Yacon (Smallanthus sonchifolius) is a tuber native from the Andes, cultivated since pre-Inca times in the inter-Andean valleys between 1500 and 2500 m.a.s.l. This tuber has multiple health benefits such as reducing of glycemic index, triglycerides and cholesterol levels, risk of colon cancer and kidney problems; help to weight control; increase the assimilation of calcium in bones; strengthening immune system response; skin rejuvenation; restoration of intestinal microflora; among others (Valentová and Ulrichová, 2003). Yacon contains between 10% and 14% of dry matter. It consists mainly of carbohydrates (70-80%), mostly fructooligosaccharides (FOS), a particular type of inulin with shorter-chain of fructans and a degree of polymerization 3-5. It also contains between 5% and 15% of sucrose; less than 5% glucose; and a significant content of antioxidants, mainly chlorogenic acid (Grancieri et al., 2017; Campos et al., 2012). Besides, other research reported between 2.4% and 4.3% of protein content and between 0.14% to 0.43% of lipids (Hermann et al., 1997).
The sweetening power of yacon with a minimum contribution of calories has generated great agro-industrial interest as a dietary supplement and natural sweetener (Bosscher, 2009). However, fresh roots have a short shelf-life because of their high water activity, showing values of 0.994±0.001; moisture content higher than 89.01% (Scher et al., 2009); type and concentration of phenolic compounds; and a high activity of polyphenol oxidase, peroxidase, and fructan 1-exohydrolase enzymes (Castillo, 2015); which all together hinder its processing and transformation.
Suspensions are thermodynamically unstable, which is directly related to different forces: van der Waals (attractive), electrostatic (repulsive), steric (absorbable macromolecules), hydration, hydrophobic, and phase separation (Piorkowski and Mcclements, 2013; Mirhosseini et al., 2008), being flocculation, coalescence, sedimentation, and Ostwald ripening the most common mechanisms (Larsson et al., 2012). In suspensions, physical-chemical instability is manifested mainly by rapid sedimentation, which can be controlled by a homogenization process that reduces particle size (Hennart et al., 2010) and increases the system’s viscosity (Bayod et al., 2008). On the other hand, viscosity and total solids of the suspension affect the pumping capacity and compromises technological characteristics of the operation (Mirhosseini et al., 2008).
According to the above, the aim of this research was to evaluate the influence of composition and homogenization process on the stability of colloidal yacon suspensions for later use in spray drying.
MATERIALS AND METHODS
Materials
Yacon tubers harvested at commercial maturity in the Municipality of Santa Elena (Antioquia) were used. Xanthan gum (XG) was used as thickener and stabilizer. Ascorbic acid and food grade citric acid were used as regulators of the suspension acidity.
Preparation of suspensions
Batches of 3 kg of yacon suspensions (YS) were prepared. Tubers stored at 25 °C were disinfected in 50 ppm NaClO solution, then they were peeled, chopped and immediately passed through an FPSTJE317 Oster® Juice extractor, where insoluble partially disintegrated material was separated from its extract. Subsequently, both were poured into a container and the first homogenization was done using an L5M-A Silverson vertical homogenizer at maximum speed (10200 rpm) for 10 min, keeping the container in a water bath at approximately 25 °C and adding the mixture of ascorbic and food grade citric acid (ratio 1:1); afterward, XG was slowly added. The resulting suspension, in this preliminary phase, was submitted to a second homogenization using an ST REGIS 3DD13-2941 Chicago USA high-pressure piston homogenizer, at 1200 psi during the time set in the experimental design to reduce particle size and improve YS stability.
Determination of the dependent variables
The zeta potential (ζ) was determined in the yacon suspension diluted in deionized water (1:100), using a Zetasizer Nano ZS90 (Malvern Instruments Ltd., Worcester, UK) at 25 °C (Rezvani et al., 2012). The spectral absorption stability index (R) was determined in a UV-Visible spectrophotometer (Thermo Scientific Evolution 60), from absorbance ratio of 800 and 400 nm (R=A800/A400) (Song et al., 2002) applied to diluted YS (1:100). Viscosity (μ) was determined on a rheometer (Brookfield DV-III Ultra, Brookfield Engineering Laboratories, Inc., USA) coupled with a Brookfield thermostatted bath model TC-502, RV5 spindle, speed from 0.01 to 100 rpm and reported at 100 rpm (Mirhosseini et al., 2008). Particle size was reported as percentiles D10, D50, and D90; using a Mastersizer 3000 (Malvern Instruments Ltd., Malvern, Worcestershire, United Kingdom) (Millqvist-Fureby and Smith, 2007). The color was determined with the CIELab coordinates, where L* measures luminosity, a* measures green (-) to red (+) chromaticity, and b* measures blue (-) to yellow (+) chromaticity; using an SP64 - X-Rite spectrophotometer, illuminant D65, and observer of 10°.
Statistical design
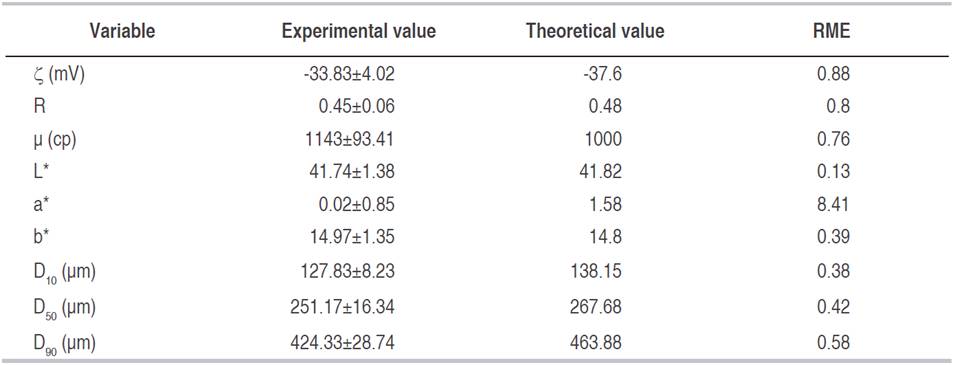
Response surface methodology with a central composite design (α=1) was used, considering as independent variables: homogenization time (TH) (4-6 min), XG added (0.1-0.2%) and acid (0.1-0.3%). The dependent variables were ζ, R, μ, color (coordinates CIELab), and particle size (D10, D50, and D90). All the variables were measured by triplicate. Results were analyzed through an Analysis of Variance with a confidence level of 95%, using Sofware Design Expert 8.0 (Stat-Ease, Inc USA). Significant differences and correlations were made by least significant difference (LSD) (level of significance ≤5%) and Pearson correlation, respectively. Experimental optimization was performed considering the statistical results obtained, and according to criteria that favored suspension stability: maximize (L*, a*, b*), minimize (ζ, D10, D50, and D90), and μ set at 1000 cP.
Mathematical modeling
A mathematical model was made using a polynomial model of order 2 (equation 1), where Y is the dependent variable, β0 is the constant, βA, βB, and βC are linear coefficients; βA 2, βB 2, and βC 2 are quadratic coefficients; βAB, βAC, and βBC are factors interaction coefficients. The adequacy of models was carried out using the lack of fit test and regression coefficient (R2).
Experimental values obtained from three additional experiments were compared with optimal conditions of the process in order to verify the accuracy of dependent variables obtained by the mathematical models.
RESULTS AND DISCUSSION
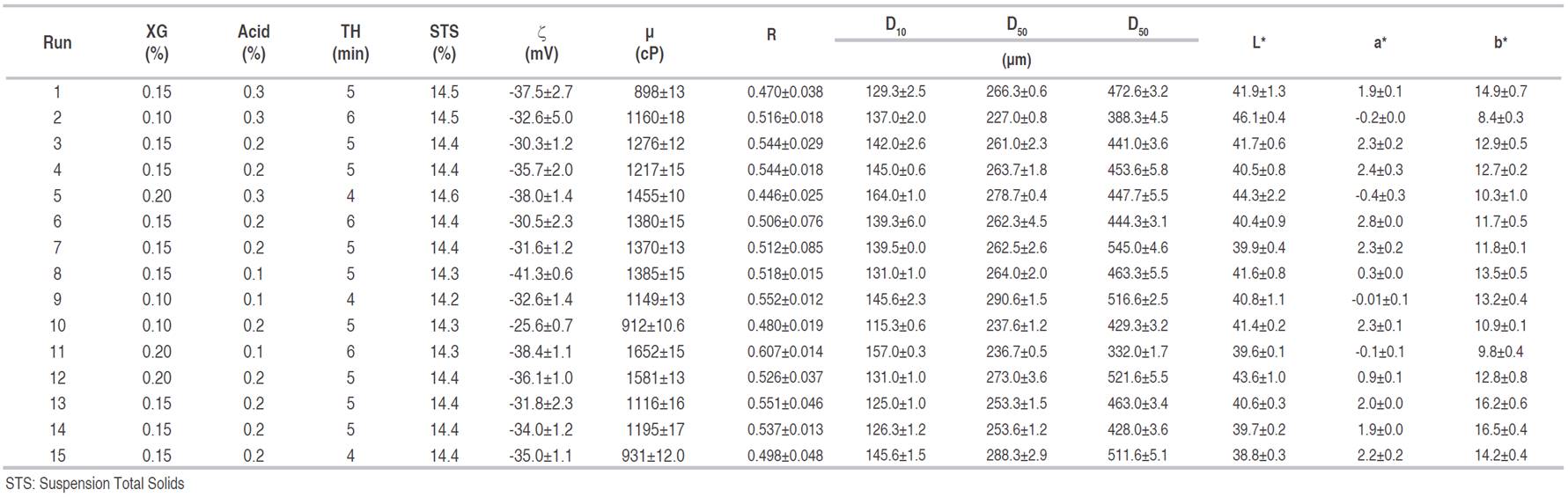
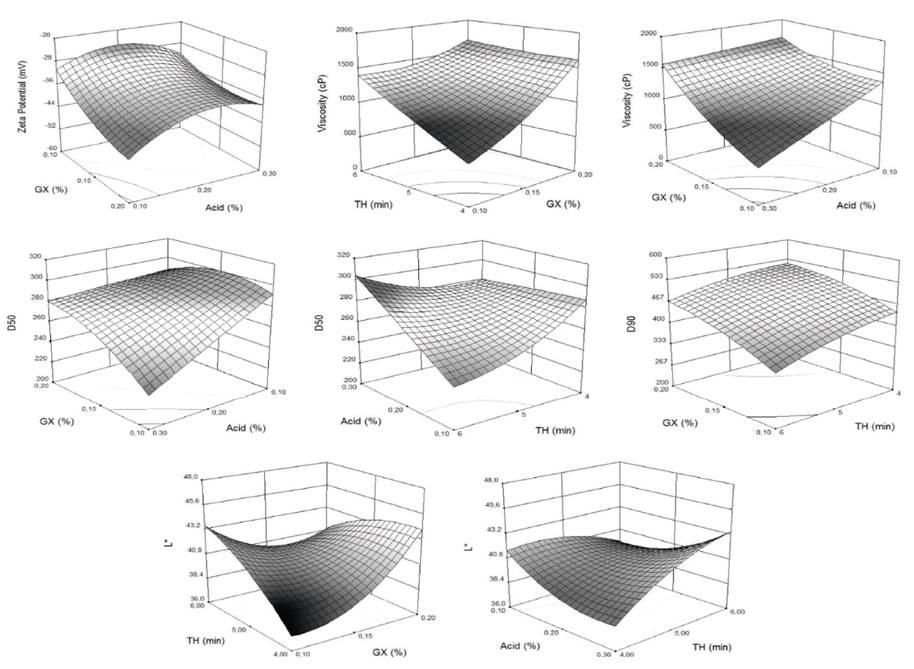
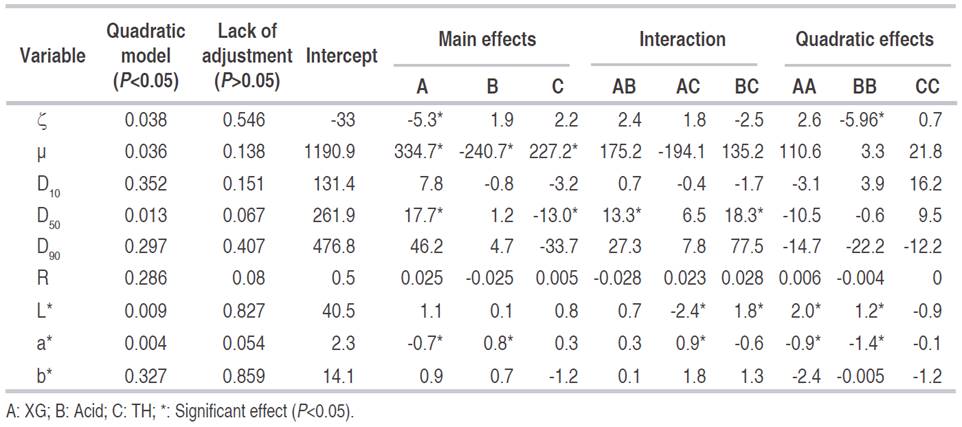
Table 1 shows mean values and standard deviation of YS dependent variables according to experimental design. The ζ of YS varied between -25.1 and -45.4 mV, identifying a predominance of negative charges outside the layer of coions, and a high density of positive charges in the double electric layer. Absolute values above 25 mV indicated that the particles were highly charged, which favors electrostatic repulsion forces and therefore, better stability of YS (Dickinson, 2009; Estevinho et al., 2014). Statistically significant (P<0.05) response surface graphs of dependent variables are shown in Figure 1. On the other hand, Table 2 shows significant effects (P<0.05) of each dependent variable concerning independent variables and their interactions.
As can be observed in Figure 1, the increase of XG confers thickening and stability to YS. It makes ζ more negative and influences net charge of YS particles, favoring repulsive forces between particles (Figueroa et al., 2016). Besides, because of the hydrophilic nature of XG, it confers steric stability (Flatt and Bowen, 2003). Similar cases have been reported in tamarillo drinks (Figueroa et al., 2016), where the inclusion of XG (0.025%) and carboxymethylcellulose, (0.050%) modified ζ to -17.9±1.1 and -30.5±1.8 mV, respectively. The same situation has been reported in orange oil emulsions adding thickeners such as XG, gum Arabic, and gelatin with positive effects on the system colloidal stability (Mirhosseini et al., 2008).
Ascorbic and citric acid showed a quadratic effect on ζ (curvature behavior), being less negative at 0.2% and more negative at 0.1%. These changes modify double layer repulsion forces and electrostatic interactions (ion-particle interactions); as well as ion-ion interaction and YS rheology (Flatt and Bowen, 2003). The pH of the YS varied between 3.8 and 4.9; whereas, the acidity varied between 0.1 and 0.3%. These dependent variables affect ζ because they influence the action of the XG and ionic strength of YS. In orange oil emulsions, it has been reported that the decrease in pH was correlated with the increase in emulsion stability and ζ magnitude (Mirhosseini et al., 2008).
μ showed a tendency to increase when XG addition increases, as well as with TH. Probably because when XG is solubilized undergoes a conformational transition from a double helix to a complex aggregates by hydrogen bonds and polymer intricacy (Jayme et al., 1999, Niu et al., 2017) forming a more complex network of polymers that includes FOS, which translates into an increase in the apparent viscosity (μa) and pseudoplastic behavior of the colloidal system.
This behavior has been observed in coconut-based emulsions (Lucas et al., 2018) and orange beverage emulsions (Mirhosseini et al., 2008). According to Stoke’s law, sedimentation or cremation rate of colloidal systems is inversely proportional to the viscosity of the aqueous phase; therefore, XG contributes to improving YS thermodynamic stability (Mirhosseini et al., 2008). The effect of TH on the μ of YS is marked mainly when the colloidal system contains low XG levels, which could be due to the formation of aggregates or flocs that confer an "elastic" behavior that is typical of suspensions known as physical gels (Castro et al., 2013).
The effect of TH when the concentration of XG was the maximum (0.2%) was less significant in the change of μ, which is corresponding to the reported by some authors, who rather attribute that change to the contribution of insoluble solids of yacon than to homogenization degree. Values of the elastic modulus (G’) between 100-150 Pa, 200-370 Pa, and 550-750 Pa have been reported for concentrations of 70, 80, and 100% of yacon pulp, respectively (Castro et al., 2013). μ presented enough coherence concerning the acid added to low XG levels, decreasing with the increase of acid in YS. H+ ions interaction decreases the ionic strength of YS and the electrostatic repulsive forces between -COO− groups located in the side chain of XG, which leads to the chains’ expansion and a decrease of the μ (Brunchi et al., 2016).
Particle size in colloidal systems is an important parameter associated with stability by phase separation phenomena (Larsson et al., 2012). Generally, particle sizes in YS varied among D10 (125-164μm), D50 (227-292μm), and D90 (332-550μm), which denotes high particle sizes and high polydispersity. The high content of insoluble fibers, which would be interacting with soluble components (inulin-type fructans) and other biopolymers, modify YS rheological properties (Bayod et al., 2008; Sharma et al., 1996). This situation is consistent with the statistical results obtained in D10 and D90 percentiles which did not show significant effects (P>0.05), nor to the interactions of independent variables considered; although there is a tendency to favor smaller sizes with higher TH.
It is observed that an increase of XG has a marked effect on the increase of D50, which is directly associated with higher μ of YS, causing an increase in resistance to homogenization. The lack of homogenization does not favor the reduction in the size of insoluble fibers (Raghavendra et al., 2006). Additionally, the effect of a higher TH is reflected in lowering D50, mainly when less acid is added (0.1%) to YS. A similar behavior (<D50) is experienced by XG-Acid interaction at lower levels of XG and higher acid addition (0.3%). The presence of H+ ions has been commented previously and also its effect on XG action; additionally, these results suggest there is an interaction between acid level and TH that could confer an increase in D50 because of YS elastic behavior, promoting the formation of small particles and aggregates (Castro et al., 2013). In the same way, other authors have reported that homogenization (speed and TH) reduces polydispersity of particles and thereby increases elastic properties (Luckham and Ukeje, 1999).
R is based on the dispersion properties of light, which is related to the average particle size and wavelength (A800/A400), low values denote better stability of the colloidal system (Cortés, 2004). Average values of R varied between 0.446 and 0.607, without reaching significant differences (P>0.05) regarding the evaluated factors, for the same polydispersibility mentioned before. In general, it is highlighted that YS was stable and did not show phase separation after 24 h, which is favored by continuous phase with high μ and the effect of high ζ (Dłużewska et al., 2006). On the other hand, some authors have reported on emulsified drinks with orange oil (Mirhosseini et al., 2008), the interaction of XG and gum arabic had no greater effect on R.
Color parameters of fresh yacon were L*=41.2±1.2, a*=3.1±0.4, and b*=10.5±0.6. In YS the dominant color was between cream and orange, varying their average values of L*, a*, and b* between (38.8 and 43.6), (-0.4 and 2.8), and (8.4 and 16.5), respectively. Yellowish and reddish tones in products are attributable to the content of carotenoids present in yacon roots (Franco et al., 2016); although b* chromaticity did not show significant differences (P>0.05) because of the effects of the evaluated factors, nor for their linear or quadratic interactions. Color parameters L* and a* of YS were the most affected statistically, varying between (38.8 and 46.1) and (-0.4 and 2.4); however, when changes in a* chromaticity are so low, they are not perceptible to the human eye. On the other hand, b* chromaticity was not significant concerning the factors studied; it could be due to some variations in yacon roots’ color because of its composition, ripeness, among others. L* represents the parameter to be controlled in YS, in which an effect of the XG-TH interaction was observed; the greatest clarity (>L*) was reached in combinations of 0.2% - 4 min and 0.1% - 6 min. This result implies an encapsulating action of the biopolymer, protecting bioactive compounds responsible for the yacon’s color; such as betalains, polyphenols, carotenoids, and antoxanthines; that works against oxidative processes and factors of the surrounding environment such as humidity, pH, light, and heat. A similar situation occurs with the TH-Acid interaction, favoring L* in combinations of 0.3% - 4 min and 0.1% - 6 min.
The fit of response surface models and statistical significance of mathematical models was evaluated through an ANOVA, lack of fit test (LOF), and regression coefficients (R2) which are presented in Tables 2 and 3.
Table 3 Regression coefficients, R2 and probability values of lack of fit of the models for dependent variables.
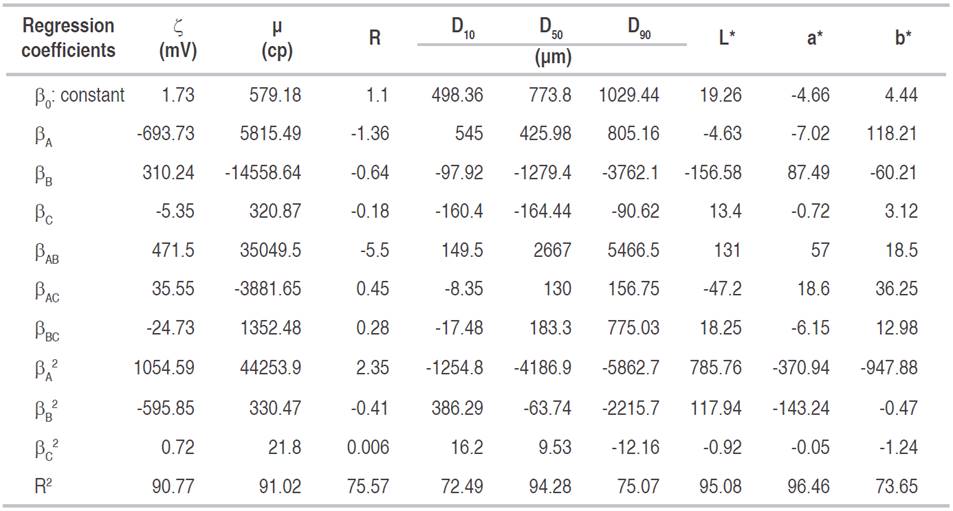
Quadratic regression models with one (P<0.05) and those with the best regression fit (R2>0.9) were ζ, μ, D50, L * and a*, showing good agreement between results predicted by the model and those observed within the experimentation range. R, D10, D90, and b* variables presented values of R2<0.9, indicating that models were not sufficiently predictive, maybe due to several factors not controlled in the research: variation in color of yacon roots, composition, ripeness, and aggregation of particles during YS repose state after homogenization, among others.
The criteria for experimental optimization of YS were set considering minimizing ζ, higher density of negative charge to favor repulsive forces; minimize R and percentiles D10, D50, and D90, characteristic of suspensions with better stability; set μ of suspension in 1000 cP, design criterion of the pilot spray dryer Vibrasec PASLAB 1.5; maximize the color parameters L*, less browning; a* and b*, greater red and yellow pigmentation respectively. These criteria allowed selecting optimal independent variables: XG=0.16% w/w, Acid=0.3% and TH=4.8 min.
Table 4 shows de comparison of experimental values of YS-dependent variables, obtained from three replicates to optimal conditions, with the theoretical values obtained from the mathematical models observing a good approximation between them with a relative mean error values (RME) lower than 10%.
CONCLUSIONS
Yacon is a tuber with health benefits; therefore, due to its industrial potential, it is necessary to generate value for its agricultural chain. The use of statistical tools from an experimental optimization allowed the development of a colloidal suspension based on yacon, stable and adequate from the technical point of view, intended for spray drying processes, which represents an effective alternative of agro-industrialization. Electrostatic forces of the colloidal system favor the stability of YS; XG increases the viscosity of the system, forming a more complex network of polymers that includes soluble and insoluble solids of yacon. Moreover, there are XG-Acid interaction that affects the electrostatic forces of the colloidal system by the H+ ions action that decreases the ionic strength of YS. In addition, the homogenization process affects the rheology of the system, being an important factor in phase separation phenomena. Mathematical modeling of dependent variables presented a good fit with relative mean error values <10%, which guarantees the right prediction of dependent variables.