Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Caldasia
Print version ISSN 0366-5232
Caldasia vol.37 no.2 Bogotá July/Dec. 2015
https://doi.org/10.15446/caldasia.v37n2.54392
DARWIN M. MORALES-MARTÍNEZ
HÉCTOR E. RAMÍREZ-CHAVES
Grupo de Mastozoología, Grupo de Conservación y Manejo de Vida Silvestre, and Instituto de Ciencias Naturales, Universidad Nacional de Colombia, Carrera 45 No 26-85, Bogotá, Colombia.
School of Biological Sciences, University of Queensland, Goddard Building 8, St. Lucia 4072, Brisbane, Australia. hera.chaves@gmail.comABSTRACT
In order to clarify the distribution of bats of the genus Lasiurus in Colombia, we performed a morphological study of 49 specimens from four Colombian collections. Our review confirms the presence of three species in Colombia (L. blossevillii, L. cinereus, and L. ega) with distribution in all natural regions that include six of the nine biogeographic provinces. These species can be easily differentiated based on external and cranial characters. Lasiurus cinereus is the largest species (forearm length > 52 mm) and L. blossevillii is the smallest (forearm < 42 mm); L. ega has a distinctive skull shape with a developed sagittal crest. While previous literature make claims that L. egregius and L. castaneus occur in Colombia, our analysis found no support for this and even contradicts the evidence cited for their presence in the country. We demonstrate that one specimen attributed to L. egregius in literature corresponds to a misidentified L. blossevillii. Furthermore, we found that there are not any specimens of L. castaneus from the Department of Cundinamarca despite several reports in the literature. The status of subspecies in Colombia remains controversial and more specimens are needed to determine whether there are clear distinctions between forms in any geographical region. Most of the specimens examined were males and in some cases, were collected opportunistically from dead individuals found in cities in the Andean region.
Key words. Andes, Amazonas, Chiroptera, misidentifications, Pacific Region.
RESUMEN
Con el objetivo de clarificar la distribución de murciélagos del género Lasiurus en Colombia, se efectuó una revisión morfológica de 49 ejemplares de cuatro colecciones colombianas. Esta revisión confirma la presencia de tres especies en Colombia (L. blossevillii, L. cinereus, and L. ega), distribuidas en todas las regiones naturales que incluyen seis de las nueve provincias biogeográficas. Estas especies pueden diferenciarse fácilmente a partir de caracteres externos y craneales. Lasiurus cinereus es la especie de mayor talla (longitud del antebrazo > 52 mm), mientras que L. blossevillii es la más pequeña (antebrazo < 42 mm). Lasiurus ega presenta una forma distintiva de cráneo con una cresta sagital desarrollada. Aunque menciones previas en literatura sugieren la presencia de L. egregius y L. castaneus en Colombia, estos análisis no apoyan dichas inclusiones y contradicen la evidencia citada de la presencia de estas especies en el país. Se demostró que un ejemplar atribuido a L. egregius en literatura es una identificación errónea de L. blossevillii. Además, se encontró que no existen ejemplares de L. castaneus para el departamento de Cundinamarca, a pesar de varias menciones en literatura. El estatus subespecífico de estas especies en Colombia sigue siendo controversial y se requiere de ejemplares adicionales para determinar si existen distinciones claras entre diferentes formas asociadas a alguna región geográfica. La mayoría de los ejemplares revisados fueron machos y en algunos casos fueron encontrados casualmente muertos en ciudades grandes localizadas en la región Andina.
Palabras clave. Andes, Amazonas, Chiroptera, identificaciones erróneas, región Pacífica.
Recibido: 03/07/2015
Aceptado: 22/10/2015
INTRODUCTION
The genus Lasiurus comprises between 15-17 species of aerial insectivores distributed along North and South America (Simmons 2005, Gardner & Handley 2008). In Colombia, the number of species present is controversial. Historically, between three (Aellen 1970, Solari et al. 2013) and five species (Cuervo Díazet al. 1986, Alberico et al. 2000) have been reported. The first account of Lasiurus in Colombia provided by Aellen (1970) included the species L. borealis (Müller, 1766), L. cinereus (Palisot de Beavois, 1796), and L. ega (Gervais, 1856). Two additional taxa have historically been included in the Colombian fauna: L. castaneus and L. egregius. Cuervo-Díazet al. (1986) introduced records of L. casteneus (sic.) (= L. castaneus Handley 1960) in the warm mountain areas of the Department of Cundinamarca, and Rodríguez-Mahecha et al. (1995) included L. castaneus in Colombia as a whole. Neither publication, however, provided voucher specimens or specific localities.
Cuervo-Díaz et al. (1986) also listed L. egregius (Peters, 1870) as potentially present in Colombia based on records from adjacent countries. Alberico et al. (2000) followed the suggestions of Cuervo-Díazet al. (1986) and claimed that specimens of L. castaneus are housed in the collections of Instituto de Ciencias Naturales (ICN) in Bogotá. Later, Muñoz (2001) indicated that the presence of L. castaneus and L. egregius is only probable in Colombia. Bejarano-Bonilla et al. (2007) listed L. egregius for the Andean region (Department of Tolima) based on the capture of one individual housed in the Zoological collections of Universidad de Tolima (CZUT). This individual is the first reported specimen of L. egregius in Colombia. The records of Cuervo-Díazet al. (1986) and Bejarano-Bonilla et al. (2007), were not properly discussed in recent accounts of the genus (Gardner & Handley 2008, Solari et al. 2013). However, the distribution of L. egregius has been recently updated (López-Baucellset al. 2014) and two literature records from Colombia (Alberico et al. 2000 and Bejarano-Bonilla et al. 2007) were included in that update.
As Aellen (1970), Gardner & Handley (2008), and Solari et al. (2013) list only three species for Colombia: L. cinereus, L. ega, and L. blossevillii (Lesson, 1826). The latter is a replacement of L. borealis, which is now known to be restricted to North America (Morales & Bickham 1995). The number of subspecies registered in Colombia varies for each species and authors. Lasiurus blossevillii presents two subspecies: L. b. blossevillii (Lesson, 1826), from the Amazon basin drainage, and L. b. frantzii (W. Peters, 1870), from northern and western Colombia (Gardner & Handley 2008). Lasiurus cinereus is represented by only one subspecies along the national territory: L. c. pallescens (W. Peters, 1870). Although the subspecies L. c. villosissimus (É. Geoffroy St.-Hilaire, 1806) has been historically reported for the country, Gardner & Handley (2008) mentioned that the subspecies found in Colombia is L. c. pallescens, while L. c. villosissimus is found from central Peru south to Argentina. Between two (Gardner & Handley 2008) or three (Kurta & Lehr 1995) subspecies of L. ega have been mentioned for the country: L. e. ega (Gervais, 1856) for eastern Colombia (Kurta & Lehr 1995), L. e. panamensis (Thomas, 1901) for northern part of the country, and L. e. fuscatus (Thomas, 1901) for western Colombia (Kurta & Lehr 1995, Gardner & Handley 2008).
In order to clarify the distribution of genus Lasiurus and to address the debate on the presence of L. castaneus and L. egregius in Colombia, we reviewed specimens from 4 natural history collections. We provide an updated distribution of the species within the genus, a comparison of their morphological and morphometric traits, and some ecological notes based on vouchered literature records and specimens reviewed.
MATERIAL AND METHODS
We reviewed 49 specimens of genus Lasiurus from the collections of the Instituto de Ciencias Naturales (ICN), Bogotá; the Instituto Alexander von Humboldt, (IAvH), Villa de Leyva; the ColecciónZoológica, Universidad de Tolima (CZUT), Ibagué; and the Museo de Historia Natural de la Universidad del Cauca (MHNUC), Popayán. For each specimen, 10 cranial and external measurements were taken using digital calipers to the nearest 0.1 mm: hind foot length (HF) forearm length (FA), greatest length of the cranium excluding incisors (GLS), condyle-basal length (CBL), zygomatic breadth (ZB), mastoid breadth (BM), braincase breadth (BBC), breadth across postorbital constriction (POC), length of maxillary toothrow (CM3), and breadth across molars (M3M3). In addition, total body length (TL), tail length (TV), ear length (EAR), and weight (W) were taken from the specimen's labels.
We reviewed all available specimens housed at the ICN, searching for vouchers of L. castaneus as well as the specimen CZUT-M 0213 identified as L. egregius by Bejarano-Bonilla et al. (2007) to assess the accuracy of their identifications. We compared each specimen with the characters provided by Handley (1960), Shump & Shump (1982) and Gardner & Handley (2008). Additionally, we describe the morphological characters and their degree of variation in each of the species distributed in Colombia to facilitate future identification under field and laboratory conditions.
We created distribution maps for each species using specimen localities in addition to those available in the literature (Handley 1960, Gardner & Handley 2008), and housed in the collections of the American Museum of Natural History (AMNH), New York; Field Museum of Natural History (FMNH), Chicago; Muséumd'histoirenaturelle de la Ville de Genève (MHNG), Geneve; and Royal Ontario Museum (ROM), Ontario. Furthermore, we analyzed the presence or absence of each species among the different geographic regions and provinces of Colombia (Table 1) proposed by Hernández Camacho et al. (1992). Finally, ecological observations were obtained from information associated with reviewed specimens.

RESULTS
Based on the reviewed specimens, we identified three distinct species distributed in Colombia: L. blossevillii, L. cinereus, and L. ega (Table 1). These species are easily differentiated by external measurements and cranial characters (Table 2, Table 3, Fig. 1). We found no evidence for the presence of L. castaneus and L. egregius in Colombia. No specimens matched the diagnostic characters of L. castaneus in the collections reviewed. Furthermore, our review of the only reported L. egregius specimen from Colombia (CZUT-M 0213; Fig. 2) reveals that it belongs to L. blossevillii, based on the following cranial, external, and morphometric characters: a developed lacrimal ridge, rusty red pelage coloration, and small size (FA 39 mm). In contrast, L. egregius lacks a lacrimal ridge, has a bright red pelage, and a larger size (FA > 46 mm) (Shump & Shump 1982, Bianconi & Pedro 2007; Table 2, Fig. 1). The probable cause for CZUT-M 0213 misidentification is the presence of only one upper premolar in the specimen, a condition generally seen in L. egregius versus the two upper premolars found in L. blossevillii. We consider this condition a dental abnormality of the individual since all other measurements of CZUT-M 0213 most resemble those of other L. blossevillii specimens from Colombia (Table 2).
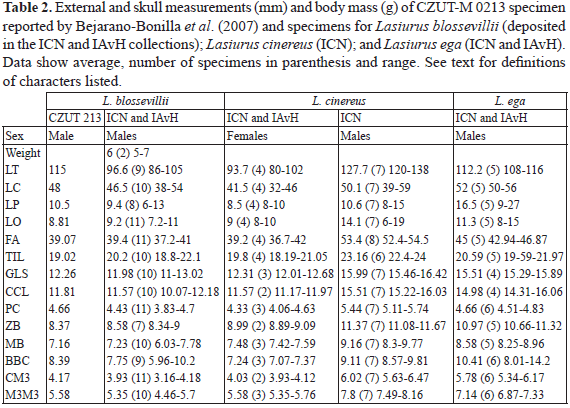


Lasiurus blossevillii was the most abundant in number of specimens in the visited collections with a total of 28 from the Amazonia, Peri-Caribbean arid belt, Guyana and North Andean provinces (Table 1; Fig. 3a). It can be identified based on the following characters: medium size (FA < 42 mm), with red-colored pelage varying from rusty red to brownish red, and dorsal pelage extending to the knees over the uropatagium. It has a small and short skull (GLS < 13.1 mm), with a developed lacrimal ridge (Fig. 1). The dental formula can be variable (I1/3 C1/1 P1-2/2 M3/3) and generally presents two upper premolars. No morphological differences associated to any geographical distribution were observed, and all specimens reviewed (n = 28) belong to the same subspecies, that we attributed to L. b. frantzii following Handley (1960) and Gardner & Handley (2008).

Lasiurus cinereus is known only from nine records in Colombian collections, all of these limited to Sierra Nevada de Santa Marta Province, the eastern slopes of Western Cordillera and western slopes of Eastern Cordillera of the North Andean Province (Table 1, Fig. 3b). It is the largest species of the genus in the country (FA 52.4-54.0 mm; Table 2) with frosty pelage mixed with dark brown, grey and white tips. The skull is comparatively large for the genus with a relatively short rostrum (Fig. 1), a poorly developed lacrimal ridge, and the dental formula I1/3 C1/1 P2/2 M3/3. All the specimens reviewed are similar in size and external morphology and belong to a single subspecies that we attributed to L. c. pallescens following Gardner & Handley (2008) suggestions.
Lasiurus ega is a medium sized bat (FA ~ 45 mm) with records in Colombian collections from the Amazon, Caribbean and Pacific regions, and six geographical provinces (Table 1, Fig. 3b, Appendix). It exhibits a high variation in pelage color, from gold to dusk white coloration, even within the same biogeographic region of Colombia. Therefore, they could not be clearly assigned to any of the three subspecies with suggested presence in Colombia neither with morphological characters nor in distribution. This species has the most distinctive cranial morphology of the three species in Colombia. It has a small but elongated and flattened skull with a pronounced sagittal crest and a poorly developed lacrimal ridge (Fig. 1). The dental formula is I1/3 C1/1 P1/2 M3/3.

Most of the Lasiurus specimens from Colombian collections were males (100% of L. cinereus, 75% of L. ega, and 75% of L. blossevillii; Table 2). For L. cinereus, the available reproductive information of six specimens show that males with inguinal testes were found from October to February. For L. ega five male specimens with reproductive activity are found from February to July. For L. blossevillii three male specimens were reproductively active from November to May. Furthermore, six female L. blossevillii nursing specimens were found from June to March. Finally, several records of L. blossevillii and L. cinereus are based on dead individuals found in large cities such as Bogotá, Villavicencio, and Cali.
DISCUSSION
Our review allows us to update and clarify some issues regarding the distribution of genus Lasiurus in Colombia. The distribution of L. cinereus (Gardner & Handley 2008) has been defined based on two marginal specimen localities in northern Colombia (Magdalena, Bonda; Allen 1900), one in Eastern Cordillera (Cundinamarca, Bogotá, Santa Isabel; Sanborn & Crespo 1957), and one in Central Cordillera, in an elevational range between 1700 and 3500 m (Solari et al. 2013), with no records for southern Colombia (Gardner & Handley 2008). We found two records from the eastern slopes of Western Cordillera in the departments of Cauca (Ramírez Chaves & Pérez 2011) and Valle del Cauca (Appendix), which extend the distribution of L. cinereus to the Eastern Andes of south-western Colombia (Fig. 3b). Although Solari et al. (2013) listed both departments in the distribution of the species, Western Cordillera was not included. These records also close the distance between mainland Colombian populations (L. cinereus pallescens) and the closest Peruvian populations (L. c. villosissimus) in more than 350 km, reducing the distance between both subspecies to 1250 km approximately. This increases the possibility that both populations belong to one single subspecies. Furthermore, the current restriction of the subspecies L. c. villosissimus to central Peru south through Bolivia, Paraguay, Uruguay, Brazil, and central Argentina (Gardner & Handley 2008) supports the inclusion of the records from Colombia (e.g. Tamsitt et al. 1964) into L. cinereus pallescens, as we found no reason to list more than one subspecies of L. cinereus for the country.
We found a similar result for L. blossevillii in Colombia, for which the distribution was based on two marginal localities: one in the western slopes of the Eastern Cordillera (Cundinamarca, Sasaima), and a second in the upper Cauca River basin in the eastern slopes of the Western Cordillera (Cauca, Mazamorrero). We found additional records from the inter-Andean valleys of upper Cauca river basin in the departments of Cauca apart of the record from Mazamorrero (Gardner & Handley 2008), the Valle del Cauca (Rojas-Díazet al. 2012; not included in Solari et al. 2013), and also from the Patía river basin (Ramírez-Chaves et al. 2010) that fill gaps in the recorded distribution of the species, and increase the elevational range to 300 to 2814 m (from 500-2600 m; Solari et al. 2013). Despite reports of two subspecies of L. blossevillii in Colombia by Gardner & Handley (2008), we found that all the reviewed specimens in Colombian collections can be attributed to only one subspecies: L. b. frantzii. Although the subspecies L. b. blossevillii has been suggested as present in the Amazon basin drainage of Colombia (Gardner & Handley 2008), the only specimen from the upper Amazon basin (ICN 16914) is indistinguishable from the L. b. frantzii specimens found in other regions of Colombia. However, to date there are not available specimens from lower part of the Amazonia Province of Colombia to raise further conclusions.
For L. ega, the distribution includes the lowlands of Colombia as reported by Gardner & Handley (2008) and Gonzalez et al. (2008). Although only four localities from Colombia have been listed in the most recent update of the distribution of L. ega in America (Leal & Gomes-Silva 2015), we found 12 confirmed localities from six of the nine geographic provinces or the country (Table 1; Appendix). Subspecies identification, however, is controversial. The external characters provided by Thomas (1901) to differentiate among subspecies reportedly distributed in Colombia (Thomas 1901, Gardner & Handley 2008) are ambiguous, especially considering the high variation of coloration observed in Colombian specimens. Thus, we assigned all L. ega specimens in Colombian collections to the nominal subspecies, pending additional comparisons using genetic information of specimens from the eastern and western part of Colombia. The elevational upper limit (1860 m; Table 1) for L. ega in Solari et al. (2013) is 900 m approx. higher than any locality of the specimens reviewed (Appendix), and we considered this elevational record as dubious until more evidence supporting Solari et al.´s (2013) range is provided.
Besides repeated mentions on the presence of L. castaneus and L. egregius in Colombia (Cuervo-Díazet al. 1986, Rodríguez-Mahecha et al. 1995, Bejarano-Bonilla et al. 2007, López-Baucellset al. 2014), we concur with Gardner & Handley (2008), and Solari et al. (2013) in excluding these taxa for the country. None of the records included in literature for these species is supported with voucher specimens, considering that "L. egregius" with specimen CZUT-M 0213 by Bejarano-Bonilla et al. (2007) here is re-identified as L. blossevillii. This misidentification was probably caused by the presence of only one upper premolar in CZUT-M 0213. However, presence/absence of a second premolar is not rare in some Lasiurus species, as shown by Handley (1960) with approximately 10% of individuals of each species showing variation. External characters also support the misidentification of CZUT-M 0213 which are not consistent with L. egregius: large body size (FA 48-50 mm), the reddish coloration pattern of the pelage with dorsal hairs with a dark base, a yellowish medium band, and reddish tips (Bianconi & Pedro 2007). Although L. egregius is potentially present in Colombia based on records from Panama and Brazil (Gardner & Handley 2008), to date there is no evidence of its presence in the country.
Confusion in the identification of Lasiurus in Colombia might be caused by inconsistencies in the characters used by different researchers. In reference to dental characters, Shump & Shump (1982) mentioned that L. egregius has two upper premolars, but the species generally has only one (Bianconi & Pedro 2007, Gardner & Handley 2008). Furthermore, wing characters are also controversial. Handley (1996), and Gardner & Handley (2008) mentioned that L. egregius has black wing membranes, with no conspicuously reddish coloration along the metacarpals and phalanges (paler than adjacent wing membranes in dried specimens). In contrast, Bianconi & Pedro (2007), and López-Baucellset al. (2014) reported that L. egregius has wings with a paler region along metacarpals and phalanges than the adjacent wing membranes. We are not able to say which combination of characters is the correct, considering that the species is not being registered to date in Colombia
Ecological notes
The species of the Lasiurus genus are not commonly caught in mist nests (Bianconi & Pedro 2007), and most records of L. blossevillii and L. cinereus are occasional and generally come from cities in which they are found dead. Although museum data are always biased due variable collection techniques (Cryan 2003), the remarkably uneven ratio of male vs. female specimens found in Colombian collections, might be caused by sex differences in migratory patterns and/or physiological constraints. For L. cinereus, the absence of female specimens from mountainous regions during summer has been suggested as possibly associated to physiological inability to raise young in these areas due to the climatic conditions and low density of resources (Cryan 2003). Alternatively, altitudinal sexual segregation likely represents partial altitudinal migrants because sexes must occur sympatrically to mate (McGuire & Boyle 2013). Both, the physiological inability and the migration events could also being affecting the populations in Colombia and might explain the absence of females from high Andean ecosystems of the country.
Using alternate methods is recommended for capturing information on these bat species (such as recording echolocation calls) and the collection of additional specimens to increase the number of the samples in the country for future comparisons. Genetic analysis would be useful to determine the validity of the subspecies classifications currently in use and some ecological aspects, such as migratory movements, and reproductive periods of the species found in Colombia.
ACKNOWLEDGEMENTS
We thank Hugo López (ICN), Claudia Medina (IAvH), Pilar Rivas (MHNUC), Derly Constanza Yará (CZ-UT), for allowing us the review of specimens under their care. Manuel Ruedi provided information of the specimens from Colombia deposited at the Muséumd'histoirenaturelle de la Ville de Genève (MHNG) and used in Appendix. Felipe Suárez-Castro and two anonymous reviewers provided useful comments that improved this work. HERC thanks Ariel Marcy and Viviana González Astudillo for comments on an early version, Bernardo Ramírez Padilla, Maatiaak Ramírez, and the University of Queensland (UQCent and UQI scholarships) for support.
LITERATURE CITED
1. Aellen, V. 1970. Catalogue raisonné des chiropterès de la Colombie. Revue Suisse de Zoologie 77: 1-37. [ Links ]
2. Alberico, M., A. Cadena, J.I. Hernández-Camacho & Y. Muñoz-Saba. 2000. Mamíferos (Synapsida: Theria) de Colombia. Biota Colombiana 1: 43-75. [ Links ]
3. Allen, J.A. 1900. List of bats collected by Mr. H. H. Smith in the Santa Marta region of Colombia, with descriptions of new species. Bulletin of the American Museum of Natural History 13: 87-94 [ Links ]
4. Bejarano-Bonilla, D.A., A. Yate-Rivas & M.H. Bernal-Bautista. 2007. Diversidad y distribución de la fauna quiróptera en un transecto altitudinal en el departamento del Tolima, Colombia. Caldasia 29: 297-308. [ Links ]
5. Cuervo-Díaz, A., J.I. Hernández-Camacho, and A. Cadena. 1986. Lista actualizada de los mamíferos de Colombia anotaciones sobre su distribución. Caldasia 15: 71-75. [ Links ]
6. Cryan, P.M. 2003. Seasonal distribution of migratory tree bats (Lasiurus and Lasionycteris) in North America. Journal of Mammalogy 84: 579-593. [ Links ]
7. Bianconi, G.V.B. & W.A. Pedro. 2007. Familia Vespertilionidae. In N.R. dos Reis, A.L. Peracchi, W.A. Pedro & I.P. Lima (eds.). Morcegos do Brasil: 167-195, Universidade Estadual de Londrina, Paraná [ Links ].
8. Gonzalez, E., R. Barquez & J. Arroyo-Cabrales. 2008. Lasiurus cinereus. In The IUCN Red List of Threatened Species.Version 2014.3. <www.iucnredlist.org>. Downloaded on 17 February 2015. [ Links ]
9. Handley, C.O., Jr. 1960. Descriptions of new bats from Panama. Proceedings of the United States National Museum 112: 459-79. [ Links ]
10. Handley, C.O., Jr. 1996. New species of mammals from northern South America: Bats of the genera Histiotus Gervais and Lasiurus Gray (Mammalia: Chiroptera: Vespertilionidae). Proceedings of the Biological Society of Washington 109: 1-9. [ Links ]
11. Handley, C.O., Jr. & A.L. Gardner. 2008. Genus Lasiurus. In A.L. Gardner (ed.). Mammals of South America. Volume 1. Marsupials, xenarthrans, shrews, and bats: 188-189, The University of Chicago Press, Chicago. [ Links ]
12. Hernández Camacho, J.I., A.H. Guerra, R.O. Quijano & T. Walschburger. 1992. Unidades biogeográficas de Colombia. In G. Halffter, (ed.). La Diversidad Biológica de Iberoamérica: 105-151, Acta Zoológica Mexicana (n.s.), México. [ Links ]
13. Kurta, A. & G.C. Lehr. 1995. Lasiurus ega. Mammalian Species 515: 1-7. [ Links ]
14. Leal, E.S.B. & F.F. Gomes-Silva. 2015. Update compilation on the geographic distribution of Lasiurus ega (Gervais, 1856) (Mammalia, Chiroptera, Vespertilionidae), including the first record for the Caatinga in the state of Paraíba, northeastern Brazil. Chiroptera Neotropical 21: 1320-1331. [ Links ]
15. López-Baucells, A., R. Rocha, G. Fernández-Arellano & P. Estefano. 2014. Echolocation of the big red bat Lasiurus egregius (Chiroptera: Vespertilionidae) and first record from the Central Brazilian Amazon. Studies on Neotropical Fauna and Environment 49: 18-25. [ Links ]
16. McGuire, L.P. & W.A. Boyle. 2013. Altitudinal migration in bats: evidence, patterns, and processes. Biological Reviews 88: 767-786. [ Links ]
17. Morales, J. C. & J. Bickham. 1995. Molecular systematics of the genus Lasiurus (Chiroptera: Vespertilionidae) based on restriction-site maps of the mitochondrial ribosomal genes. Journal of Mammalogy 76: 30-749. [ Links ]
18. Ramírez-Chaves, H.E. & W. Pérez. 2011. Mamíferos (Mammalia: Theria) del departamento del Cauca, Colombia. Biota Colombiana 11: 139-169. (Dated 2010, printed 2011). [ Links ]
19. Ramírez-Chaves, H.E., F. Ayerbe-Quiñones, and O. Mejía-Egas. 2010. Mamíferos de la cuenca Alta del río Patía en el departamento del Cauca, Colombia. Boletín Científico, Centro de Museos, Universidad de Caldas 14: 92-113. [ Links ]
20. Rodríguez-Mahecha, J. V., J.I. Hernández-Camacho, T.R. Defler, M. Alberico, R.B. Mast, R.A. Mittermeier & A. Cadena. 1995. Mamíferos colombianos: sus nombres comunes e indígenas. Occasional Papers in Conservation Biology, Conservation International 3: 1-56. [ Links ]
21. Rojas-Díaz, V., M. Reyes-G. & M. Alberico. 2012. Mamíferos (Synapsida, Theria) del Valle del Cauca, Colombia. Biota Colombiana 13: 99-116. [ Links ]
22. Sanborn, C.C. & J.A. Crespo. 1957. El murciélago blanquizco (Lasiurus cinereus) y sus subespecies. Boletín del Museo Argentino de Ciencias Naturales "Bernardino Rivadavia" e Instituto Nacional de Investigaciones de las Ciencias Naturales, Buenos Aires 4: 1-13. [ Links ]
23. Simmons, N.B. 2005. Order Chiroptera. In D.E. Wilson and D.M. Reeder (eds.). Mammals Species of the World: a taxonomic and geographic reference 3a ed., Vol. 1, 2: 312-529, The Johns Hopkins University Press, Baltimore, USA. [ Links ]
24. Shump, K.A. & A.U. Shump. 1982. Lasiurus borealis. Mammalian Species 183: 1-6. [ Links ]
25. Solari, S., Y. Muñoz-Saba, J.V. Rodríguez-Mahecha, T.R. Defler, H.E. Ramírez-Chaves & F. Trujillo. 2013. Riqueza, endemismo y conservación de los mamíferos de Colombia. Mastozoología Neotropical 20: 301-365. [ Links ]
26. Tamsitt, J.R., D. Valdivieso & J. Hernández Camacho. 1964. Bats of the Bogota savanna, Colombia, with notes on altitudinal distribution of Neotropical bats. Revista de Biología Tropical 12: 107-15. [ Links ]
27. Thomas, O. 1901. New Neotropical Mammals, with a Note on the Species of Reithrodon. Annals and Magazine of Natural History 8: 246-255. [ Links ]













