doctor of Phylosophy thesis. Lincoln University. Christchurch, New Zealand. 219 p. [ Links ]
Kramer, D.M., G. Johnson, O. Kiirats and G.E. Edwards. 2004. New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynthesis Research 79: 209-218. [ Links ]
Layne, D.R. and J.A. Flore. 1993. Physiological responses of Prunus cerasus to whole-plant source manipulation. Leaf gas exchange, chlorophyll fluorescence, water relations and carbohydrate concentrations. Physiologia Plantarum 88:44-51. [ Links ]
Macedo, T.B., R. Peterson, C.L. Dausz and D.K. Weaver. 2007. Photosynthetic responses of wheat, Triticum aestivum L., to defoliation patterns on individual leaves. Environmental Entomology 36(3):602-608. [ Links ]
Maxwell, K. and G.N. Johnson. 2000. Chlorophyll fluorescence. A practical guide. Journal of Experimental Botany 51(345): 659-668. [ Links ]
McNaughton, S.J. 1983. Compensatory plant-growth as a response to herbivory. Oikos 40(3): 329-336. [ Links ]
Otronen, M. and H.M. Rosenlund. 2001. Morphological Asymmetry and chlorophyll fluorescence in Scots pine (Pinus sylvestris), responses to variation in soil moisture, nutrients and defoliation. Annales Botanici Fennici 38:285-294. [ Links ]
Prins, A.H. and H.J. Verkaar. 1992. Defoliation - Do physiological and morphological responses lead to (over) compensation. pp. 13-31. In: Ayres, P.G. (ed). Pests and Pathogens. Plant responses to foliar attack. Bios Scientific Publishers, Oxford. 220 p. [ Links ]
Prins, A.H., H.J. Verkaar and M. Van Den Herik. 1989. Responses of Cynoglossum officinale L. and Senecio jacobaea L. to various degrees of defoliation. New Phytologist 111: 725-731. [ Links ]
Quijano, M. 2003. La luz como factor de tipicidad y calidad en el terroir tropical de altitud, el ejemplo de la vid y el vino en el Valle del Sol. Viñedo y Cava Loma de Puntalarga, Nobsa. 9 p. [ Links ]
Quijano, M. 2006. Investigación e innovación. Promoción y defensa del terroir regional. Cultura Científica 4: 35-41. [ Links ]
Retuerto, R, B. Fernández-Lema and J.R. Obeso. 2006. Changes in photochemical efficiency in response to herbivory and experimental defoliation in the dioecious tree Ilex aquifolium. International Journal of Plant Sciences 167(2): 279-289. [ Links ]
Richards, J.H. 1993. Physiology of plants recovering from defoliation. pp. 85-94. In: Proceedings of the XVII International Grassland Congress. New Zealand Grassland Assoc., Palmerston North, N.Z. 186 p. [ Links ]
Ruban, A.V., M. Wentworth, A.E. Yakushevska, J. Andersson, P.J. Lee, W. Keegstra, J.P. Dekkerk, E.J. Boekema, S. Jansson and P. Horton. 2003. Plants lacking the main lightharvesting complex retain photosystem II macro-organization. Nature 421:648-653. [ Links ]
Schneiter, A.A. and B.L. Johnson. 1994. Response of sunflower plants to physical injury. Canadian Journal of Plant Science 74:763-766. [ Links ]
Schneiter, A., J.M. Jones and J.J. Hammond. 1987. Simulated hail research in sunflower: defoliation. Agronomy Journal 79:431-434. [ Links ]
Vaudour, E. 2002. The quality of grapes and wine in relation to geography: notions of terroir at various scales. Journal of Wine Research 13(2):117-141. [ Links ]
Vieira, D.A. de P., T. de A. Portes, E. Stacciarini-Seraphin and J.B. Teixeira. 2010. Fluorescência e teores de clorofilas em abacaxizeiro cv. pérola submetido a diferentes concentrações de sulfato de amônio. Revista Brasileira de Fruticultura 32(2):360-368. [ Links ]
Wang, G., Z. Hao, R.H. Anken, J. Lu and Y. Liu. 2010. Effects of UV-B radiation on photosynthesis activity of Wolffia arrhiza as probed by chlorophyll fluorescence transients. Advances in Space Research 45(7):839-845. [ Links ]
Weis, E. and J.A. Berry. 1987. Quantum efficiency of Photosystem II in relation to 'energy'-dependent quenching of chlorophyll fluorescence. Biochimica et Biophysica Acta. General Subjects 894(2):198-208. [ Links ]













