Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Actualidades Biológicas
Print version ISSN 0304-3584
Actu Biol vol.31 no.90 Medellín Jan./June 2009
ARTÍCULOS DE INVESTIGACIÓN
Histology and morphometry of dorsal root ganglia and their neurons in a fish of indeterminate growth the White Cachama (Piaractus brachypomus)
HISTOLOGÍA Y MORFOMETRÍA DE LOS GANGLIOS DE LA RAÍZ DORSAL Y SUS NEURONAS EN UN PEZ DE CRECIMIENTO INDETERMINADO, CACHAMA BLANCA (PIARACTUS BRACHYPOMUS)
Jenny Jaramillo1, Edwin Gómez-Ramírez1,3, María L. Caldas1, Daniel Rodríguez1, Hernán Hurtado1,2
1 Professors. Histoembriology Laboratory, Ichthyology Group, Applied Biology Program, Faculty of Sciences, Nueva Granada Military University, Campus Rio Grande, Cajicá, Cundinamarca
Correos electrónicos: hernan.hurtado@unimilitar.edu.co; edwin_biologia@yahoo.com.
Abstract
A histological and morphometric analysis of the Dorsal Root Ganglia (DRGs) and their sensorial neurons were carried out to contribute to the understanding of how do their nervous system adapt to their fast body growth. Two age group of fish (age 1 = 20 dph; age 2 = 30 dph), and three spinal sections were used (anterior, middle, and posterior). Histologically, the DRGs and their sensorial neurons are similar to those described for mammals, birds, amphibians, and reptiles. On the other hand, the DRGs and their neurons are larger in age group 2 fish compared with age group 1 fish. There were also differences in the DRGs spinal sections in volume and number of neurons, and the sensorial neurons differed in area. The distribution of the types A and B subpopulations was 36 and 64%, respectively for age group 1 fish; and 25 and 75%, respectively for age group 2 fish. The differences between the DRGs and their sensorial neurons can be attributed to the proportion of target tissue (muscular, cutaneous, and visceral) that each of the DRGs and their neurons must innervate depending on their location. In these species, even a small increment in body mass, represents a change in DRGs and their sensory neurons, suggesting that White Cachama is a good model for the study of the adaptation of the nervous system to large changes in body size.
Key words: dorsal root ganglion, indeterminate growth, morphometry, Piaractus brachypomus, sensory neurons
Resumen
Se realizó un análisis histológico y morfométrico de los Ganglios de la Raíz Dorsal (GRD) y sus neuronas sensoriales, para contribuir al entendimiento de cómo el sistema nervioso se adapta al rápido incremento en el tamaño corporal. Se utilizaron peces en dos clases de edad (edad 1 = 20 dph; edad 2 = 30 dph), y se tomaron GRD de tres secciones de la espina del pez (anterior, medio y posterior). Histológicamente, los GRDs son similares a los de mamíferos, aves, anfibios y reptiles. Los GRDs y sus neuronas son de mayor tamaño en los peces del grupo de edad 2. Se presentaron diferencias en el volumen de los GRDs al igual que en el número y área neuronal en los diferentes niveles espinales. También la ubicación de los GRD influye en el tamaño de los ganglios, y el número y tamaño de las neuronas. Las neuronas tipos A y B representan el 36 y 64%, respectivamente para los peces del grupo de edad 1; y 25 y 75%, respectivamente para los peces del grupo de edad 2. Estas diferencias pueden atribuirse a sus eferentes y a la proporción de tejido blanco (muscular, cutáneo o visceral) que cada GRD y sus neuronas debe inervar dependiendo de su ubicación anatómica. En esta especie, un pequeño incremento en masa corporal representa un cambio importante en los GRD y sus neuronas sensoriales, sugiriendo que la ''cachama blanca'' es un buen modelo para el estudio de la adaptación del sistema nervioso a grandes cambios en tamaño corporal.
Palabras clave: crecimiento indeterminado, ganglio de la raíz dorsal, neuronas sensoriales, morfometría, Piaractus brachypomus
INTRODUCCIÓN
The sensorial neurons of the Dorsal Root Ganglia (DRGs) provide information about mechanical, thermal, chemical, and noxious external and internal stimuli (Friedel et al. 1997). Aspects such as the development of the sensorial systems and DRG formation in fish are little known. Thus increase the interest to study the Peripheral Nervous System (PNS) in fish (Laudel and Lim 1993, Moorman 2001).
The PNS is involved in the adaptation processes of animals since by its means they can gather sufficient external information to correlate the environmental changes and their physiological processes (Carrillo and Rodríguez 2001).
In general, the information that fish receive from the environment depends on the structure and function of its nervous system, specifically from DRGs and their respective sensorial neurons. The study of the DRG cellular bodies shows distinctions that may be particular evidences in the natural history of a species and the environment in which it has evolved (Perl 1992).
In many aspects the DRG neuronal population is heterogeneous, which permits classifying them into subpopulations and eventually, subpopulations according to their morphology, biochemistry and physiology (membrane properties, peripheral and central connections, among others), which makes each subpopulation behave in a different manner when facing internal and external stimuli. The morphological classification is essentially based on the size of the soma in two neurons types; neurons type A (large and light) and type B (small and dark) (Lawson 1992, Martínez et al. 2000), even though Rambourg et al. (1983) could characterize ultrastructurally each neuronal type, which were divided into subtypes (A1, A2, A3 and B1, B2, B3) according to the distribution of the Nissl substance in the cytoplasm, adding as well a third group (neurons type C), the smallest among the DRGs.
The caliber of the sensorial afferences and the propagation velocity of the nervous impulse depend on the neuronal size. The largest neurons in the DRGs have myelinated fibers and the smallest have unmyelinated fibers. This is related as well to the velocity of propagation, which is greater in the myelinated fibers than in the unmyelinated ones (Harper and Lawson 1985, Lawson 1992, Lawson and Waddell 1991). Harper and Lawson (1985) demonstrated that there is a direct relationship between the neuronal size, the type of fiber, the presence or absence of myelin and the velocity of propagation.
For these reasons, the interest in obtaining a model for the histology and morphometry of DRGs and sensorial neurons in P. brachypomus fry arises, allowing the consolidation of this information to increase the knowledge of these structures in teleosts, and thus, understand the relationship between the fish and its environment. Another important consideration is that at present there do not exist enough elements to explain neuronal development in teleosteous of indeterminate growth since the study model in fish is D. rerio, which has a limited and finite growth, reasons for which it would not be the best model if considering that most fish species tend to present a indeterminate growth (Mommsen 2001). In the present study, we tried elucidate that problem describing the normal histology and morphometry of DRGs and their sensory neurons in White Cachama (P. brachypomus) fry of two age groups and in different spinal sections (anterior, middle, and posterior).
MATERIALS AND METHODS
Biological material. Piaractus brachypomus fry of two different sizes (ages group) were used: the age group 1 with 20 days post hatching (dph), a total length of 2,5 ± 0,23 cm and mean weight of 0.16 g. The age group 2 with 30 dph, a total length of 3,71 ± 0,34 cm and weight of 0,69 ± 0,18 g. The P. brachypomus fry used for these studies came from a hatching farm in Villavicencio (Colombia). The fish had been maintained at water temperature range from 25 to 28 ºC. The fish were fed to apparent satiety six times daily with fish pellets at 38% protein. Natural photoperiod was maintained at 12L:12D in a continuous flow.
Tissue processing. The fish were deeply anaesthetized by thermal shock, 10 specimens from each age group were put in a beaker with 250 ml water and placed in freezer until they were immobile, which occurred after about 15 minutes. Then, they were quickly sacrificed by cutting the spinal cord in the posterior part of the head behind the supraoccipital ridge. Incisions were made in the fish's abdominal wall to allow penetration of the fixer (formaldehyde at 4% in 0.1M phosphate buffer, pH 7.2-7.3 range), and stored at 4 °C for at least five days (Hinton 1990, Prophet 1995). After of fixation, the fish were decalcified with a solution of 100ml formic acid at 8% and chlorhydric acid at 8% at a 1:1 proportion at 4 °C during seven days. The solution was changed daily. Immediately, dehydration procedure was performed with ascending ethanol series and following cleared in xylene and embedded in paraffin (MERCK; fusion point 52-54 °C). Serial sections of the whole ganglia were cut with a ratatory microtome (MICROM HM 340E) at 5 µm thickness (Hinton 1990, Prophet 1995). Sections were mounted onto glass slides, and stained with haematoxylin and eosin using standard techniques (Hinton 1990, Prophet 1995).
Histological analysis. The slides were examined with a light microscope (ZEISS®) and the structures such as neurons, fibroblasts and satellite cells were described. Emphasis was made on the distribution, position and staining properties of the DRG sensorial neurons to distinguish the large and light, type A neurons from the small and dark, type B ones.
Morphometric analysis. To carry out this analysis, ten specimens from each age group were taken and a pair of serial DRGs from spinal sections (anterior, middle, and posterior; figure 1) was taken for each specimen.

Figure 1. Spinal sections used for evaluation in 30 dph P. brachypomus fry. Scale in centimeters. Anterior (A) from the supraoccipital crest to the anterior end of the dorsal fin. Middle (M) it corresponds to the section of the dorsal fin. Posterior (P) includes from the anterior end to the middle of the adipose fin. The total length (TL) was taken from the mouth to the posterior part of the caudal fin
Digitalized imaging for each one of the serial sections was done with a light microscope (ZEISS) with an adaptor whose zoom is 2x for the digital cameras (MVC-FD200 SONY and CANNON POWER SHOT). Then, the images were processed to improve contrast, eliminate background noise and thus, create the montage. This procedure was carried out by using Software Scion Image PC (2000), freely distributed on internet (www.scioncorp.com).
DRG volume. In order to define the area of each DRG in each serial section was used the relation VDRG = S(Ai * e), where ''VDRG'' is the volume of the whole ganglion, ''A'' is the ganglion area in a sectional cut and ''e'' is the thickness of the section.
A second method, each one of the images had to go through the Convert software to change its format, and thus, be readable on Align Software. The sections were aligned based on the associated structures such as the spinal chord and the notochord among others. Then, using previously calibrated Trace Software, the regions corresponding to the DRGs were defined, obtaining, as a result, the ganglion total volume.
Number, area, and diameter of DRG neurons. On the Scion Image PC (2000) Software-processed images for each DRG, the area and diameter of each differentiated neuron were counted and measured. The criterion used to count and measure each neuron was: the presence of a nucleus with well defined nucleoli, which should be darker than the surrounding cytoplasm. In sensorial neurons, the nucleus is centrally located. The counts and measures of neurons were taken at the section at which the nucleus and nucleoli presented either highest extension. Each measure was registered under the number corresponding to the neuron (numbered in each image) and the section. This was done to avoid counting the same neuron more than once and for the later data analysis to know if it corresponded to a type A or B neuron.
Statistical analysis. The variables area and diameter of the sensorial neurons and the number and volume of the neurons in each DRG were tested for normality with the Shapiro–Wilk test. Significant differences in the variables between age group (1 and 2) and spinal sections (anterior, middle, and posterior) were determined using one-way ANOVA followed by Tukey's test. A difference of p < 0.05 was considered statistically significant and p < 0.01 difference highly significant. Statistical analysis was performed using the software SAS 8.2 for Windows.
RESULTS
DRG histology. The DRGs in P. brachypomus fry were observed as clusters of encapsulated cells in condensed connective tissue, as described by Ross et al. (1995) in mammals. In addition, the DRGs are continuous to the cover of the peripheral nerves that enter and leave them (Ross et al. 1995).
DRG neuronal structure. Two neuronal subpopulations, one of large and light cells, denominated type A, and the other of small and dark cells, denominated type B, were identified in this study. Type A cells were characterized by their roundly shaped cytoplasm. A denser substance, called Nissl's substance or granular endoplasmic reticulum can be identified. The nucleus is located centrally and a prominent and strongly stained nucleolus was identified (figures 2 and 3). Cells type B are smaller in size, and their nucleus is also recognized by being more strongly stained than the surrounding cytoplasm. Likewise, the nucleolus is differentiated by its more acute staining. Their nucleolus is not as prominent as those of the type A cells. Another type of cells that could be distinguished in the DRGs of P. brachypomus was the satellite cells, which were seen around the sensorial neurons (figures 2 and 3).
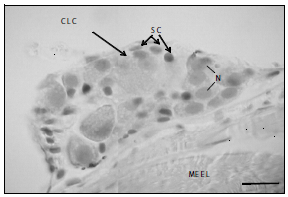
Figure 2. Anterior DRG longitudinal section, P. brachypomus, size 1. Large and light cells (CLC), satellite cells (SC), nuclei (N), connective tissue (CT), longitudinal striated skeletal muscle (MEEL). H & E. 1200X. Bar = 10 µm
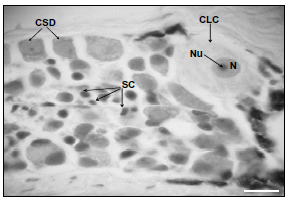
Figure 3. Anterior DRG longitudinal section, P. brachypomus, size 2. Large and light cells (CLC), cells small and dark (CSD), satellite cells (SC), nuclei (N), nucleolus (Nu). H & E. 1200X. Bar = 10 µm
Morphometry. DRG Volume. The DRGs in the group age 2 fish have a considerable larger volume than those in fish age group 1 (P < 0.01). There are no significant differences in DRGs in fish age group 1 at the analyzed spinal sections. On the contrary, the DRG volume in fish age group 2 differ at each spinal sections, with those at the anterior level being the largest, followed by those at the posterior level and, finally, those in the middle (figure 4).
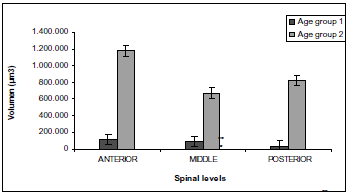
Figure 4. Comparison of P. brachypomus DRG volume at different spinal sections. Each value represents mean ± the standard error. The volume of age group 2 DRG is larger than age group 1 DRG *(P < 0.01). In age group 1 DRG no differences were observed between spinal sections. However in age group 2, DRG volumes were different between spinal sections, being larger anterior section, followed by posterior section, and finally, middle spinal sections **(P < 0.05)
Number of neurons. A total of 5.012 sensorial neurons that exhibit the mentioned criteria were counted for this study, corresponding to ten fish per age group. Type B neurons were the largest number with a total of 64% for fish age group 1 and 75% for age group 2, while neurons type A were found at a lower frequency with a total of 36% for size 1 and 25% for size 2 (table 1).
Table 1. Number and percentage of type A and B differentiated neurons for each age group in P. brachypomus fry [number of sensorial neuron differentiated types A and B in the GRDs by age group and spinal levels. There are not differences between the number of type A neuron the age group 1 and the age group 2. Different small letters within age groups indicates statistical differences between different types of neurons (P < 0.05)]

For both age groups, the number of type A neurons differs according to the spinal sections, being similar at the anterior and posterior sections, and differing from the middle one, which shows a lower number of neurons. The number of type B neurons is similar at all spinal sections (anterior, middle, and posterior) in age group 2. In age group 1, at the middle level, the number of neurons is higher than at the anterior and posterior sections, which are similar in number (P < 0.05) (figure 5).
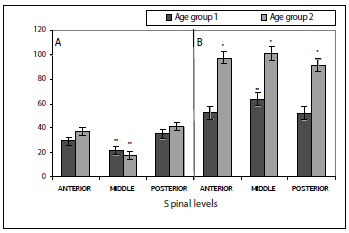
Figure 5. Number of differentiated type A (A) and type B (B) neurons on DRG of P. brachypomus fry. Each value represents mean ± the standard error. Number of type A neurons shows no differences between age groups 1 and 2 (P < 0.05). However, the number of type B neurons is different between age groups *(P < 0,01). Number of type A neurons is smaller in middle spinal section for both age groups **(P < 0.05). The number of type B neurons is similar for all spinal sections in size 2 individuals, but in age group 1 individuals, middle section presents more B neurons **(P < 0.05)
Neuron area. The areas of neurons type A and those of type B are different, with those of type A exhibiting a larger one than those of type B. The area of the differentiated type A and B neurons in the DRGs was evaluated for age group and spinal sections. Type A neuron area showed highly significant differences between fish age group 1 and age group 2 (P < 0,0001). Likewise, type B neuron area also show highly significant differences between the two sizes (P < 0,0001), the fish of age group 2 showing the largest area for neurons type A and type B (figures 5, 6). The mean of area for neurons type A in size 1 differs at each spinal level, with the posterior site showing the highest area mean, followed by the anterior level and, finally, the middle level (figure 6).
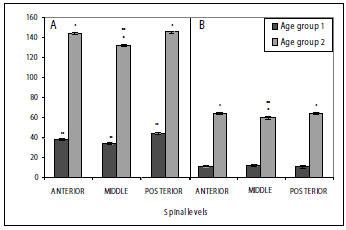
Figure 6. Area of type A (A) and type B (B) neurons on DRG of P. brachypomus fry. Each value represents mean ± the standard error. Area from type A and B neurons is different between age groups *(P < 0.01). Mean area of type A neurons is different between sections spinal in age group 1 individuals, with posterior section neurons being larger, followed by anterior section neurons, and finally, middle section neurons **(P < 0.05). In age group 2 individuals, middle section A neurons are smaller **(P < 0.05). Mean area of type B neurons is similar for all spinal sections in size 1 individuals, while in age group 2 individuals middle section neurons are smaller **(P < 0.05)
In the age group 2, the results were different, since the anterior and posterior sites have a similar mean area of neurons type A, while the middle level is the one showing the lowest value of mean area when compared to the ones mentioned (figures 5, 6). On the other hand, the mean area of type B neurons in age group 1 showed similar results in all the evaluated spinal sections. As seen in the type A for age group 2, the value for the mean area was similar at the anterior and posterior spinal sections, again with the middle level having the lowest area values (figures 5, 6).
DISCUSSION
Histology. Dorsal root ganglions. The neuronal types found in DRGs of P. brachypomus fingerlings are similar to what is described for mammals (Coggeshall et al. 1994, Ross et al. 1995), birds (Eide and Glover 1997), amphibians (Peter et al. 1994), and reptiles (Geuna et al. 1998). Neurons in DRGs are characterized by their round shape (Lawson et al. 1992, Ross et al. 1995); however, the sensorial neurons in this study are not completely round, and sometimes, presented elongated shapes (figures 2 and 3), which is characteristic of immature neurons (Berg and Farel 2000).
The growth of the neurons depending on the fish age could be explained by the growth and differentiation of the sensorial neurons in relation to the growth and development of the individual (Heathcote and Sargent 1987). The DRG cells at the middle level were predominantly smaller and darker than those at the anterior and posterior spinal sections, difference that could be attributed to the different maturing time among the various DRGs (An et al. 2002).
With the histological observations we found a low number of satellite cells in the DRGs of P. brachypomus fry may be explained, as previously mentioned, by immaturity of the DRGs: a large number of cells were found to be undifferentiated and it si possible that many of these would be satellite cells which had not differentiated yet (Coggeshall et al. 1994, Ross et al. 1995)
Morphometry. The highly significant increase in volume of the DRGs in P. brachypomus fry observed between the two age groups might be due to the increased body size of the fish. Being a rapid-growing species it would be expected that the DRG volume increase in order to sensorial innervate the growing muscle and skin. Pearson et al. (1978) mention that in rats, it is normal that the DRGs increase their sizes, as they get older. This growth in volume is also produced by the increase in the number of neurons. On the other hand, the differences in volume at the different spinal sections in fish of age group 2 could be explained by the anatomy of the fish and its corresponding sensorial innervations. The DRGs at the anterior level showed the largest volume; likewise, this level exhibits a larger amount of muscular tissue, and there is a larger area of exposed skin. This is very important since the main targets of the DRGs are the cutaneous and muscle terminals (Eide and Glover 1997, Sharma and Frank 1998). It was found that the DRGs at the posterior level followed the ones at the anterior one as far as volume is concerned. The posterior level of a P. brachypomus fry possesses neither the largest amount of muscle nor exposed skin, but it is the tail the main instrument for movement and displacement of the fish. In the specific case of the P. brachypomus fry, these are more active than at their adult stage, so it is necessary to have superior sensorial feed back for controlling the tail and for their displacement, either to find food or to escape from predators. These conditions are more determining in fry than in adults, since adult fish have advantages in relation to the fry in the rate of predation or the search for food (Helfman et al. 1997). Contrary to what was expected, the DRGs at the middle level presented a lower volume, since it was supposed that this area had the largest area of exposed skin and the most amount of muscle. However, upon studying the internal anatomy, it was found that it did not possess as much muscle tissue as expected. In addition, most vital organs are placed in this region, and since the innervations to these tissues is mainly autonomous, this might explain the lower DRGs volume at the middle level, although DRGs play a role in proprioception (Caspary and Anderson 2003, Friedel et al. 1997). These same arguments could explain the variations in the number of neurons depending on the spinal level. This hypothesis may be supported by An et al.'s study (2002), which found a different number of sensorial neurons between the anterior and posterior sites in zebra fish (Danio rerio). Similarly, in adult rats, differences in the number of neurons between the DRGs in the cervical and lumbar sites have been found; for example, Mille-Hamard et al. (1999), who studied the C4, C5, and C6 cervical DRGs in adult rats, found that they contained 7.508 ± 299, 6.825 ± 950, and 6.858 ± 923 sensorial neurons, respectively. In contrast, Schmalbruch (1987), who evaluated the L4, L5, and L6 lumbar site DRGs, found that each of these contained approximately 12.000, 15.000, and 14.000 sensorial neurons, respectively.
Previous studies on mice and rats show that type B neurons are more numerous than those of type A (Altman and Bayer 1984, Kitao et al. 1996, Lawson 1992). Szarijanni and Rethelyi (1979) determined in DRGs at the sacrococcygeal part in cats that 69% of the population corresponded to small, dark neurons. Tandrup (1993), in a study carried out on the L5 site in male rats of the Wistar strain, found a total mean number of 17.900 neurons, of which 28% were type A and 70% were type B. These percentages are similar to those obtained in this study and which would indicate that the subpopulations in P. brachypomus fry have a similar distribution in comparison with mammals.
Two hypotheses have been proposed to explain the increase in the number of neurons in the DRGs at later stages or when the individual increases its muscular mass. One of them exposes that there is no neurogenesis (Berg and Farel 2000, Fernández and Nicholls 1998, La Forte et al. 1991, Mohammed and Santer 2001, Pover et al. 1994). Instead, a later differentiation of precursor cells occurs (Peter et al. 1994, Popken and Farel 1997). The other hypothesis poses a continuous neurogenesis in organisms with indeterminate growth (as is P. brachypomus), inclusive in adult individuals (Devor and Govrin-Lippmann 1991). The increase in the number of type B neurons in fish of age group 2 could be explained by a late differentiation of precursor cells that were not considered in fish of age group 1 because they did not meet the established criteria for counting due to their immaturity. However, we can not discard that this increase is due to a process of neurogenesis because we do not perform these tests in this investigation. An et al. (2002) detected by immunehistochemical tests in Zebra fish (Danio rerio with determined growth) neurogenesis in individuals up to 28 days post fertilization, which could be another reason not to reject neurogenesis as the explanation for the increase in the number of type B neurons in fish of age group 2, in view of the fact that P. brachypomus is a fish of indeterminate growth.
Type A cells did not show differences in the mean number between the two age groups, and their number was lower than the type B cells, which could be due to the fact that the precursors of type A neurons cease their proliferation before those of the type B cells (Ninomiya et al. 1994, in: Martínez et al. 2000). Previous studies in rat embryos on the development of the subpopulations of sensorial neurons found that these develop oscillatory, that is, the large light (type A) ones develop first, and then, the small dark (type B) ones, which are found in larger numbers, do (Altman and Bayer 1984, Kitao et al. 1996).
Furthermore, on rat embryos has been reported that it is common that the two subpopulations overlap at immature stages (Jackman and Fitzgerald 2000). The area differences would indicate that neurons belonging to one same subpopulation do not mature at the same time. Therefore, in the same DRG, inclusive in the same sections spinal, type A or B neurons differing in area value could be found. These differences could be attributed to trophic factors that are provided by the tissue target (Jackman and Fitzgerald 2000). Also, factors such as neuronal birth, the time and pattern of subsequent development, as well as the neurotrophic factors and the innervations patterns are critical in determining the area of the sensorial neurons at a specific moment (Goldstein et al. 1996, Kishi et al. 2002, Martínez et al. 2000, Rohrer et al. 1988, Zochodne et al. 2001).
REFERENCES
1. Altman JS, Bayer A. 1984. The development of the rat spinal cord. Advances in Anatomy Embryology and Cell Biology, 85: 1-166. [ Links ]
2. An M, Rushu L, Henion PD. 2002. Differentiation and maduration of Zebra fish dorsal root and sympathetic ganglion neurons. Journal of Comparative Neurology, 446: 267-275. [ Links ]
3. Berg JS, Farel PB. 2000. Developmental regulation of sensory neuron number and limb innervation in the mouse. Developmental Brain Research, 125: 21-30. [ Links ]
4. Carrillo M, Rodríguez JA. 2001. Bases fisiológicas de la reproducción de peces tropicales. In: Rodríguez H, Daza P, Carrillo M editors. Fundamentos de acuicultura continental. Bogotá (Colombia): Instituto Nacional de Pesca INPA. p. 205-206. [ Links ]
5. Caspary T, Anderson K. 2003. Pattering cell types in the dorsal spinal cord: what the mouse mutants say. Nature Reviews, 4: 289-297. [ Links ]
6. Coggeshall RE, Pover CM, Fitzgerald M. 1994. Dorsal root ganglion cell death and surviving cell numbers in relation to the development of sensory innervation in the rat hindlimb. Developmental Brain Research, 82: 193-212. [ Links ]
7. Devor M, Govrin-Lippmann R. 1991. Neurogenesis in adult rat dorsal root ganglia: on counting and the count. Somatosensory and Motor Research, 8: 9-12. [ Links ]
8. Eide AL, Glover JC. 1997. Developmental dynamics of functionally specific primary sensory afferent projections in the chicken embryo. Anatomy and Embryology. 195: 237-250. [ Links ]
9. Fernández J, Nicholls JG. 1998. Fine structure and development of dorsal root ganglion neurons and Schwann cells in the newborn opossum Monodelphis domestica. Journal of Comparative Neurology, 396: 338-350. [ Links ]
10. Friedel RH, Schnürch H, Stubbusch J, Barde Y. 1997. Identification of genes differentially expressed by nerve growth factor- and neurotrophin-3-dependent sensory neurons. Proceedings of the National Academy of Sciences, 94: 12670-12675. [ Links ]
11. Geuna S, Borrione P, Corvetti G, Poncino A, Giacobini-Robecchi MG. 1998. Types and sub-types of neurons in dorsal root ganglia of the lizard Podarcis sicula: a light and electron microscope study. European Journal of Morphology. 36: 37-47. [ Links ]
12. Goldstein ME, Grant P, House SB, Henken DB, Gainer H. 1996. Developmental regulation of two distinct neuronal phenotypes in rat dorsal root ganglia, Neuroscience, 71: 243-258. [ Links ]
13. Harper AA, Lawson SN. 1985. Conduction velocity is related to morphological cell type in rat dorsal root ganglion neurons. Journal of Physiology, 359: 31-46. [ Links ]
14. Heathcote RD, Sargent PB. 1987. Growth and morphogenesis of an autonomic ganglion. I. Matching neurons with target. Journal of Neurosciences, 7: 2493-2501. [ Links ]
15. Helfman GS, Collette BB, Facey DE. 1997. The diversity of fishes. Massachusetts (USA): Blackwell Science Inc, Malden. [ Links ]
16. Hinton DE. 1990. Histological techniques. In: Schreck CB, Moyle PB editors. Methods for fish biology. Bethesda: Exxon Company. p. 191-211. [ Links ]
17. Jackman A, Fitzgerald M. 2000. Development of peripheral hindlimb and central spinal cord innervation by subpoblations of dorsal root ganglion cells in the embryonic rat. Journal of Comparative Neurology, 418: 281-298. [ Links ]
18. Kishi M, Tanabe J, Schmelzer JD, Low PA. 2002. Morphometry of dorsal root ganglion in chronic experimental diabetic neuropathy. Diabetes, 51: 819-824. [ Links ]
19. Kitao Y, Robertson B, Kudo M, Grant G. 1996. Neurogenesis of subpopulation of rat lumbar dorsal root ganglion neurons including neurons projecting to dorsal column nuclei. Journal of Comparative Neurology, 371: 249-257. [ Links ]
20. La Forte RA, Melville S, Chung K, Coggeshall RE. 1991. Absence of neurogenesis of adult rat dorsal root ganglion cells. Somatosensory and Motor Research, 8: 3-7. [ Links ]
21. Laudel TP, Lim TM. 1993. Development of the dorsal root ganglion in a teleost, Oreochro mossambicus (Peters). Journal of Comparative Neurology, 327: 141-150. [ Links ]
22. Lawson SN, Waddell PJ. 1991. Soma neurofilament immunoreactivity is related to cell size and fibre conduction velocity in rat primary sensory neurons. Journal of Physiology, 435: 41-63. [ Links ]
23. Lawson SN. 1992. Morphological and biochemical cell types of sensory neurons. In: Scott SA, editor. Sensory Neurons: Diversity, development, and plasticity. New York (USA): Oxford University Press. p. 27-52. [ Links ]
24. Martínez M, Quiroga NY, Castellanos JE, Hurtado H. 2000. Subpoblaciones neurales presentes en el ganglio de la raíz dorsal. Biomédica, 20: 248-260. [ Links ]
25. Mille-Hamard L, Bauchet L, Baillet-Derbin C, Horvat JC. 1999. Estimation of the number and size of female adult rat C4, C5 and C6 dorsal root ganglia (DRG) neurons. Somatosensory and Motor Research, 16: 223-228. [ Links ]
26. Mohammed HA, Santer RM. 2001. Total neuronal numbers of rat lumbosacral primary afferent neurons do not change with age. Neuroscience, 105: 249-263. [ Links ]
27. Mommsen TP. 2001. Paradigms of the growth in fish. Comparative Biochemistry and Physiology, 129: 207-219. [ Links ]
28. Moorman SJ. 2001. Development of sensory systems in Zebrafish (Danio rerio), ILAR Journal. 4: 292-298. [ Links ]
29. Pearson J, Pytel BA, Grover-Johnson N, Axelrod F, Dancis J. 1978. Quantitative studies of dorsal root ganglia and neuropathologic observations on spinal cords in familial dysautonomia. Journal of the Neurological Sciences, 35: 77-92. [ Links ]
30. Perl ER. 1992. Function of dorsal root ganglion neurons: An overview. In: Scott SA, editor. Sensory Neurons: Diversity, development, and plasticity. New York (USA): Oxford University Press. p. 3-20. [ Links ]
31. Peter GR, Wecker ST, Farel PB. 1994. Hindlimb sensory neuron number increases with body size (Rana catesbeiana). Journal of Comparative Neurology, 357: 117-123. [ Links ]
32. Popken GJ, Farel PB. 1997. Sensory neuron number in neonatal and adult rats estimated by means of stereologic and profile-based methods. Journal of Comparative Neurology, 386: 8-15. [ Links ]
33. Pover CM, Barnes MC, Coggeshall RE. 1994. Do primary afferent cell numbers change in relation to increasing weight and surface area in adult rats? Somatosensory and Motor Research, 11: 163-167. [ Links ]
34. Prophet EB, Mills B, Arrington JB, Sobin LH. 1995. Métodos histotecnológicos. Washington: Registro de Patología de los Estados Unidos de América. [ Links ]
35. Rambourg A, Clermont Y, Beaudet A. 1983. Ultraestructural features of six types of neurons in rat dorsal root ganglia. Journal of Neurocytology, 12: 47-66. [ Links ]
36. Rohrer H, Hofer M, Hellweg R, Korsching S, Stehle AD, Saadat S, Thoenen H. 1988. Antibodies against mouse nerve growth factor interfere in vivo with the development of avian sensory and sympathetic neurons. Development, 103: 545-552. [ Links ]
37. Ross MH, Romrell LJ, Kaye GI. 1995. Histology, A text and atlas. Third edition. Baltimore (USA): Williams and Wilkins Publ. [ Links ]
38. Schmalbruch H. 1987. The number of neurons in dorsal root ganglia L4-L6 of the rat. Anatomical Record, 219: 315-322. [ Links ]
39. Sharma K, Frank E. 1998. Sensory axons are guided by local cues in the developing dorsal spinal cord. Development, 125: 635-643. [ Links ]
40. Szarijanni N, Rethelyi M. 1979. Differential distribution of small and large neurons in the sacrococcygeal dorsal root ganglia of the cat. Acta Morphologica Academiae Scientiarum Hungaricae, 27: 25-35. [ Links ]
41. Tandrup T. 1993. A method for unbiased and efficient estimation of number and mean volume of specified neuron subtypes in rat dorsal root ganglion. Journal of Comparative Neurology, 329: 269-276. [ Links ]
42. Zochodne DW, Verge VM, Cheng C, Sun H, Johnston J. 2001. Does diabetes target ganglion neurones? Progressive sensory neurone involvement in long-term experimental diabetes. Brain, 124: 2319-2334. [ Links ]
Recibido:enero 2009
Aceptado: junio 2009














