Gmelina arborea Roxb. ex Sm is one of the most promising forest species known worldwide in the use of different reforestation programs due to its rapid growth, it is a secure source of raw wood in a short time. This plant behaves as an opportunistic in rainforests, and it is classified as a pioneer (Moya, 2004; Rojas et al., 2004; Hernández and Salas, 2008). This species is currently being used in commercial forest plantations, and it is commonly used on the Atlantic coast with around 11,000 planted hectares in Colombia (Cadena and Guauque 2009; Romero, 2004). The growing importance that this species is having is due to relative homogeneity and stability of its wood which makes it adequate for obtaining cellulose as raw material for paper making (Dvorak, 2004). Other uses are furniture manufacturing (Moya, 2004; Romero, 2004), living fences, windbreaker curtains and timber boundaries (Rojas et al., 2004).
Gmelina arborea is a perennial plant, which makes it a key step in nursery seedling production that determines the success of the future plantation, making necessary to look for alternatives that improve the development and fertilization at this stage of the production cycle. This is how the use of Arbuscular Mycorrhizal Fungi (AMF) becomes a viable option to fulfill this objective because of these microorganisms support the growth, survival and seedling development in different vegetable species under tropical conditions (Osorio, 2011; Ramírez-Gil et al., 2013; Ramírez-Gil et al., 2014; Ramírez-Gil et al., 2015).
AMF improves the growth of its host, especially in soils with low levels of phosphorus and under stress due to lack of water (Yano and Takaki, 2005; Osorio, 2011; Osorio et al., 2017). This symbiosis also helps the absorption of other nutrients, improves the response to the attack of pathogenic microorganisms, among other benefits (Osorio, 2011; Ramírez-Gil et al., 2014; Osorio et al., 2017). It is expected that inoculation with AMF in G. arborea improves the productivity of this species in Colombia since most areas planted are arid zones and have soils with low natural fertility (Cadena and Guauque, 2009). Besides they are an economical and friendly alternative to protect the environment by reducing fertilizer applications on soil, especially with phosphorus (P) fertilizers (Osorio, 2011; Osorio et al., 2017).
The first step for an adequate use of AMF in agriculture consists in determining the relationship between microorganism strains with the host species, which can be quantified through mycorrhizal colonization and dependency (Osorio, 2011; Ramírez-Gil et al., 2013; Ramírez-Gil et al., 2014; Ramírez-Gil et al., 2015; Osorio et al., 2017). Given the need to understand the symbiotic relationship between G. arborea and different AMF species, this work aimed to determine the mycorrhizal dependency and colonization, so as the plant development when was inoculated with five AMF species (R. fasciculatum, R. aggregatum, R. irregulares, G. fistulosum, and E. colombiana) in three different P levels in a soil solution (0.002, 0.02, and 0.2 mg L-1).
MATERIAL AND METHODS
Location
The experiment was performed between 2012 and 2013, in the Environmental Microbiology and Tropical Phytotecnia laboratories and greenhouse at the Universidad Nacional de Colombia, in Medellín (6°15'51.7"N, 75°34'40.1"W, and 1495 m of altitude). The greenhouse environmental conditions were: temperature at 18-22 °C, relative humidity in a range of 75-95%, and photosynthetically active radiation of 650-1920 μmol photons m-2 s-1.
Obtaining soil and seedlings
The soil used in these assays was collected from the subsurface horizon (Bt) in Volador hill, Medellín, Antioquia, Colombia (6°15'56.4"N, 75°34'57.1"W). The soil sample was collected under 30-60 cm of deep to reduce the influence of native organic P available on the soil (Ramirez-Gil et al., 2015). The soil sample was analyzed based on protocols used in the Soil Fertility Laboratory at Universidad Nacional de Colombia-Sede Medellín. Soils results were: sand: 65%, silt: 15%, clay: 20% (Bouyoucos); pH 4.8 (water 1:2, v:v); Al: 0.7 cmolc kg-1 (KCl 1 M); Ca, Mg, K: 1.2, 0.5, 0.02 cmolc kg-1 (1 M ammonium acetate); Fe, Mn, Cu, Zn: 37, 3, 1, 2 mg kg-1 (Olsen-EDTA), B: 0.1 mg kg-1 (hot water); S: 9 mg kg-1 (0.008 M Calcium phosphate); NO3 -: 2 mg kg-1 (0.025 M ammonium sulfate); NH4 +: 2 mg kg-1 (KCl 1M); P: 2 mg kg-1 (Bray II); P soluble: 0.001 mg L-1 (0.01 M of CaCl2.) and organic material: 2% (Walkley and Black method).
The soil was sterilized following the recommendations reported by Ramírez-Gil et al. (2015) to eliminate microorganism interferences. Using an autoclave at 0.1 MPa and 121 °C, for two cycles of one hour each. Besides, the aim to improve the AMF ecological condition (Osorio et al., 2017), lime was added to adjust soil pH at 5.6, based on lime incubation method (Uchida and Hue, 2000). On the other hand, KH2PO4 was added to achieve a soil solution of P concentration of 0.002 0.02 and 0.2 mg L-1, based on P sorption isotherm (Fox and Kamprath, 1970), which is considered optimal for the studied mycorrhizal dependence and activity (Habte and Manjunath, 1991; Osorio 2011; Osorio et al., 2017).
Seeds of G. arborea were collected from healthy trees in Santa Fe de Antioquia at the Cotove Agriculture Experimental Station (6°31'55.2"N, 75°49'33.8"W) of Universidad Nacional de Colombia Sede Medellín. The process of extraction, cleaning, disinfection, and germination was developed according to the indications reported by Ramírez-Gil (2017). The plants were maintained in autoclaved quartz until the experiment was assembled.
Obtaining microorganisms and their inoculation
The AMF strains used were Rhizoglomus fasciculatum, Rhizoglomus aggregatum, and Rhizoglomus irregulare. Originally provided by Dr. M. Habte from University of Hawaii (Honolulu, USA). They were subsequently multiplied following the indication of Environmental Microbiology Laboratory at the Universidad Nacional de Colombia Sede Medellín, using roots of sorghum plants. Glomus fistulosum and Entrophospora colombiana strains were supplied by a private laboratory and multiplied based on the indication reported before.
The inoculation in soil was developed based on methodology reported by Ramírez-Gil et al. (2015) using plastic pots containing 2 kg of the chosen soil. In each, 35 g of crude inoculum per kg of soil was applied. The inoculum presented a concentration of 45-50 infective mycorrhizal propagules per g of soil (Table 1), determined according to the most probable number method (Porter, 1979). In controls, an autoclaved (120 °C, 0.1 MPa, for 60 min) crude inoculum (35 g kg-1) and 10 mL of a suspension (10%) obtained from inoculant filtered through a 10 µm filter paper were used (Ramírez-Gil et al., 2015). Then two seedlings were transplanted into plastic pots, a month later one of the plants was removed. The plants were kept for three months in net-house conditions under 50-60% of the soil's maximum water retention capacity. Every week 50 mL of a P-free Hoagland solution was added (Ramírez-Gil et al., 2015).
Colonization and mycorrhizal dependence and development of Gmelina arborea to five species of AFM
A completely randomized design was used, with a factorial arrangement 6×3, five strains of AMF + control (uninoculated plans), and three doses of phosphorus in the soil solution, with five replicate per each treatment and twice through time. Experimental units consisted of three pots with one plant each.
Ninety days after the treatment started, biometrics and symbiotic variables were evaluated. The height (cm) was measured using a Mitutoyo Digimatic Caliper®, dry biomass (g) was quantified in by Binder® stove at 60 °C for 72 h, leaf area (cm2) was measured using a LI-Cor® LI 3000A foliar area meter, and foliar phosphate concentration (%) was determined by non-destructive samples using the blue molybdate method (Murphy and Riley, 1962; Aziz and Habte, 1987). Mycorrhizal Colonization (MC) was evaluated based on the following steps (i) roots were discolored using KOH (10%) for 24 h (Phillips and Hayman, 1970), (ii) coloration of roots by fuchsine acid staining (0.15%) for 48 h (Kormanik et al., 1980), (iii) excess of fuchsine on roots were removed using glycerin-lactic acid-water (0.7-0.2-0.1 v:v), and (iv) intensity of mycorrhizal colonization using the interception lines method (Giovannetti and Mosse, 1980). Mycorrhizal dependency (MD) was calculated based on total dry material value, using equation 1 proposed by Plenchette et al. (1983). MD classification was performed following the indications reported by Habte and Manjunath (1991), MD values of 75% or higher, 50-75%, 25-50%, less than 25% and 0% represents very highly, highly, moderately, marginally, and independent mycorrhizal dependency, respectively.
Statistics analysis of data
For each variable evaluated, data homoscedasticity and normality were determined by Levene and Kolmogorov Smirnov methods. Once homoscedasticity and normality were proved, an ANOVA test was performed. Means were compared with the Tukey test (P<0.05). The evaluation was performed using the R software (R Core Team, 2013).
RESULTS AND DISCUSSION
Mycorrhizal colonization and dependency of Gmelina arborea with five AMF species
All plants of G. arborea inoculated with R. fasciculatum, R. aggregatum, R. irregulare, G. fistulosum, and E. colombiana presented mycorrhizal colonization, which was evidenced by the presence of hyphae in these fungi, particularly within intracellular spaces in the root. In uninoculated plants, no associated structures of AMF were found (Figure 1A and B).

Figure 1 Presence and absence of arbuscular mycorrhizal fungi in roots of Gmelina arborea. A. root inoculated with an Arbuscular Mycorrhizal Fungi (AMF) (Rhizoglomus fasciculatum) (40x); B. root uninoculated (40x).
Most mycorrhizal colonization (P<0.05) in G. arborea was found with inoculation of R. fasciculatum and R. aggregatum under 0.002 and 0.02 mg L-1 of P in the soil solution. These values were 62.5-55.5% and 55.2-54.5%, respectively. In decreasing order and without statistical differences within them (P>0.05), were found the inoculation R. irregulare, and G. fistulosum under 0.02 and 0.002 mg L-1 of P in the soil solution, with values of 38.3-36.2% and 35.3-34.8%, respectively. E. colombiana inoculation showed the lowest values (P<0.05) concerning other AMF species that were evaluated with a 22.3 and 26.2% of colonization under 0.02 and 0.002 mg L-1 of P in the soil solution, respectively. Also, the mycorrhizal colonization of these five AMF species under 0.2 mg L-1 of P did not exceed 5.9% (Figure 2A).
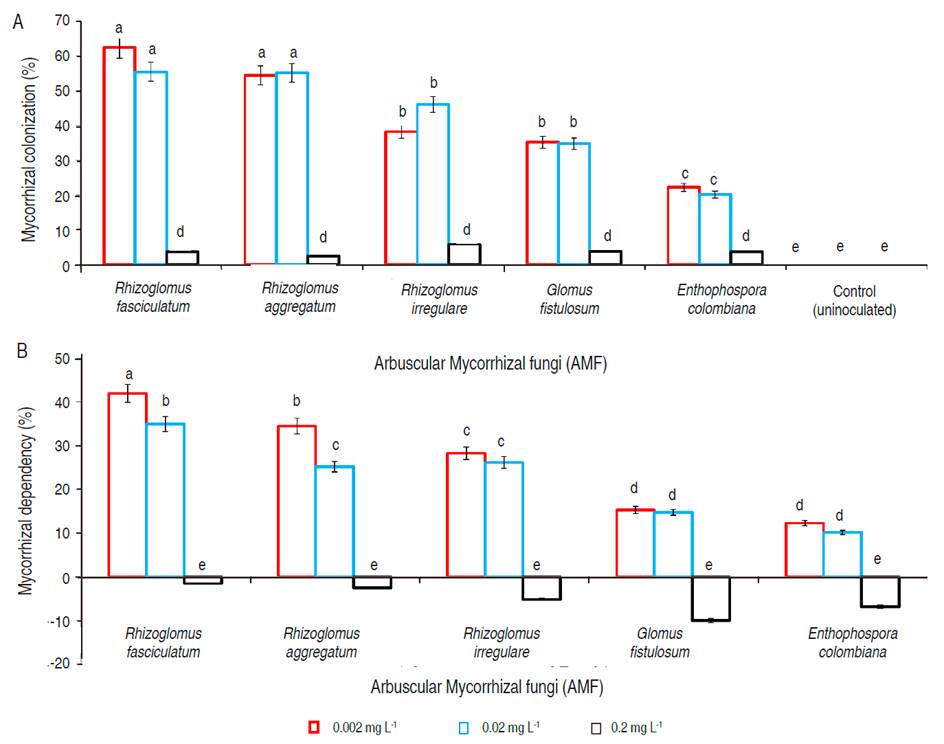
Figure 2 A. Mycorrhizal colonization; B. Mycorrhizal dependence in Gmelina arborea inoculated with five species of Arbuscular Mycorrhizal Fungi (AMF). P levels evaluated in the soil solution were 0.002, 0.02 and 0.2 mg L-1. Statistical analyzes were performed for each level of P and each AMF species evaluated. Equal letters on the bars represent no significant differences with a significance level of 95%. Lines on the bars represent standard error of the mean.
Based on Plenchete et al. (1983) formula (equation 1), and determined according to the classification criteria of Habte and Manjunath (1991), the mycorrhizal dependency (MD) in G. arborea, showed that this species presents a moderate dependency with respect to R. fasciculatum, R. aggregatum, and R. irregulare under P soil solution of 0.02 and 0.002 mg L-1 with values of 42.3-35.1%, 34.5-26.2%, and 28.3-26.2%, respectively (Figure 2B). Meanwhile, G. fistulosum and E. colombiana MD was classified as marginally dependent for P levels of 0.02 and 0.002 mg L-1 with values of 14.8-15.3 and 10.2-12.3%, respectively. In this sense the G. arborea presented the highest MD to species of AMF R. fasciculatum (P<0.05) (Figure 2B). For the AMF species evaluated and with a P level of 0.2 mg L-1 in the soil solution, the MD values in G. arborea were negative, indicating that under these conditions of P in the soil the symbiotic relation becomes adverse for the plant (Figure 2B).
The high MC and medium MD found in G. arborea show that this species needs positive symbiotic relations with AMF to achieve better development and survival, resulting in an improvement of the plant development under these circumstances. This relation was highly conditioned by the AMF species and the content of P in the soil solution, the best results were associated with P levels of 0.002 and 0.02 mg L-1 and R. fasciculatum inoculation, followed by other associated species of the Glomus genera. These results coincide with other works in different species, where inoculation with this same strain (R. fasciculatum) and under these soil conditions (P dose, 0.02 mg L-1) presented high values of mycorrhizal colonization and dependency (Ramírez-Gil et al., 2013; Ramírez-Gil et al., 2014; Ramírez-Gil et al., 2015). According to Sieverding (1991), Rhizoglomus is one of the most aggressive and effective generous for the establishment of symbiotic relations with different taxonomic plant groups under natural conditions.
The best development from symbiotic relation of G. arborea and the AMF species was found in a soil P dose of 0.02 mg L-1. It takes place, thanks to the fact that under this condition the symbiotic relationship is favored, leading to a closer interaction and dependency where the plant through radial exudates supplies an energy source to the fungus, which increases exploration surface in the plant root system and improves nutrient absorption, especially of P (Osorio, 2011; Osorio et al., 2017). The P levels in soil solution and foliar tissue in the host are so important that they can activate or deactivate P transporters in mycorrhizal hyphae, directly affecting P plant absorption by this mechanism (Zandavalli et al. 2004; Osorio, 2011; Osorio et al., 2017).
Growth and development of Gmelina arborea inoculated with five strains of AMF in P levels in soil solution
The height (P<0.05) of G. arborea was found in treatments with inoculation of R. fasciculatum, followed by R. aggregatum, R. irregulare, G. fistulosum, and E. colombiana, for P levels in a soil solution of 0.02 and 0.002 mg L-1, respectively. Meanwhile, for 0.2 mg L-1 of P in soil solution and control (no-inoculated plant), the values were not statistically different (P>0.05) and presented the lowest height (P<0.05) (Figure 3A).
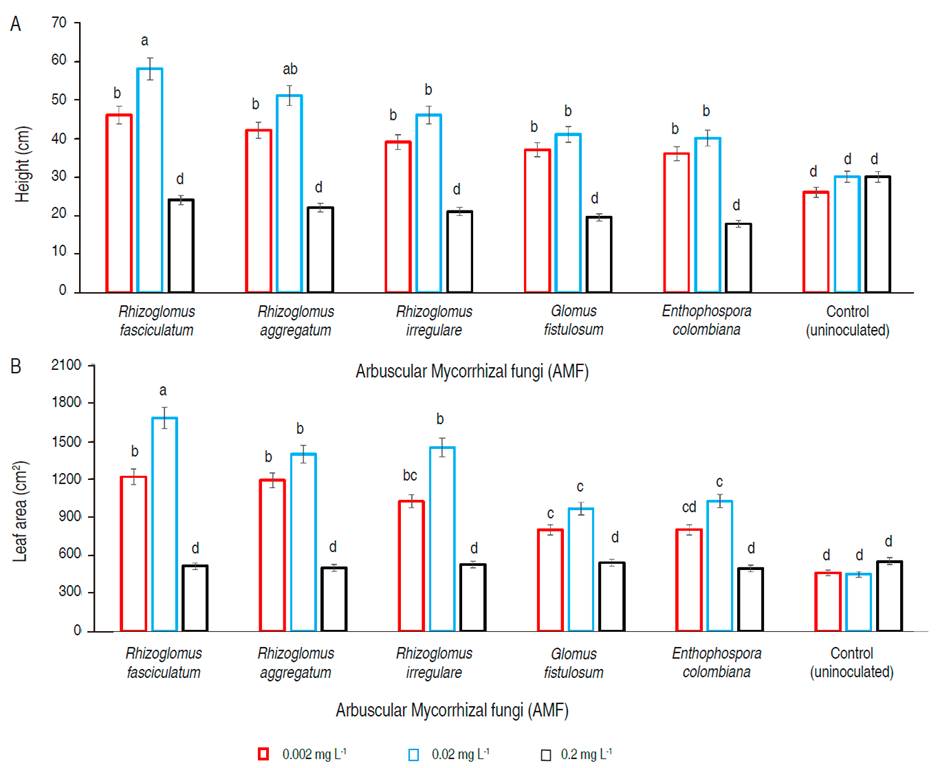
Figure 3 A. Height; B. leaf area of Gmelina arborea inoculated with five species of arbuscular mycorrhizal fungi. P levels evaluated in the soil solution were 0.002, 0.02 and 0.2 mg L-1. Statistical analyzes were performed for each level of P and each AMF species evaluated. Equal letters on the bars represent no significant differences with a significance level of 95%. Lines on the bars represent standard error of the mean.
Leaf area presented same behavior that height, where the greatest values (P<0.05) were for P level of 0.02 mg L-1 in soil solution, and inoculated with R. fasciculatum (1668 cm2), followed by R. aggregatum (1394.4 cm2) and R. irregulare (1447.4 cm2). No statistical differences were found (P>0.05) among G. fistulosum (964.2 cm2) and E. colombiana (1024.4 cm2), and the control (447.3 cm2) presented the lowest values (P<0.05) (Figure 3B).
Total biomass was significantly greater (P<0.05) when G. arborea was inoculated with AMF species in the soil that contained 0.02 mg L-1 of P, followed by P dose of 0.002 mg L-1 and with less value when P concentration was 0.2 mg L-1 (Figure 4A). Concerning the AMF strains evaluated, the biggest biomass (P<0.05) was obtained when plants were inoculated with R. fasciculatum, followed by inoculation of R. aggregatum, R. irregulare, and G. fistulosum. Meanwhile, treatment with E. colombiana did not show a statistical difference (P>0.05) with G. fistulosum, with these two strains the biomass was inferior (P<0.05) to the other three Rhizoglomus species, but superior (P<0.05) to the uninoculated plants (control) (Figure 4A).
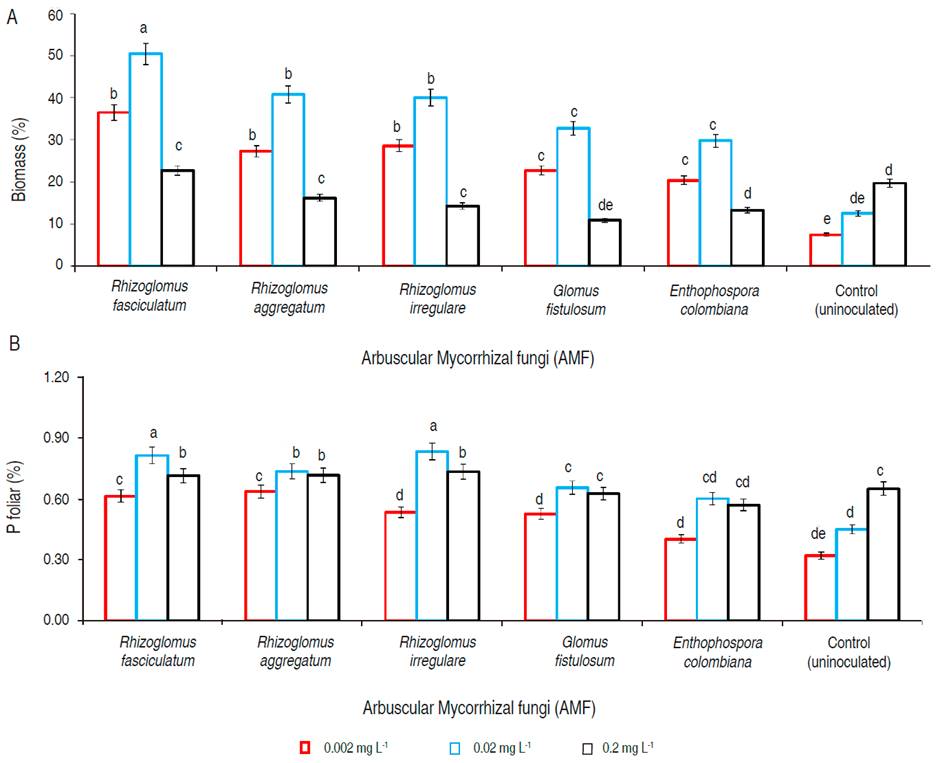
Figure 4 A. Biomass; B. foliar P of Gmelina arborea inoculated with five species of arbuscular mycorrhizal fungi. P levels evaluated in the soil solution were 0.002, 0.02 and 0.2 mg L-1. Statistical analyzes were performed for each level of P and each AMF species evaluated. Equal letters on the bars represent no significant differences with a significance level of 95%. Lines on the bars represent standard error of the mean.
In the case of foliar P (Figure 4B) the highest concentration (P<0.05) was found when the soil had a P level of 0.02 mg L-1 and the plants were inoculated with R. fasciculatum and R. irregulare. They were followed by treatments associated with the same P concentration and the other inoculated AFM strains. Control and plants inoculated with all strains under phosphorus level of 0.2 mg L-1 did not show statistical differences (P>0.05) to the group above mentioned. All treatments associated with AMF inoculation and a P soil level of 0.002 mg L-1 were classified as a third group. The lowest P amount (P<0.05) were presented in the control with a P concentration of 0.002 and 0.02 mg L-1 (Figure 4B).
Same to MC and MD, growth and development of G. arborea was associated with P concentration in soil solution and specific AMF species inoculated. The highest values were obtained under 0.02 mg L-1 of P in the soil solution and R. fasciculatum presence. On the other hand, plant growth and development improved with all AMF treatments, and a higher P concentration in plant leaves was found concerning the plants that were not inoculated. These results agree with Hernández and Salas (2008) findings, indicating that inoculation of G. arborea with R. fasciculatum presented an increase in height, diameter, foliage weight, and roots of 10, 6.2, 6.7 and 44.7%, respectively. It also showed an improvement in inoculated seedlings behavior at nursery stage when transplanted to field.
Many comparative advantages were found in G. arborea when it established a positive symbiotic relation with AMF. Given these interactions, the plant improves its ability to increase the P absorption among other beneficial effects not evaluated in this study, especially under low P levels in soil solution (Osorio, 2011, Osorio et al., 2017). This effect has been evaluated in many other species of economic importance for the Colombian tropics, in which all show better growth and development when the symbiotic relationship is successfully generated (Osorio, 2011; Osorio et al., 2017; Ramírez-Gil et al., 2013; Ramírez-Gil et al., 2014; Ramírez-Gil et al., 2015).
This work is a pioneer in reporting a detailed study of the interactions between G. arborea and five AMF species, it is a fundamental part of the search process for sustainable alternatives for fertilization, where the use of AMF may compensate the low nutrient levels in tropical soil and systems, especially P (Randhawa et al., 2006; Osorio, 2011; Osorio et al., 2017). The use of this biotechnology may help to decrease the use of chemical fertilizers with many economic and environmental benefits since the frequent use of these compounds increase production cost and could have adverse effects on the environment (Brady and Weil, 1999).
The inoculation of the strains R. fasciculatum, R. aggregatum, and R. irregulare is recommended during nursery stage which may increase the yield of the productive systems with this species since its cultivation is carried out in Colombia with low levels of natural fertility (Cadena and Guauque, 2009). Besides, it shows a high capacity of P retention, limiting phosphoric fertilization because the phosphate ion is rapidly precipitated and absorbed (Osorio, 2011; Osorio et al., 2017).
Moreover, understanding that the symbiotic relations with AMF show comparative advantages such as better adaptability to adverse conditions like drought, tolerance to disease attack among others (Yano and Takaki, 2005; Ramírez-Gil et al., 2014; Osorio et al., 2017). It could be a solution to face adverse effects of climate change, each time more frequent and intense, by alleviating the devastating effects on some vegetable species, especially those which are in tropical zones where climate variability is a problem that threatens the sustainability of different production systems.
CONCLUSIONS
The interaction of G. arborea and the five AMF species evaluated favored its development under low values of P (0.02 and 0.002 of mg L-1). The best results were obtained with R. fasciculatum, R. aggregatum and R. irregulare strains. It is important to continue the research with the objective of improving AMF usage in forest plantation in order to improve the sustainability in tropical areas with infertile soils.
















