Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
Revista Facultad Nacional de Agronomía Medellín
versão impressa ISSN 0304-2847
Rev. Fac. Nac. Agron. Medellín vol.69 no.2 Medellín jul./dez. 2016
https://doi.org/10.15446/rfna.v69n2.59142
DOI: http://dx.doi.org/10.15446/rfna.v69n2.59142
Effect of different 1-methylcyclopropene doses on the postharvest period of pitahaya fruits (Selenicereus megalanthus Haw.)
Efecto de diferentes dosis de 1-metilciclopropeno sobre la poscosecha de frutos de pitahaya (Selenicereus megalanthus Haw.)
Javier Giovanni Álvarez-Herrera1, Yuli Alexandra Deaquiz2 and Anibal O. Herrera3
1 Grupo de Investigaciones Agrícolas (GIA). Facultad de Ciencias Agropecuarias. Universidad Pedagógica y Tecnológica de Colombia (UPTC). Avenida Central del Norte, No. 39-115, Tunja, Colombia. <jgalvarezh@gmail.com>
2 Fundación Universitaria Juan de Castellanos. Calle 17 No. 9-85, Tunja, Colombia.
3 Facultad de Ciencias Agrarias. Universidad Nacional de Colombia. A.A. 14490, Bogotá, Colombia.
Received: March 25, 2016; Accepted: June 8, 2016
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

ABSTRACT
Colombia is one of the leading producers of yellow dragon fruit, but exports of this fruit is low when compared to the domestic production because most do not meet the requirements of international markets in terms of quality and preservation. As a result, this study aimed to determine the effect of the application of 1-methylcyclopropene (1-MCP) on the preservation and quality of dragon fruit, given that it has been effective in the conservation and postharvest quality of many agricultural species allowing longer life, using a completely randomized experiment design with three treatments: 0, 300 and 600 mg L-1 of 1-MCP, with four replications. The 600 mg L-1 of 1-MCP dose was able to maintain the quality of the dragon fruit longer (28 days after harvest (dah)) because it managed to reduce the respiratory rate. The application of 1-MCP did not affect the firmness, loss of mass, total soluble solids, total titratable acidity, or total carotenoids in the fruits. The application of 1-MCP slowed the loss of fruit quality in terms of color because the 600 mg L-1 dose maintained the lightness of the fruits longer, reduced the color changes as expressed in a* and b* values and decreased the chlorophyll degradation.
Key words: Storage, Total carotenoids, Climacteric fruit, Ripening
RESUMEN
Colombia es uno de los principales productores de pitahaya amarilla, pero las exportaciones de este fruto representan un bajo porcentaje en comparación a la producción nacional, debido a que la mayor parte de los frutos no cumplen con los requisitos de los mercados internacionales, en cuanto a calidad y conservación. Por lo cual, esta investigación tuvo como objetivo determinar el efecto de la aplicación de 1-metilciclopropeno (1-MCP) en la conservación y calidad de frutos de pitahaya, dado a que ha sido eficiente en la conservación y calidad poscosecha de muchas especies agrícolas lo que permite prolongar su vida útil, para lo cual se utilizó un diseño experimental completamente al azar con tres tratamientos 0, 300 y 600 mg L-1 de 1-MCP, con cuatro repeticiones. La dosis de 600 mg L-1 de 1-MCP logró mantener la calidad de los frutos de pitahaya por más tiempo (28 días después de cosecha (ddc)), debido a que logró disminuir la tasa respiratoria. La aplicación de 1-MCP no afectó la firmeza, la pérdida de masa, los sólidos solubles totales, la acidez total titulable, ni los carotenoides totales de los frutos de pitahaya. La aplicación de 1-MCP retarda la pérdida de calidad de los frutos de pitahaya en cuanto al color, debido a que la dosis de 600 mg L-1 mantiene por mas tiempo la luminosidad de los frutos y disminuye los cambios de color expresados en los valores de a* y b*, así mismo disminuyó la degradación de la clorofila.
Palabras claves: Amacenamiento, Carotenoides totales, Fruto climatérico, Maduración
The yellow dragon fruit is one of the more exotic fruits from Colombia and, currently, it has a growing international market thanks to its nutritional content, sodium, potassium and vitamin A, flavor and aroma (Le Bellec and Vaillant, 2011), mainly with high demand for quality, flavor and nutraceutical properties (ICA, 2010); however, proper technology production packages and postharvest management that would allow this crop to be competitive do not exist, which implies the need to apply advanced techniques that have proven effective in maintaining the quality of fruits after harvesting, such as the application of 1-MCP (Selvarajah et al., 2001), thermal treatments (Dueñas et al., 2008), controlled atmospheres and wax (Hu et al., 2011; Hu et al., 2012), among others as packaging and use of potassium permanganate.
1-methylcyclopropene (1-MCP) has been widely used to control senescence in minimally processed fruits and vegetables (Toivonen, 2008; Watkins, 2008), which can delay ripening and maintain fruit quality longer during storage (Jiang et al., 2014). It also reduces the respiratory rate and ethylene production and helps inhibit the activity of 1-aminocyclopropane-1-carboxylic (ACC) oxidase (Amornputti et al., 2016). 1-MCP has been tested in many fruits, as reported in the nectarine (Öskaya et al., 2016), apple (Xiaotang et al., 2016), guava (Cerqueira et al., 2009), kiwi (Mao et al., 2007), plum (Khan and Singh, 2007), banana (Zhu et al., 2015), melon (Han et al., 2015) and broccoli (Ma et al., 2009) among others, where the best results have been observed in climacteric fruits (Serek et al., 2006). It has also been used in the preservation of flowers and foliage, increasing the postharvest shelf-life (Cerqueira et al., 2009).
The concentration of 1-MCP needed to block the action of ethylene varies by species, variety, degree of ripeness, production of new ethylene receptors, exposure time and temperature (Amornputti et al., 2016). The greater the time of exposure to the product, the lower the concentration needed to achieve the desired effect because there is an interaction between the concentration and exposure time. In this regard, Bassetto et al. (2005) applied concentrations of 900 nL L-1 and 100 nL L-1 of 1-MCP for 3 and 12 h on 'Pedro Sato' guava fruits and observed the same results, indicating that a 1-MCP application alone does not guarantee preservation of fruits.
Although the dragon fruit is a climacteric fruit and water loss does not exceed 4.3% during the first three weeks of storage, during ripening, the fruit loses a lot of its peel firmness at the time of harvest, making it a highly perishable fruit when stored under ambient conditions (Le Bellec and Vaillant, 2011).
Therefore, the objective of this research was to determine the effect of 1-MCP applications on the postharvest behavior of yellow dragon fruit (Selenicereus megalanthus Haw.) in an attempt to maintain fruit quality longer during storage.
MATERIALS AND METHODS
Ubication
The study was conducted in the postharvest laboratory of the Faculty of Agriculture Sciences of the Universidad Nacional de Colombia, Bogotá. The fruits were collected from a commercial orchard (Piedras Verdes farm) in the municipality of Miraflores (Boyacá, Colombia), located at an altitude of 1600 m, latitude 5°11' and longitude 73°08', with an average annual rainfall of 2500 mm, a temperature ranging between 18 and 24 °C and an average relative humidity of 87%.
Vegetable material
The plant material used was yellow dragon (or pitahaya) fruits (Selenicereus megalanthus Haw.), which were collected in maturity grade 3 (4580 norm of Icontec, 1996). The fruits presented uniform size, no mechanical damage and good phytosanitary condition. 1-MCP (powdered form from Rohm and Haas, Bogotá) was used.
The experimental design was completely randomized with three treatments that corresponded to the doses of 1-MCP (0, 300 and 600 mg L-1) with four replications, for a total of 12 experimental units (EU). Each EU was composed of six fruits.
The fruits were disinfected with a solution of 2% NaCl and washed with distilled water, then subjected to the treatment applications. The 1-MCP was vaporized according to the methodology of Herrera (2007), where 300 and 600 mg L-1 of 1-MCP was weighed in a 10 mL glass tube, which was sealed with a rubber stopper, through which the hot water (45-50 °C), provided by the manufacturer, was injected. The dissolution of 1-MCP in the hot water generated the release of gaseous 1-MCP in the headspace of the tube. This was introduced into a 2 L sealed chamber containing the fruits; then the chamber was opened to release the compound and immediately sealed for 24 h.
Variables
The evaluated variables were: fruit firmness (N), determined with a digital PCE-PTR200 penetrometer (PEC Ibérica SL, Albacete, Spain); fresh mass loss (%), by measuring the fresh weight of the fruits with a 0.0001 g precision balance (Ohaus, Ohio, OH); total soluble solids (TSS), using a digital Hanna refractometer (Hanna Instruments, Woonsocket, RI), 0 to 85% range at 20 °C; total titratable acidity (TTA; % citric acid), using the formula: % Acidity = (A* B* C)*100/D, where: A = volume of NaOH used, B = normality of NaOH (0.097), C = equivalent weight in g of predominant acid in the fruit (citric acid 0.064 meq g-1), D = weight in grams of the sample used (5 g). The maturity index (MI) was calculated with the TSS /TTA ratio. The color was measured using a Minolta CR-300 reflectance colorimeter (Minolta Co., Japón). Three replications were performed on three fruits directly on the surface at the equatorial axis. CIELab system parameters "L", "a" and "b" were determined. L indicates brightness, where 0 is black and 100 white; values "a" < 0 indicate trend towards green and > 0 into the red; "B" has the same range but values < 0 indicate a tendency towards blue and > 0 towards yellow.
For the extraction and quantification of total carotenoids, approximately 1 g of pulp was weighed, 5 mL of acetone was added, vortexed for 1 min and then centrifuged for 10 min at 4,000 rpm. The supernatant was poured into a 25 mL flask; acetone was added to the pellet, vortexed and then centrifuged. This procedure was repeated three times. Acetone was added to the obtained supernatant to obtain a volume of 25 mL. The absorbance was measured in a spectrophotometer at 450, 663 and 645 nm.
Respiratory rate was measured taking approximately 300 g of fruit which were placed in an airtight 2 L chamber VER BC-2000 (Vernier Software & Technology, United States), in which an infrared CO2 sensor VER CO2-BTA (Vernier Software & Technology, United States) was located, which was connected to a LabQuest2 (Vernier Software & Technology, United States). Every 4 sec for 5 min, CO2 values were recorded. With these values, the slope was calculated, which corresponded to the respiration rate. Also, the fruit weight and volume of the chamber were taken into account to convert the data to mg CO2 kg-1 h-1.
Statistic analysis
An analysis of variance (Anova) was conducted to determine statistical differences between the treatments and to classify them with a Tukey test (P=0.05). Analyses were performed using SAS statistical software v. 9.1e (SAS Institute Inc., Cary, NC).
RESULTS AND DISCUSSION
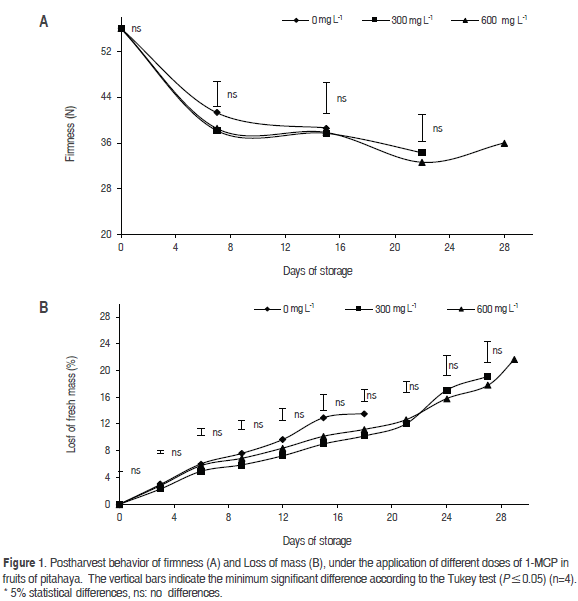
Firmness
In the case of yellow dragon fruit, the loss of firmness at 15 and 22 days after harvest (dah) showed significant differences between the doses of 300 and 600 mg L-1 of 1-MCP; however, these doses did not differ significantly from the dose control, where the fruits only maintained quality for 15 dah (Figure 1a). Still, only the fruits of treatment than received the dose of 600 mg L-1 reached the 28 dah, whereas treatment of 300 mg L-1 only reached 26 dah.
This indicates that the yellow dragon fruit responded to the application of 300 mg L-1 doses of 1-MCP during the first 22 dah, while the dose of 600 mg L-1 extended life 6 more days (until 28 dah); day from which the fruit quality consumer is lost due that this continue to mature normally, with the respective softening. This effect occurs due to the inhibition of the enzymes involved in the degradation of cell walls, such as cellulose, pectin methyl esterase, polygalacturonase, β-1,4-D-glucanase, β-galactosidase, exopolygalacturonase, endopolygalacturonase and polyphenol oxidase (Öskaya et al., 2016; Khan and Singh, 2007), which has been observed in apples (Gago et al., 2015), where applications of 1-MCP reduced the softening of stored fruits, as well as in nectarines (Öskaya et al., 2016) and pears (Li et al., 2016). On the other hand, Rizzolo et al. (2015) reported that pear fruit firmness only began to decline after 20 d of storage, similar to what was found by Li et al. (2016).
Mass loss
This variable showed no significant difference during the storage time, notwithstanding the fact that the application of 1-MCP extended the postharvest shelf-life of the fruits 28 dah, as compared with the control fruits, which only reached 15 dah with consumption quality. The 600 mg L-1 dose showed the greatest loss of mass (9.84%, 11.89%, 13.01% and 16.09%) at 22, 24, 26 and 28 dah. However, this dose was not significantly different from the 300 mg L-1 dose or the control (Figure 1b).
This indicates that the 600 mg L-1 dose of 1-MCP could generate a negative effect on the fruits, increasing the processes of transpiration and respiration and causing further losses of water as compared to the 300 mg L-1 dose, in agreement with the results reported by Serna et al. (2011) for the sweet yellow pitahaya, where an application of 1-MCP had the highest loss of mass. Likewise, Gago et al. (2015) found that the mass loss in apple fruits increased during the storage period equally in treatments with 1-MCP and in untreated fruits. In this regard, Aguayo et al. (2006) found that 1-MCP in pineapples did not affect the values of mass loss and firmness. In addition, it has been proven that when applications of 1-MCP are carried out on fruits stored at room temperature, treatments tend to be less effective against the responses of ethylene (Serek and Sisler, 2005), which coincides with the results found in dragon fruit.
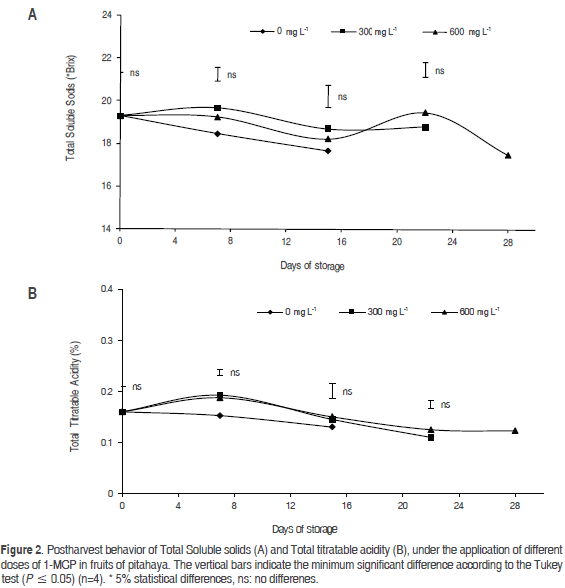
Total soluble solids (TSS)
The behavior of the TSS did not show significant differences during storage (Figure 2a), which implies that the application of 1-MCP does not affect the sugar content of dragon fruit during storage, which is similar to results observed with applications of 1-MCP in the apple (Gago et al., 2015), pear (Rizzolo et al., 2015), plum (Khan and Singh, 2007) and feijoa (Rupavatharam et al., 2015), where 1-MCP had little or no effect on the content of TSS.
It is known that, during the ripening of fruits on the tree, the TSS content increases due to the conversion of organic acids to sugars and the biosynthesis or degradation of polysaccharides in cell walls (Cerqueira et al., 2009), forming part of simple sugars such as glucose, fructose and sucrose, which generates an increase in the content of soluble sugars. Still, the TSS in the fruits decreased after 28 dah in all of the treatments, which probably occurred due to the use of sugars as a respiratory substrate (Paliyath and Murr, 2008).
Contrasting results have been reported for the effect of 1-MCP on TSS because, according to Hofman et al. (2001), 1-MCP increased soluble sugars in papaya, contrary to the results found for the nectarine (Öskaya et al., 2016), guava (Cerqueira et al., 2009), Durio zibethinus (Amornputti et al., 2014 ), apple (Marin et al., 2009), kiwi (Mao et al., 2007) and melon (Han et al., 2015), where 1-MCP delayed the conversion of organic acids to sugars and maintained fewer TSS throughout storage when compared to the control.
Total titratable acidity (TTA)
Statistically significant differences (P<0.05) were obtained during the first two postharvest measurements at 7 and 15 dah between the doses of 300 and 600 mg L-1 of 1-MCP, but they did not show significant differences from the control. The 600 mg L-1 dose had the highest values of TTA (Figure 2b). Similarly, it was observed that the TTA values decreased as the storage time passed, similar to that seen by Obenland et al. (2016) in dragon fruit, which is attributed to the activity of dehydrogenases because organic acids are used as respiratory substrates (Paliyath and Murr, 2008); it has also been shown that 1-MCP affects the metabolism of polymeric carbohydrates (peptic and hemicellulose substances) and decreases the TTA in the apple (Marin et al., 2009), guava (Bassetto et al., 2005), pineapple (Selvarajah et al., 2001), pear (Rizzolo et al., 2015) and tomato (Beckles, 2012); however, the efficiency of 1-MCP may depend on the endogenous levels of ethylene (Zhang et al., 2009).
Maturity index (MI)
This characteristic only had significant differences at 15 dah, where the application of the 600 mg L-1 dose of 1-MCP (MI = 12.6) significantly reduced the MI values as compared with the fruits in the control treatment (MI = 14). The behavior of the MI during storage presented initial values of 12 at harvest and then decreased at 7 dah with a value of 10.4 (probably at the climacteric peak); then it began to increase during the storage period to a value of 16.7 at 28 dah. Increasing MI occur when fruits reach maximum respiratory intensity, associated with energy consumption in the form of ATP and other compounds in order to maintain the homeostasis of the fruits (Piriyavinit et al., 2011), which includes conversion processes for starch into glucose and fructose that are used as respiration substrates (Lima et al., 2011).
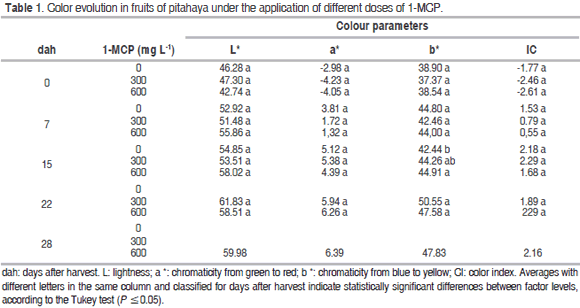
Color
In general, the different color parameters measured in the fruits showed no significant differences in the 1-MCP applications (Table 1), similar to that found in apples (Gago et al., 2015) and feijoa (Rupavatharam et al., 2015); however, it can be seen that the treatments with a dose of 600 mg L-1 had fruits with more lightness because the postharvest shelf-life (7 and 15 dah) was prolonged, indicating that the 1-MCP applications resulted in fruits that had higher luminosity, which slows the loss of quality, which agrees with the results of Villalobos et al. (2011), who stated that 1-MCP is important for increasing the postharvest shelf-life and delaying color changes in the peel.
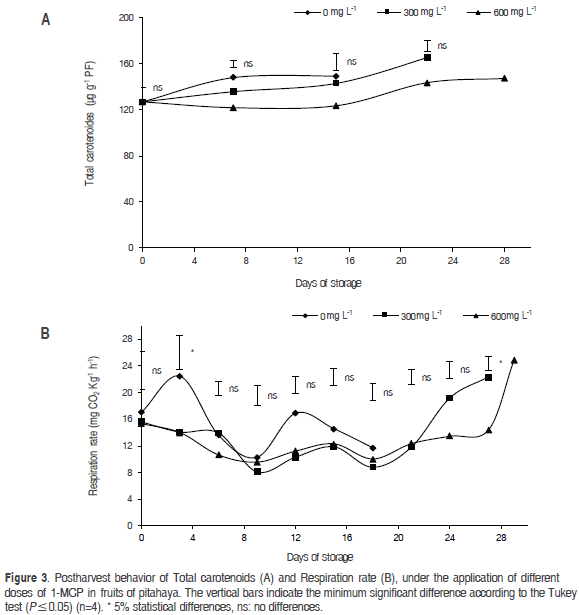
Total carotenoid content
The content of total carotenoids in the fruits had no significant differences between the 1-MCP doses and the control during the first 15 dah (Figure 3a). However, they continued to increase throughout the storage period, reaching values of 179 mg g-1 PF, which means that the application of 1-MCP prolonged the postharvest shelf-life of the fruits, which retained their nutraceutical properties.
Because the fruits were stored under ambient conditions, it is likely that the total carotenoid content increased due to the accelerated loss of chlorophyll, which was expressed as total chlorophyll, from 0.068 mg g-1 PF at the beginning of storage to 0.014 mg g-1 PF at 28 dah, which coincides with that reported in citrus fruits stored at 12 °C (Carmona et al., 2012). Rubinowska et al. (2012) stated that an increase in the synthesis of carotenoids (β-carotene and lycopene) is associated with the ability to scavenge free oxygen radicals that are generated in greater amounts during the oxidative stress seen in the postharvest phase of fruits. However, 1-MCP applications likely slow the increase in carotenoids, which coincides with that reported by Guillén et al. (2007) who showed that ripening tomatoes require the application of much higher levels of 1-MCP in order to extend the shelf life.
Respiratory rate (RR)
This variable showed significant differences between the control and the doses of 300 and 600 mg L-1 of 1-MCP (Figure 3b), with an increased RR in the fruits. Similarly, the 1-MCP doses had similar results during the first 22 dah; towards the end of the postharvest period, differences (27 dah) occurred between the doses of 300 and 600 mg L-1 1-MCP, where the higher doses preserved fruit quality longer. This occurred because, in prolonged periods of storage, fruit tissues synthesize more ethylene receptors, which increases the RR at the end of storage (Serek et al., 2006). However, the 1-MCP greatly reduced the RR during the storage time of the fruits. Similarly, this decrease in CO2 production is consistent with that found in the nectarine (Öskaya et al., 2016), guava (Cerqueira et al., 2009), and melon (Han et al., 2015), where 1 -MCP decreased the respiration rate and significantly delayed the onset of the climacteric peak.
CONCLUSIONS
The 600 mg L-1 dose of 1-MCP was able to maintain the quality of the fruits longer (28 dah), which was reflected in a decreased respiration intensity in the fruits. The application of 1-MCP did not affect the firmness, loss of mass, TSS, TTA, or carotenoids of the fruits. The application of 1-MCP slowed the loss of fruit quality in terms of color because the 600 mg L-1 dose maintained the lightness of the fruits longer and reduced the color changes expressed as a* and b* values.
ACKNOWLEDGMENTS
This work was supported by Colciencias and the Universidad Pedagógica y Tecnológica de Colombia (UPTC) by the national program for training young researchers and innovators 2013 "Virginia Gutiérrez de Pineda" which selected Yuli Alexandra Deaquiz Oyola.
REFERENCES
Aguayo E, Jansasithorn R and Kader AA. 2006. Combined effects of 1-methylcyclopropene, calcium chloride dip, and/or atmospheric modification on quality changes in fresh-cut strawberries. Postharvest Biology Technology 40: 269-278. doi: 10.1016/j.postharvbio.2006.01.016 [ Links ]
Amornputti S, Ketsa S and van Doorn W. 2014. Effect of 1-methylcyclopropene (1-MCP) on storage life of durian fruit. Postharvest Biology Technology 97: 111-114. doi: 10.1016/j.postharv bio.2014.06.011 [ Links ]
Amornputti S, Ketsa S and van Doorn W. 2016. 1-Methylcyclopropene (1-MCP) inhibits ethylene production of durian fruit which is correlated with a decrease in ACC oxidase activity in the peel. Postharvest Biology Technology 114: 69-75. doi: 10.1016/j.postharvbio.2015.11.020 [ Links ]
Bassetto E, Jacomino AP, Pinheiro AL. and Kluge RA. 2005. Delay of ripening of 'Pedro Sato' guava with 1- methylcycloproprene. Postharvest Biology Technology 35: 303-308. doi: 10.1016/j.postharvbio.2004.08.003 [ Links ]
Beckles D. 2012. Factors affecting the postharvest soluble solids and sugar content of tomato (Solanum lycopersicum L.) fruit. Postharvest Biology Technology 63: 129-140. doi: 10.1016/j.postharvbio.2011.05.016 [ Links ]
Carmona L, Zacarias L and Rodrigo M. 2012. Stimulation of coloration and carotenoid biosynthesis during postharvest storage of "Navelina" orange fruit at 12 °C. Postharvest Biology Technology 74: 108-117. doi: 10.1016/j.postharvbio.2012.06.021 [ Links ]
Cerqueira TS, Jacomino AP, Sasaki FF and Amorim L. 2009. Controle do amadurecimento de goiabas 'kumagai' tratadas com 1-metilciclopropeno. Revista Brasileira de Fruticultura 31(3): 687-692. doi: 10.1590/S0100-29452009000300010 [ Links ]
Dueñas Y, Narváez CE and Restrepo LP. 2008. Inhibición de lesiones por frío de pitaya amarilla (Acanthocereus pitajaya) a través del choque térmico: catalasa, peroxidasa y polifenoloxidasa. Acta Biologica Colombiana 13(1): 95 -106. [ Links ]
Gago C, Guerreiro A, Miguel G, Panagopoulos T, Sánchez C and Antunes M. 2015. Effect of harvest date and 1-MCP (SmartFreshTM) treatment on 'Golden Delicious' apple cold storage physiological disorders. Postharvest Biology Technology 110: 77-85. doi: 10.1016/j.postharvbio.2015.07.018 [ Links ]
Guillén F, Castillo S, Zapata PJ, Martínez-Romero D, Serrano M and Valero D. 2007 Efficacy of 1-MCP treatment in tomato fruit: 1. Duration and concentration of 1-MCP treatment to gain an effective delay of postharvest ripening. Postharvest Biology Technology 43: 23-27. doi: 10.1016/j.postharvbio.2006.07.004 [ Links ]
Han C, Zuo J, Wang Q, Xu L, Wang Z, Dong H and Gao L. 2015. Effects of 1-MCP on postharvest physiology and quality of bittermelon (Momordica charantia L.). Scientia Horticulturae 182: 86-91. doi: 10.1016/j.scienta.2014.07.024 [ Links ]
Herrera A. 2007. Emisión de compuestos volátiles durante la postcosecha de frutos de mandarina: Efecto de las bajas temperaturas y del tratamiento con etileno. Ph.D. thesis. Faculty of Sciences, Universidad Autónoma de Madrid, Madrid. 103 p. [ Links ]
Hofman PJ, Jobin-Décor M, Meiburg GF, Macnish AJ and Joyce D. 2001. Ripening and quality responses of avocado, custard apple, mango and papaya fruit to 1-methylcyclopropene. Australian Journal of Experimental Agriculture 41: 567-572. doi: 10.1071/EA00152 [ Links ]
Hu HG, Li XP, Dong C and Chen W. 2011. Effects of wax treatment on quality and postharvest physiology of pineapple fruit in cold storage. African Journal of Biotechnology 10: 7592-7603. doi: 10.5897/AJB10.2474 [ Links ]
Hu HG, Li XP, Dong C and Chen W. 2012. Effects of wax treatment on the physiology and cellular structure of harvested pineapple during cold storage. Journal of Agricultural and Food Chemistry 60(26): 6613-6619. doi: 10.1021/jf204962 [ Links ]
Icontec. 1996. NTC 3554. Frutas frescas: pitahaya. Instituto Colombiano de Normas Técnicas y Certificación, Bogotá [ Links ].
Instituto Colombiano Agropecuario, ICA. 2010. Pitahaya colombiana podría llegar a Corea. En: http://www.ica.gov.co/Noticias/Agricola/2010/Pitahaya-colombiana-podria-llegar-a-Corea.aspx. 1 p; consulta: febrero 2014. [ Links ]
Jiang L, Zhang L, Shi Y, Lu Z and Yu Z. 2014. Proteomic analysis of peach fruit during ripening upon post-harvest heat combined with 1-MCP treatment. Journal Proteomics 98: 32-43. doi: 10.1016/j.jprot.2013.11.019 [ Links ]
Khan A and Singh Z. 2007. 1-MCP regulates ethylene biosynthesis and fruit softening during ripening of 'Tegan Blue' plum. Postharvest Biology Technology 43: 298-306. doi: 10.1016/j.postharvbio.2006.10.005 [ Links ]
Le Bellec F and Vaillant F. 2011. Pitahaya (Hylocereus spp.). pp. 247-271. In: Yahia E. (Ed.). Postharvest Biology and Technology of Tropical and Subtropical Fruits. Woodhead Publishing Series in Food Science, Technology and Nutrition 534 p. doi: 10.1533/9780857092618.247. [ Links ]
Li G, Jia H, Li J, Li H and Teng Y. 2016. Effects of 1-MCP on volatile production and transcription of ester biosynthesis related genes under cold storage in 'Ruanerli' pear fruit (Pyrus ussuriensis Maxim.). Postharvest Biology Technology 111: 168-174. doi: 10.1016/j.postharvbio.2015.08.011. [ Links ]
Lima FV, Saavedra J, Marcos EM and Kluge RA. 2011. Pós-colheita de lichia 'Bengal' tratada com etileno e 1-metilciclopropeno. Ciência Rural 41(7): 1143-1149. doi: 10.1590/S0103-84782011005000095 [ Links ]
Ma G, Wang R, Wang C, Kato M, Yamawaki K, Qin F and Xu H. 2009. Effect of 1-methylcyclopropene on expression of genes for ethylene biosynthesis enzymes and ethylene receptors in post-harvest broccoli. Plant Growth Regulation 57: 223-232. doi: 10.1007/s10725-008-9339-7 [ Links ]
Mao M, Wang G and Que F. 2007. Application of 1-methylcyclopropene prior to cutting reduces wound responses and maintains quality cut kiwifruit. Journal of Food Engineering 78(1): 361-365. doi: 10.1016/j.jfoodeng.2005.10.004 [ Links ]
Marin A, Colonna A, Kudo K, Kupferman E and Mattheis J. 2009. Measuring consumer response to "Gala" apples treated with 1-methylcyclopropene (1-MCP). Postharvest Biology Technology. 51: 73-79. doi: 10.1016/j.postharvbio.2008.06.008 [ Links ]
Obenland D, Cantwell M, Lobo R, Collin S, Sievert J and. Arpia ML 2016. Impact of storage conditions and variety on quality attributes andaroma volatiles of pitahaya (Hylocereus spp.). Scientia Horticulturae 199: 15-22. doi: 10.1016/j.scienta.2015.12.021 [ Links ]
Öskaya O, Yildirim D, Dündar Ö and Tükel S. 2016. Effects of 1-methylcyclopropene (1-MCP) and modified atmosphere packaging on postharvest storage quality of nectarine fruit. Scientia Horticulturae 198: 454-461. doi: 10.1016/j.scienta.2015.12.016 [ Links ]
Paliyath G and Murr D. 2008. Biochemistry of fruits. pp. 19-50. En: Paliyath, G, Murr D, Handa A and Lurie. S. (eds.). Postharvest Biology and Technology of Fruits, Vegetables and Flowers. Wiley-Blackwell Publishing. Iowa, USA. 482p. [ Links ]
Piriyavinit P, Ketsa S and van Doorn W. 2011. 1-MCP extends the storage and shelf life of mangosteen (Garcinia mangostana L.) fruit. Postharvest Biology Technology 61(1): 15-20. doi: 10.1016/j.postharvbio.2011.02.007 [ Links ]
Rizzolo A, Grassi M and Vanoli M. 2015. Influence of storage (time, temperature, atmosphere) on ripening, ethylene production and texture of 1-MCP treated 'Abbé Fétel' pears. Postharvest Biology Technology 109: 20-29. doi: 10.1016/j.postharvbio.2015.06.003 [ Links ]
Rubinowska K, Pogroszewska E. and Michalek W. 2012. The effect of polyamines on physiological parameters of post-harvest quality of cut stems of Rosa 'red berlin'. Acta Scientiarum Polonorum Hortorum Cultus 11(6): 81-93. [ Links ]
Rupavatharam S, East A and Heyes J. 2015. Re-evaluation of harvest timing in 'Unique' feijoa using 1-MCP andexogenous ethylene treatments. Postharvest Biology Technology 99: 152-159. doi: 10.1016/j.postharvbio.2014.08.011 [ Links ]
Selvarajah S, Bauchot A and John P. 2001. Internal browning in cold-stored pineapples is suppressed by a postharvest application of 1-methylcyclopropene. Postharvest Biology Technology 23: 167-170. doi: 10.1016/S0925-5214(01)00099-0 [ Links ]
Serek M, Sisler EC. 2005. Impact of 1-MCP on postharvest quality of ornamentals. APEC Symposium on Quality Management of Postharvest Systems Proceedings. pp. 121-8. [ Links ]
Serek M, Woltering EJ, Sisler EC, Frello S and Sriskandarajah S. 2006. Controlling ethylene responses in flowers at the receptor level. Biotechnology Advances 24: 368-381. doi: 10.1016/j.biotechadv.2006.01.007 [ Links ]
Serna L, Torres LA and Ayala A. 2011. Efecto del empaque y del 1-MCP sobre características físicas, químicas y fisiológicas de pitahaya amarilla. Biotecnología en el Sector Agropecuario y Agroindustrial 9(2): 139-149. [ Links ]
Toivonen PMA. 2008. Application of 1-methylcyclopropene in fresh-cut/ minimal processing systems. HortScience 43: 102-105. [ Links ]
Villalobos M, Biasi W, Mitcham E and Holcroft D. 2011. Fruit temperature and ethylene modulate 1-MCP response in 'Bartlett' pears. Postharvest Biology Technology 60: 17-23. doi: 10.1016/j.postharvbio.2010.11.005 [ Links ]
Watkins CB. 2008. Postharvest effects on the quality of horticultural products: Using 1-MCP to understand the effects of ethylene on ripening and senescence processes. Acta Horticulturae 768: 19-32. doi: 10.17660/ActaHortic.2008.768.1 [ Links ]
Xiaotang Y, Jun S, Lina D, Forney C, Leslie CP, Sherry F, Wismer P and Zhaoqi Z. 2016. Ethylene and 1-MCP regulate major volatile biosynthetic pathways in apple fruit. Food Chemistry 194: 325-336. doi: 10.1016/j.foodchem.2015.08.018 [ Links ]
Zhang Z, Huber DJ, Hurr BM and Rao J. 2009. Delay of tomato fruit ripening in response to 1-methylcyclopropene is influenced by internal ethylene levels. Postharvest Biology Technology 54: 1-8. doi: 10.1016/j.postharvbio.2009.06.003 [ Links ]
Zhu X, Shen L, Fu D, Si Z, Wu B, Chen W and Li X. 2015. Effects of the combination treatment of 1-MCP and ethylene on the ripening of harvested banana fruit. Postharvest Biology Technology 107: 23-32. doi: 10.1016/j.postharvbio.2015.04.010 [ Links ]