Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Actualidades Biológicas
Print version ISSN 0304-3584
Actu Biol vol.36 no.101 Medellín July/Dec. 2014
RESEARCH PAPERS
First reports and generic descriptions of the achlorophyllous holoparasites Apodanthaceae (Cucurbitales) of Colombia
Primeros registros y descripciones genéricas de la familia de holoparásitos aclorófilos Apodanthaceae (Cucurbitales) de Colombia
Favio González1, Natalia Pabón-Mora2
1 Instituto de Ciencias Naturales, Universidad Nacional de Colombia. A. A. 7495. Bogotá, Colombia. fagonzalezg@unal.edu.co
2 Docente e investigadora, Instituto de Biología, Universidad de Antioquia. A. A. 1226. Medellín (Antioquia), Colombia. lucia.pabon@udea.edu.co
Received: February 2014; accepted: May 2014.
Abstract
The Apodanthaceae (Cucurbitales) are one of the 12 parasitic clades of flowering plants and the only holoparasitic family with a vegetative phase entirely endophytic, growing primarily on Fabaceae and Salicaceae stems. The two genera of the family, Apodanthes and Pilostyles, are broadly distributed in Colombia, although they remain extremely under-collected and poorly known. Based on field and herbarium work, and a review of the literature, we provide detailed generic-level descriptions of the family. We also update the records of the family in Colombia and discuss the homology and taxonomic implications of floral characters such as perianth scales, pollen sacs, and the central column. Finally, we provide photographic records of Colombian Apodanthaceae, and some guidelines for properly collecting these plants.
Key words: Apodanthaceae, Apodanthes, Cucurbitales, Colombian flora, parasitic flowering plants, Pilostyles.
Resumen
Apodanthaceae (Cucurbitales) es uno de los doce clados de angiospermas parasíticas, y la única familia de especies holoparasíticas con la fase vegetativa completamente endofítica en tallos principalmente de especies de Fabaceae y Salicaceae. Los dos géneros de la familia, Apodanthes y Pilostyles están ampliamente distribuidos en Colombia, aunque han sido muy poco recolectados y permanecen poco conocidos. Con base en trabajo de campo y de herbario, y una extensa revisión bibliográfica, se describen en detalle los caracteres genéricos de la familia. A la vez, se actualizan los registros de la familia en Colombia y se discute la homología e implicaciones taxonómicas de caracteres florales tales como piezas del perianto, sacos polínicos y columna central. Finalmente, presentamos fotografías de las Apodanthaceae colombianas, así como algunas recomendaciones para recolectar adecuadamente estas plantas.
Palabras clave: Apodanthaceae, Apodanthes, Cucurbitales, flora colombiana, plantas con flor parasíticas, Pilostyles.
INTRODUCTION
The family Apodanthaceae Takhtajan consists of 11 species in the genera Apodanthes Poit. (1 species from the New World) and Pilostyles Guill. (10 spp. from North-, Central- and South America, Iran, Irak, Syria, Asia Minor, subtropical eastern Africa and southwestern Australia) (Bellot and Renner, 2014). All members of the family are achlorophyllous holoparasites with an intrincate but histologically simple endophyte and an exophyte consisting of small gregarious flowers that emerge from the parasitized stems of a limited group of hosts (table 1). The wide distribution of the family on four continents strongly suggest a long evolutionary history (Filipowics and Renner, 2010) and make intercontinental dispersal a less plausible biogeographic scenario.
The affinities of Apodanthaceae have long been debated. They have been placed as part of Rafflesiales/Rafflesiaceae primarily based on the parasitic life style, the dioecy and the central column of the flower (e.g., Brown, 1845; Endlicher, 1841; Eichler, 1878; Eliasson, 1994; Engler, 1912; Gómez, 1983; Harms, 1935; Hutchinson, 1959; Karsten, 1856; Kuijt, 1969; Meijer, 1993; Melchior, 1964; Mitchell, 2004; Takhtajan, 1997; Yatskievych and Meijer, 2004) or related to Malvales (Blarer et al. 2004; Heide-Jørgensen, 2008) based on floral development and morphology. However, recent molecular-based phylogenetic analyses have shown that Rafflesiaceae s.l. is polyphyletic, and placed Apodanthaceae as sister to the remaining seven families of the order Cucurbitales (Filipowicz and Renner, 2010; Nickrent, 2008; Nickrent et al. 2004; Schaeffer and Renner, 2011). According to Filipowicz and Renner (2010), inferior ovaries, parietal placentation and dioecy support the inclusion of the Apodanthaceae within Cucurbitales. Thus, Apodanthaceae and the redefined Rafflesiaceae s. str. (now part of Malpighiales) represent two of the 12 independent origins of parasitism among flowering plants (Barkman et al. 2007; Nickrent, 2008).
At a specific level, the equivocal morphological characters in Apodanthaceae have led some authors (e.g., Harms, 1935; Solms-Laubach, 1878) to use extrinsic traits to distinguish species, particularly the hosts on which they grow, or the geographical area they occupy. This is evident in names such as Apodanthes caseariae Poit., Pilostyles aethiopica Welw., P. mexicana (Brandegee) Rose, or P. ingae or P. calliandrae, the latter two lacking any differential trait in Solms-Laubach's (1878) treatment besides the hosts they parasitize.
Although members of Apodanthaceae from the United States, Mexico, Venezuela, Brazil and Chile have been collected and are thus relatively well documented, they remain poorly known and undercollected in Colombia. This is mainly because of their small flowers, the endophytic life form, the absence of conspicuous hypertrophy of the infected tissues in the host, and the short flowering time. Thus, the goals of this work are: (1) to report new findings of Apodanthaceae in Colombia, (2) to update and revisit historical data regarding the presence of the family in Colombia, (3) to contribute to the ongoing discussion of the homology of the perianth, the central column, the androecium, and the fruit and (4) to provide guidelines to help Colombian botanists collect and make careful field observations of this extremely undercollected family. At present, limited collections prevent detailed developmental, morphological, ecological and taxonomic studies of these interesting holoparasitic flowering plants.
MATERIALS AND METHODS
We examined all the Colombian specimens (ca. 2500) of the genera expected or reported as hosts of Apodanthaceae, e.g., Adesmia, Bauhinia, Calliandra, Cassia, Dalea, Dioclea, Galactia, Inga, Mimosa, and Schnella (Fabaceae), and Banara, Casearia and Xylosma (Salicaceae) housed at Herbario Nacional Colombiano (COL; Universidad Nacional de Colombia, Bogotá, Colombia), Herbario Universidad de Antioquia (HUA; Universidad de Antioquia, Medellín, Colombia), New York Botanical Garden (NY; New York, U. S. A.), and Herbario Toli (TOLI; Universidad del Tolima, Ibagué, Colombia). The historical collections of Apodanthaceae cited below and deposited at Gray Herbarium (GH; Harvard University, Massachusetts, U. S. A.) and Royal Botanic Gardens, Kew (K; London, U. K.) were also examined for the present study. Based on this herbarium search, fieldwork was carried out in selected localities where Apodanthaceae and their hosts are expected to occur in Boyacá (Colombia).
The descriptions that follow are based on our field and herbarium observations, as well as relevant literature, especially Bellot and Renner (2013, 2014), Blarer et al. (2004), Blassingame (1968), Dell et al. (1982), Endriss (1902), Felger (1988), Guillemin (1834), Harms (1935), Heide-Jørgensen (2008), Kuijt (1969), Kuijt et al. (1985), Nickrent (2006), Robinson (1891), Rutherford (1970), Solms-Laubach (1874), Takhtajan (1997), Thiele et al. (2008), and Vattimo-Gil (1955, 1956, 1978). The taxonomy of Dalea follows Barneby (1977).
RESULTS AND DISCUSSION
Morphological traits. Apodanthaceae are perennial, achlorophyllous holoparasites (figures 1, 2, 3, 4) and their vegetative tissues are reduced to a mycelium-like system that is completely endophytic, that is, it develops entirely within the host. Both monoecy and dioecy have been reported to occur in the family. In Pilostyles aethiopica Welw. (formerly Berlinianche aethiopica) from Zimbabwe and P. haussknechtii Boiss. from Iran, Bellot and Renner (2013) reported a higher number of host individuals carrying staminate flowers. They also found that the presence of flowers of both sexes on the same host is occassional or nule. This issue is further complicated by the fact that it is not easy to determine an individual in Apodanthaceae (see below). Nevertheless, we have detected the presence of staminate and carpellate flowers in the same floral row in P. boyacensis, which indicates monoecy at least in this species.
The flowers of Apodanthaceae are gregarious, sessile (figures 1, 2, 3, 4), unisexual, globose, ovoid or ellipsoid in side view, small (< 6 mm in length and 8 mm in diameter, some of the smallest among the holoparasitic plants; Kuijt, 1969), bright yellow, white, pink, red or purple colored, with a tannoid or ''indoloid'' odor (pers. obs.; Blassingame, 1968; Sipes et al. 2014). Prior to the emergence from the host tissue, floral buds are apparent as they form wart-like outgrowths (called ''floral cushions'' by Solms-Laubach, 1874; figure 1E). Flower emergence occurs from endogenous primordia by rupturing the outer layer of the host stem (figures 1E-G, 3) such that several to many flowers can break through (Kuijt, 1969: figure 1, 3O), thus forming floral rows that presumably belong to a single individual (figure 1E, F). The perianth is formed by 12 to 15 opposite to alternate organs, in 3 or 4 poorly defined whorls; aestivation of these organs in the floral bud is irregular, although cochleate appear to be the most frequent type in both the staminate and the carpellate flowers (figures 1F, 3C, D). The outer perianth whorl is formed by two or three scale-like free organs more or less integrated to the rest of the flower (figures 1, 3 B, E, 4D), whereas the middle and inner whorls (calyx and corolla sensu Guillemin; figure 3) are tri- to hexamerous. At least in Pilostyles, the outer and middle perianth organs are purple and the inner ones are yellow, pink, red, brown, purple or, less often, white (figure 1 D, G, H). The inner perianth organs in P. aethiopica have a hair carpet at the base of its adaxial side. A tubular column basally surrounded by a nectary disk is formed at the center of both staminate and carpellate flowers; stomata are present in the outer epidermis of the nectary disk.
Staminate flowers. The androecium is a synandrium composed of two to four rings of globose, sessile pollen sacs located laterally to the central column (figure 3). These dehisce through a transversal slit between adjacent rings. The number of pollen sacs per ring varies from 15 to 30, and usually decreases centripetally. The pollen sacs lack endothecia. Pollen grains are tricolpate in Apodanthes and Pilostyles, except in P. aethiopica, which has non-aperturate pollen. Above the rings of pollen sacs, a rooflike ring of vesicular papillae is formed. These papillae were interpreted as rudiments of the stigma by Solms-Laubach (1874). Gynoecial rudiments in the form of a style with a stylar canal and a mound-like, massive stigmatic surface are present in the staminate flowers. Vestiges of locules or placentae have not been found, although ovule-like rudimentary tissue is occasionally formed in the stylar canal.
Carpellate flowers. Rudiments of androecium have not been found. The gynoecium is four-carpellate with a unilocular ovary that is quadrangular or circular in transverse section. There are four (sometimes more due to proliferating placentae) discrete parietal placentae or a broad and diffuse placenta with the ovules covering the whole cavity may be present. The position of the ovary appears to be inferior or half-inferior; however, this assesment implies that the outer scales, clearly located below the ovary, are not part of the flower (figures 3, 4). The central column is differentiated into a short, massive and vascularized style with a stylar canal bearing transmitting tissue, and a stigma formed by a series of large vesicular papillae located just below the apex of the column. The ovules are bitegmic, tenuinucellate, anatropous to halfway anatropous; the embryo sac is Polygonum type. Fruits have been described either as berries or as fleshy capsules; they are small (<1 cm in diameter), ovoid-turbinate, conical to globose, orange, yellow or red, and are either exposed (figure 4D) or enclosed by the persistent perianth organs (figure 1G, I), they contain up to 100 or more minute (< 0.5 mm long) ovoid to pyriform seeds, which are sticky and hyaline. Both the embryo and the endosperm are reduced.
Floral and fruit traits of uncertain homology. The origin and homology of several floral organs in Apodanthaceae have long been in dispute, and developmental studies are urgently needed for a better understanding of the perianth organs, the central column, the androecium, and the fruit type.
Perianth organs. The homology of the outer, middle and inner perianth parts has been controversial, although a detailed comparative study by Blarer et al. (2004) suggests that the outer whorl does not correspond to the perianth. This is consistent with the lack of integration of the outer scale-like bracts with the rest of the flower, especially in Apodanthes (figure 4D), as a short internode separates the scale-bracts and the middle perianth organs, rendering the ovary as inferior or halfway inferior.
Central column. The central column in the flowers of Apodanthaceae (figure 3 E, G) has long been interpreted as a gynostemium despite the fact that flowers are functionally unisexual. In fact, Guillemin (1834) placed his new genus Pilostyles as a member of the class Gynandria Polyandria. However, the vestigial gynoecium in the staminate flowers and the absence of vestigial androecium in the carpellate flowers complicates the interpretation of the central column as the result of the fusion between carpellary and androecial tissue.
A close relationship between Apodanthaceae (as members of Rafflesiaceae) and Aristolochiaceae (cf. Huber, 1960; Solms-Laubach, 1891) was based on the superficial resemblance of the ring-shaped stigmatic surface in these families. A detailed comparison shows, however, that the central column of the former is mainly carpellary tissue, either vestigial or functional (cf. Blarer et al. 2004; Igersheim and Endress, 1998), whereas the gynostemium of the latter is formed by the fusion of 5, 6 or more stamens and stigmatic lobes, both functional and extremely specialized for pollination (González and Stevenson, 2000).
Androecium. The structure of the androecium in Apodanthaceae is unique because the individual, monosporangiate pollen sacs are sessile and do not conform the typical dithecal, tetrasporangiate anthers (pers. obs.; Blarer et al. 2004; Endress and Stumpf, 1990; Guillemin, 1834; Kuijt, 1969; figure 3). In addition, the presence of numerous, contiguous pollen sacs per ring, the transverse dehiscence between two contiguous pollen sacs, and the lack of endothecium do not support the interpretation of each pollen sac as a monosporangiate anther.
Fruits. Fruits in Apodanthaceae have been described as either capsules or berries (cf. Harms, 1935; Karsten, 1856; Solms-Laubach, 1878). However, the histological changes after fertilization and during fruit formation are still unknown. Thus, a developmental/ anatomical study to examine the histology of the pericarp and the mechanism(s) of fruit dehiscence, if any, is underway to unequivocally answer this question. It will also be useful to better understand the plesiomorphic state in fruit evolution in Cucurbitales, as within each of the remaining families of the order (Anisophyllaceae, Begoniaceae, Coriariaceae, Cucurbitaceae, Datiscaceae, and Tetramelaceae) both fleshy (drupes, pepos or berries) and dry (capsules or samaras) fruits occur.
Systematic and taxonomic considerations. The molecularbased placement of Apodanthaceae in Cucurbitales appears to be well supported. Filipowicz and Renner (2010) stated that the inferior ovary, the parietal placentation, and the dioecy are the morphological synapomorphies for the order. However, these traits need both developmental studies and extensive fieldwork (currently in progress by the authors), as they are still unclear in the family. For instance, the occurrence of dioecy or monoecy in the species of Apodanthaceae is not easy to discern, because of the persistence of many fruits from previous years in a single host individual, the formation of numerous seeds per fruit, and the uncertainty to assert what constitutes an individual in Apodanthaceae (figure 1A, B). According to Bellot and Renner (2013:1091), the endoparasitic lifestyle in the family ''makes it difficult to decide whether cases of monoecy in fact represent multiple infections with two or more parasite individuals, each representing one sex''.
The systematics and taxonomy within Apodanthaceae is challenging, mainly due to the extreme reduction of the individuals as obligate parasites, the endoparasitic lifestyle, and the lack of clear-cut characters to distinguish genera and species. A first taxonomic approach to distinguish Apodanthes and Pilostyles used the features of the floral scales as the primary criteria to key out the two genera (Harms, 1935; Solms-Laubach, 1878). These authors distinguished Apodanthes by the ''perigonial scales'' always epygynous, clawed, caducous, and inner scales slightly connate, versus Pilostyles, which has hypogynous outer scales, persistent, with a broad (not clawed) base, and free inner ones. In addition, Harms's (1935) classification included characters such as the number of perianth series and organs, the number of rings of pollen sacs, and the extent of the placenta. However, these characters are variable at specific and generic levels and therefore are taxonomically unreliable. For instance, the number of rings of pollen sacs is equivocal, as there are two rings in Apodanthes (see Blarer et al. 2004), two to four in Pilostyles, and one to two rings in Berliniache (treated as Pilostyles section Berliniache by Harms, 1935).
The recognition of Berlinianche as a distinct genus from Pilostyles (Vattimo-Gil, 1955) has been followed by other authors (e.g., Blarer et al. 2004: Heide-Jørgensen, 2008), who have provided several morphological characters to key out the three genera. We have summarized them as follows:
1. Whorls of scales three or six (rarely seven)-merous; perianth formula 3-6-6 or 3-3-5; inner whorl organs with a hair carpet at the base of the adaxial surface. Androecium forming a tubular synandrium with two whorls of pollen sacs, the inner ones forming a cylindrical column at the center of the flower, and the outer ones forming a hollow tube; sterile gynoecium in staminate flowers not fused to the synandrium; nectary disk conspicuous. Pollen inaperturate. ....................... Pilostyles (Berlinianche) aethiopica
1' Whorls of scales di- or tetramerous; perianth formulas 2-4-4 or 3-4-4; inner whorl organs lacking a basal hair carpet on the adaxial surface. Stamens in one whorl; sterile gynoecium in staminate flowers almost completely fused to the androecium; disk nectary inconspicuous. Pollen tricolpate. .......................................... 2
2. Flowers subtended by alternate bracts; perianth formula 2-4-4; organs of the outer whorl of scales caducous; organs of the inner whorl of the perianth fused in a small extension at their bases. Locule of ovary cruciform in transverse section, placentae not diffuse. Parasites primarily on Salicaceae. ..................................................... Apodanthes
2'. Flowers subtended by imbricate bracts; perianth formula 3-4-4; organs of the outer whorl of scales persistent; organs of the inner whorl of the perianth fused by a large portion of their bases. Locule of the ovary nearly circular in transverse section, placentae diffuse. Parasites on legumes. .............................................................. Pilostyles spp.
Alternatively, Blarer et al. (2004) identified the following sets of shared features, which are inconsistent with the key presented above: a) Apodanthes and Pilostyles (as Berlinianche) aethiopica share the doubling in the number of scales from the outer to the middle whorl, the isomerous middle and inner whorl, the stigmatic papillae covering the whole apical platform in carpellate flowers, and the style not narrower than the stigma; b) P. aethiopica and the remaining species of Pilostyles share legume parasitism, the perianth organs of the inner whorl caducous, with broad insertion, the presence of hairs in a ring in staminate flowers, the simple stigmatic unicellular papillae in carpellate flowers, and the placentae less protruding and bearing fewer ovules than in Apodanthes.
The recent combined (morphological plus nuclear plus mitochondrial) analysis carried out by Bellot and Renner (2014) for all members of the family renders the genus Berlinianche as nested within Pilostyles. Thus, the morphological traits of P. aethiopica described in the Apodanthes + P. aethiopica taxonomic scenario mentioned above appear to be homoplasic.
Apodanthaceae in Colombia. Two historical records of Apodanthaceae in Colombia are known. The first was reported by Karsten (1856), who briefly described Pilostyles ingae (under the generic name of Sarna), from the department of Cauca (''Ramis Ingae specierum insidens, in fluminis Cauca valle prope oppidum Popayan observata''; Karsten 1856: 415). The second record comes from the herbarium specimen I. F. Holton 84 (GH!, K!) collected on February 24, in La Paila, Valle del Cauca (Colombia) (figure 2). This specimen, initially identified as Apodanthes sp., bears two latter identifications as Pilostyles thurberi Gray in the GH specimen, and P. ingae (H.Karst.) Hook.f. (now P. blanchetii) in the K specimen. Isaac Farwell Holton (1812-1874) collected by mid-XIX century in Colombia as part of the project ''Flora Neogranadina Caucana''. The Kew specimen (available at http://plants.jstor.org/specimen/viewer/k000601220) is not fragmented and bears at least 52 flowers and floral buds that are densely arranged on the proximal portion of the host's branch. It also has two drawings of a flower and a floral bud. The Harvard specimen consists of approximately 20 flowers in different developmental stages, the largest of about 4 mm in diameter. The flowers are scattered in a short branch of ca. 5.5 cm long and 6 mm in diameter (figure 2). In both specimens, the leaves of the host are pinnately-compound, and the leaflets are oblong and minute (ca. 3 x 1 mm), which suggests that it belongs to the genus Calliandra (Mimosaceae). Both of these historical collections cannot be further identified as the material is rather fragile and fragmented, although according to their hosts, they could correspond to P. blanchetii.
Currently, the collections of Apodanthaceae from Colombia sufficiently documented are here identified at a species level as follows:
Apodanthes caseariae Poit.: PANAMA/COLOMBIA border: Southernmost peak of Cerro Pirre massif, Alto de Nique, 1300-1520 m, cloud forest, parasitizing a trunk of Casearia sp., flowers yellow, fruits brown, 19 Apr 1980 (fl, fr), A. Gentry et al. 28659 (COL)''. COLOMBIA: Department of Antioquia: Remedios, eastern slopes of the Central Cordillera, 500-1500 m of elevation, parasitizing Trema micrantha, 4 Jul 1989 (fl, fr), R. Callejas et al. 8062 (HUA); San Roque, Granja Experimental Corpoica, vereda Marbella, 800-1200 m, parasitizing Casearia, 14 Fb 1998 (fl, fr), R. Fonnegra et al. 6726 (HUA). Department of Santander: 150 km of Barrancabermeja, 74°4'W, 8°15'N, Mico Ahumado camp, 22 Aug 1966 (fl), 900 m, found by the authors in the specimen J. de Bruijn (COL; figure 4 F-H) filed under Casearia aculeata Jacq. Department of Casanare: carretera a Yopal, bosque de galería, río Charte, 19 Oct 1962, found by the authors in the specimen J. M. Idrobo 5225 (COL) filed under Casearia sylvestris Sw. (figure 4 A-D). Department of Valle del Cauca: Western Cordillera, ''vertiente occidental, hoya del río Sanquinini, lado izquierdo, La Laguna, 1250-1400 m, 10-20 Dic 1943'', found by the authors in the specimen J. Cuatrecasas (COL) filed under Casearia megacarpa Cuatr. (figure 4 E); finca Kyburz, eastern slope above the Bitaco River, Bitaco valley 1 km above and east of Bitaco, Pacific slope of the Western cordillera, 4500 ft, 16 Nov 1963, found by the authors in the specimen P. C. Hutchison & J. M. Idrobo 3010 (COL), filed under Casearia megacarpa Cuatr.
Pilostyles boyacensis F. González & Pabón-Mora: COLOMBIA: Department of Boyacá: Chíquiza, vereda Juan Díaz, 2415 m, 30 Nov 2013 (fl, fr), F. González 4508 (COL, HUA, NY); Sáchica, vereda El Espino, 2202 m, 01 Dec 2013 (fl fr), F. González 4509, 4510 (COL, HUA, NY); Sáchica, vereda El Espino, 2200 m, 15 Dec 2013 (fl fr), F. González 4517, 4518 (COL, HUA, NY); Sáchica, vereda El Espino, 2205 m, 01 Mar 2014 (fl fr), F. González 4519 (COL, HUA, NY); Villa de Leyva, vereda La Cañuela, 2135 m, 450 m después de la entrada al parque Gondava, 23 Mar 2014 (fl, fr), F. González et al. 4521 (COL, HUA, NY); Soatá, Cañón del Chicamocha, 2000-2100 m, Dec 1952 (fl, fr), found by the authors on the Dalea cuatrecasasii specimen J. Hernández 665 (COL).
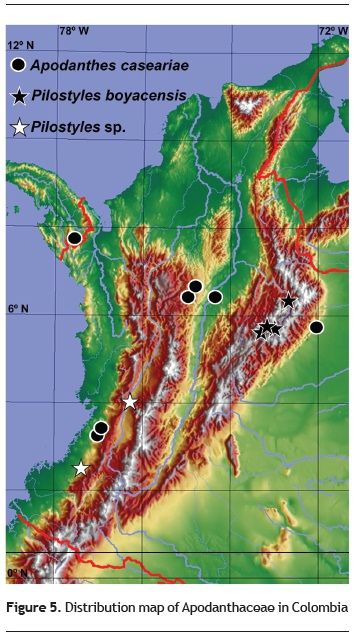
Ecological and chorological aspects. The two genera Apodanthes and Pilostyles do not share hosts, as the first parasitizes primarily Salicaceae (formerly Flacourtiaceae) and the second parasitizes Fabaceae (table 1). Worldwide, the geographic areas of distribution of the hosts are much wider than those of the parasites (table 1). This might also be the case in Colombia, as the distribution of Apodanthaceae known to occur in the country (summarized in figure 5) are considerably narrower that those of the hosts (not mapped), either members of Fabaceae, Burseraceae, Meliaceae or Salicaceae, or the genus Trema (Cannabaceae).
A map of the distribution of Apodanthaceae in Colombia (figure 5) indicates that they can grow in wet or dry forests, at elevations between 500 and 2415 m, on both sides of the Andes, as well as in mid-elevation forests of the Cauca and the Magdalena valleys, and in inter-Andean dry valleys of the Eastern Cordillera. In Colombia, they appear to set flowers and fruits mainly during or after the rains, in April, and from October to December.
A swelling in the host stem appears to be the first indication of infection. Flower emergence moves progressively up the host stem such that the next season's flowers emerge from the distalmost parts of the host twigs (Dell et al. 1982). According to Solms-Laubach (1874), the branching of the primary root of Pilostyles hausknechtii is not superficial but it occurs in the cambial region of its host. However, this observation requires further corroboration. The endophyte consists of uniseriate filaments and radial sinkers (Kuijt et al. 1985), formed by cells with large nuclei, two nucleoli and a dense cytoplasm (Riopel and Timko, 1995). At least in Pilostyles, the endophyte develops isophasically with respect to the host branch (Kuijt, 1969), that is, it keeps pace with the primary growth of the host shoot apex; however, the vegetative cells of the parasite do not reach the host apical meristem (but see Heide-Jørgensen, 2008, who described that the endophyte develops multicellular strands that can penetrate host tissue to the very shoot tip at least in Pilostyles). The endophytic cells of Pilostyles develop extensions from the host cortex to the vascular tissue. According to Dell et al. (1982), the vegetative endophyte of Pilostyles takes primarily phloem-derived nutrients, whereas the reproductive portion takes nutrients both from the host phloem and xylem. Pilostyles ingae affects its host Mimosa maguirei by causing an increase in branching but a decrease in the branch length (Gomes and Fernandes, 1994). Host anatomical responses also occur in Mimosa spp. parasitized by P. ulei and include changes in the size of the plant, vessel diameter, length and number and fiber length, perhaps to compensate for the required hydraulic properties of the host (Amaral, 2007; Amaral and Ceccantini, 2011).
The life cycle, reproduction and dispersal mechanisms of the New World species have not been investigated in detail. The flowers of Apodanthaceae last only a few days. Pilostyles thurberi is perennial, but plants appear to flower for only two years, dying afterwards (Heide-Jørgensen, 2008). African and Iranian members of Apodanthaceae are fly-pollinated, although flowers are also visited by ants, bees, butterflies and wasps (Bellot and Renner, 2013; Sipes et al. 2014). At least in the ripened fruits remain attached to the host until the following flowering period (figure 1G, I); thus, the same individual host can have floral buds, anthetic flowers, fruits and seeds simultaneously (figure 1).
In the New World Apodanthes caseariae, Trigona bees and Thraupis birds have been reported as flower visitors and fruit eaters, respectively (Gómez, 1983). According to Heide-Jørgensen (2008), harvest ants (Messor pergandei) are likely the pollinators and fruit dispersers of P. thurberi, as they need to walk over the parasite on their way up to collecting the rewards of the host Dalea emoryi. We have also seen frequent visits of a number of floral visitors (including Camponotus ants, Vespidae, shield bugs, and small beetles) in the dry areas where the Pilostyles-Dalea association occurs in Colombia. According to Sipes et al. (2014), only the bee Augochloropsis metallica and potter wasps are effective pollinators or Pilostyles thurberi, a process that could be mediated by the secretion of raspberry ketones and eugenols during anthesis of this species.
Guidelines to collect Apodanthaceae. The following field observations are necessary to improve the information contained in the herbarium specimens of Apodanthaceae, and in turn, to better understand the natural history, morphology, and taxonomy of these plants:
1. Occurrence of isophasic development of the parasite in the case of Pilostyles. This can be detected if the floral buds are found primarily towards the distal portions of the branch host, and the anthetic flowers and fruits towards the proximal portions of the host.
2. Detection of dioecy versys monoecy on each floral row. The staminate flowers can be detected by the presence of a white, collar row of digitiform, vesicular hairs above the pollen sacs, which is visible from the outside of the flower. The carpellate flowers are more globose at the base, and lack the collar-like roof of hairs on the central column.
3. Discharge of floral scent, particularly strong at noon.
4. Presence of flower and/or fruit visitors. The peaks of visitors detected by the authors, at least in Pilostyles, occur at noon.
5. Given that dry specimens are extremely hard to be dissected and loose much of the diagnostic characters, it is highly recommended to fix either in FAA or 70% ethanol each infected host branch in individual tubes or jars with its distal end visually marked, and to record each sample with a good quality and scale documented photographic series. These preserved samples will be critical for further dissection and standard transverse and longitudinal anatomical sections in the laboratory.
6. Silica gel preserved tissues are also critical, as molecular markers have shown to be promising for species recognition (e.g., Thiele et al. 2008; Bellot and Renner, 2014). Here also the flower rows (preferably at pre-anthesis) become the ''sampling units'' at least in Pilostyles, assuming that all flowers of each row are connected underneath by the same endophyte, and that they all conform a single individual. Thus, each floral row tangentially cut-off from the host tissue needs to be collected as a separate sample in silica gel.
ACKNOWLEDGEMENTS
We thank Michaela Schmull, Emily Wood, Kanchi Gandhi and the curatorial staff of the Gray Herbarium of Harvard University, Cambridge (MA), for the permission to publish images of figure 2. We thank Sebastian González (Universidad Nacional de Colombia) and Vanessa Suaza- Gaviria (Universidad de Antioquia) for assistance in the field and Fernando Fernández (Instituto de Ciencias Naturales, Universidad Nacional de Colombia) for the identification of the insects reported here as floral visitors. We also thank the three anonymous reviewers for their valuable comments. NPM acknowledges the Fondo Primer Proyecto 2012, and the Estrategia de Sostenibilidad 2013-2014 at the Universidad de Antioquia (Medellín), Colombia.
REFERENCES
Amaral MM do. 2007. A strutura da angiosperma endoparasita Pilostyles ulei (Apodanthaceae): interface e impacto no lenho do Mimosa spp. [Dissertação de Mestrado]. [São Paulo (Brasil)]: Instituto de Biociências, Departamento de Botânica, Universidade de São Paulo. p. 85. [ Links ]
Amaral MM do, Ceccantini G. 2011. The endoparasite Pilostyles ulei (Apodanthaceae - Cucurbitales) influences wood structure in three host species of Mimosa. International Association of Wood Anatomists Journal, 32: 1-13. [ Links ]
Barkman TJ, McNeal JR, Lim SH, Coat G, Croom HB, Young ND, De Pamphilis CW. 2007. Mitochondrial DNA suggests at least 11 origins of parasitism in angiosperms and reveals genomic chimerism in parasitic plants. BMC Evolutionary Biology, 7: 248. [ Links ]
Barneby RC. 1977. Daleae Imagines. An illustrated revision of Errazurizia Philippi, Psorothamnus Rydberg, Marina Liebmann, and Dalea Lucanus emend. Barneby, including all species of Leguminosae tribe Amorphae Borissova ever referred to Dalea. Memoirs of the New York Botanical Garden, 27: 1-892. [ Links ]
Bellot S, Renner SS. 2013. Pollination and mating systems of Apodanthaceae and the distribution of reproductive traits in parasitic angiosperms. American Journal of Botany, 100: 1083- 1094. [ Links ]
Bellot S, Renner SS. 2014. The systematics of the worldwide endoparasite family Apodanthaceae (Cucurbitales), with a key, a map, and color photos of most species. Phytokeys, 36: 41-57. [ Links ]
Blarer A, Nickrent DL, Endress PK. 2004. Comparative floral structure and systematics in Apodanthaceae (Rafflesiales). Plant Systematics and Evolution, 245: 119-142. [ Links ]
Blassingame JL. 1968. Revision of the genus Pilostyles in the southwestern United States. [M. Sc. Thesis]. [Harlingen (U. S. A.)]: Graduate Faculty, Texas Technological College. p. 63. [ Links ]
Brown R. 1845. Description of the female flower and fruit of Rafflesia arnoldii, with remarks on its affinities and an illustration of the structure of Hydnora africana. Transactions of the Linnean Society of London, 19: 221-248. [ Links ]
Cerros-Tlatilpa R, Espejo-Serna A. 1998. Contribución al estudio florístico de los cerros El Sombrerito y Las Mariposas (Zoapapalotl) en el municipio de Tlayacapan, Morelos, México. Polibotánica, 8: 29-46. [ Links ]
Dell B, Kuo J, Burbidge AH. 1982. Anatomy of Pilostyles hamiltonii C. A. Gardner (Rafflesiaceae) in stems of Daviesia. Australian Journal of Botany, 30: 1-9. [ Links ]
Eichler AH. 1878. Blüthendiagramme, vol. 2. Leipzig (Germany): W. Engelmann, p. 575. [ Links ]
Eliasson UH. 1994. Rafflesiaceae. In: Harling G, Andersson L, editors. Flora of Ecuador. Copenhagen (Denmark): Council for Nordic Publications in Botany. p. 43-49. [ Links ]
Endlicher S. 1841. Enchiridion Botanicum. Leipzig (Germany): Sumptibus G. Engelmann, p. 763. [ Links ]
Endress PK, Stumpf S. 1990. Non-tetrasporangiate stamens in the angiosperms: Structure, systematic distribution and evolutionary aspects. Botanische Jahrbücher für Systematik, 112: 193-240. [ Links ]
Endriss W. 1902. Monographie von Pilostyles ingae (Karst.) (Pilostyles Ulei Solms-Laub.). Flora, 91: 209-236, Table XX. [ Links ]
Engler A. 1912. Syllabus der Pflanzenfamilien. Berlin (Germany): Gebrüder Borntraeger. p. 387. [ Links ]
Felger RS. 1988. Pilostyles, strange desert parasite. The Plant Press, 12: 6-7. [ Links ]
Filipowicz N, Renner SS. 2010. The worldwide holoparasitic Apodanthaceae confidently placed in the Cucurbitales by nuclear and mitochondrial gene trees. BMC Evolutionary Biology, 10: 219. [ Links ]
Gomes AL, Fernandes GW. 1994. Influence of parasitism by Pilostyles ingae (Rafflesiaceae) on is host plant, Mimosa naguirei (Leguminosae). Annals of Botany, 74: 205-208. [ Links ]
Gómez LD. 1983. Rafflesiaceae. In: Burger W, editor. Flora costarricensis. Chicago (U. S. A.): Fieldiana Botany. p. 89-93. [ Links ]
González F, Stevenson DW. 2000. Gynostemium development in Aristolochia (Aristolochiaceae). Botanische Jahrbücher für Systematik, 122: 249-291. [ Links ]
Guillemin M. 1834. Mémoire sur le Pilostyles, nouveau genre de la famille des Rafflesiacées. Annales des Sciences Naturelles, Botanique, séries 2, 2: 19-25. [ Links ]
Harms H. 1935. Rafflesiaceae. In: Engler A, Prantl K, editors. Die natürlichen Pflanzenfamilien. 2th ed. Leipzig (Germany): W. Engelmann 16b: 243-281. [ Links ]
Heide-Jørgensen H. 2008. Parasitic flowering plants. Leiden (Netherlands): Brill. p. 438. [ Links ]
Huber H. 1960. Zur Abgrenzung der Gattung Aristolochia L. Mitteilungen der Botanischen Staatssammlung München, 3: 531-553. [ Links ]
Hutchinson J. 1959. The families of flowering plants. Vol. I. Dicotyledons. Oxford (U. K.): Oxford University Press. p. 511. [ Links ]
Idárraga A. 2011. Apodanthaceae. In: Idárraga A, Ortiz R, Callejas R, Merello M, editores. Flora de Antioquia. Catálogo de las Plantas Vasculares, Listado de las plantas vasculares del departamento de Antioquia. Medellín (Colombia): Editorial D'Vinni. Vol. 2. p. 268. [ Links ]
Igersheim A, Endress PK. 1998. Gynoecium diversity and systematic of the paleoherbs. Botanical Journal of the Linnean Society, 127: 289-370. [ Links ]
Jaffuel F. 1933. Notas sobre mis colectas entomológicas en las termas de Chillan. Revista Chilena de Historia Natural, 37: 148-152. [ Links ]
Karsten H. 1856. Plantae Columbianae. Linnaea, 28: 387-462. [ Links ]
Kuijt J. 1969. The biology of parasitic flowering plants. Berkeley (U. S. A.): University of California Press. p. 246. [ Links ]
Kuijt J, Bray D, Olson AR. 1985. Anatomy and ultrastructure of the endophytic system of Pilostyles thurberi (Rafflesiaceae). Canadian Journal of Botany, 63: 1231-1240. [ Links ]
Laudermilk J. 1945. Desert midget and jungle giant. The Desert Magazine, 8: 9-10. [ Links ]
Meijer W. 1993. Rafflesiaceae. In: Kubitzki K, editor. The families and genera of vascular plants. Berlin (Germany): Springer. Vol. 2. p. 557-563. [ Links ]
Melchior H. 1964. A. Engler's Syllabus der Pflanzenfamilien. Berlin (Germany): Gebrüden Borntraeger. Vol. 2. p. 666. [ Links ]
Mitchell JD. 2004. Rafflesiaceae. In: Smith N, Mori SA, Henderson A, Stevenson DW, Heald SV, editors. Flowering Plants of the Neotropics. Princeton (U. S. A.): Princeton University Press. p. 318-319. [ Links ]
Nickrent DL [Internet]. 2006. Apodanthaceae. The Parasitic Plant Connection. Accessed date: 15 November 2013. Available in: http://www.parasiticplants.siu.edu/Apodanthaceae/index.html. [ Links ]
Nickrent DL. 2008. Parasitic Plants. In: McGraw-Hill, editors. Yearbook of Science & Technology. New York (U. S. A.): McGraw-Hill. p. 251-253. [ Links ]
Nickrent DL, Blarer A, Qiu Y-L, Vidal-Russell R, Anderson FE. 2004. Phylogenetic inference in Rafflesiales: the influence of rate heterogeneity and horizontal gene transfer. BMC Evolutionary Biology, 4: 40. [ Links ]
Riopel JL, Timko MP. 1995. Haustorial initiation and differentiation. In: Press MC, Graves JD, editors. Parasitic plants. London (U. K.): Chapman & Hall. p. 39-79. [ Links ]
Robinson BL. 1891. Two undescribed species of Apodanthes. Botanical Gazette, 16: 82-84. [ Links ]
Rodríguez R, Grau J, Baeza C, Davies A. 2008. Lista comentada de las plantas vasculares de los Nevados de Chillán, Chile. Gayana Botánica, 65:153-197. [ Links ]
Rose JN. 1909. Studies of Mexican and Central American plants. No. 6. Rafflesiaceae. The North American species of Pilostyles. Contributions from the United States National Herbarium, 12: 262-265. [ Links ]
Rutherford RJ. 1970. The anatomy and cytology of Pilostyles thurberi Gray (Rafflesiaceae). Aliso, 7: 263-288. [ Links ]
Schaefer H, Renner SS. 2011. Phylogenetic relationships in the order Curcurbitales and a new classification of the gourd family (Cucurbitaceae). Taxon, 60: 122-138. [ Links ]
Sipes S, Huff Hartz K, Amin H, Anterola A, Nickrent D. 2014. Floral scent and pollinators of the holoparasite Pilostyles thurberi (Apodanthaceae). Journal of Pollination Ecology, 12: 31-39. [ Links ]
Solms-Laubach H. 1874. Ueber den Thallus von Pilostyles haussknechtii. Botanische Zeitung, 32: 49-59, 65-74, Taf. [ Links ] I.
Solms-Laubach H. 1878. Rafflesiaceae. In: von Martius CFP, Eichler AG, Urban I, editors. Flora Brasiliensis. Leipzig (Germany): Frid. Fleischer. Vol. 27 (4): 114-126. [ Links ]
Solms-Laubach H. 1891. Über die Species in der Gattung Rafflesia, insonderheit über die auf den Philippinen sich findenden Arten. Annales du Jardin Botanique de Buiterzorg, 9: 184-246. [ Links ]
Takhtajan AL. 1997. Diversity and classification of flowering plants. New York (U. S. A.): Columbia University Press. p. 643. [ Links ]
Thiele KR, Wylie SJ, Maccarone L, Hollick P, McComb JA. 2008. Pilostyles coccoidea (Apodanthaceae), a new species from Western Australia described from morphological and molecular evidence. Nuytsia, 18: 273-284. [ Links ]
de Vattimo-Gil I. 1955. Notice sur la tribu Apodantheae R. Br (Rafflesiaceae). Taxon, 4: 211-212. [ Links ]
de Vattimo-Gil I. 1956. Notes on Apodanthes caseariae Poit. and Pilostyles calliandrae (Gardn.) R. Br. (Rafflesiaceae- Apodantheae). Notulae Systematicae, 15: 225-229. [ Links ]
de Vattimo-Gil I.1978. Contribuição ao conhecimento da distribuição geográfica do gênero Pilostyles Guill. (Rafflesiaceae). Rodriguesia, 30: 7-11. [ Links ]
Yatskievych G. 1993. Rafflesiaceae. Journal of the Arizona-Nevada Academy of Science, 27: 239. [ Links ]
Yatskievych G, Meijer W. 2004. Rafflesiaceae. In: Steyermark JA, Berry PE, Yatskievych K, Holst BK, editors. Flora of the Venezuelan Guayana. St. Louis (U. S. A.): Missouri Botanical Gardens Press. Vol. 8. p. 407-410. [ Links ]