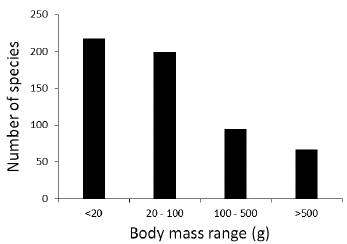INTRODUCTION
The Dry Tropical Forest (DTF) biome has a global distribution and encompasses a broad variety of ecosystem types, from open woodlands to dense forests that may be mostly or partially deciduous. There are multiple definitions of dry forest, but a defining characteristic is a pronounced seasonality with a drought period that represents a time of hydric stress for plants and other organisms (Portillo-Quintero and Sánchez-Azofeifa 2010). Fifty-four percent of the global extent of DTF occurs in South America and 12.5 % in Central America (Miles et al. 2006). Colombian dry forests represent 6.5 % of the biome's extent in the American continent and occur in seven regions, four of which are in the Andes; the other three are the Caribbean lowlands and islands, and the Llanos del Orinoco or Orientales (Portillo-Quintero and Sánchez-Azofeifa 2010, Pizano et al. 2014, 2016).
Dry forests are among the most threatened ecosystems on a global scale, due to anthropogenic transformation for agriculture and other uses (Portillo-Quintero and Sánchez-Azofeifa 2010). According to one estimate, 97 % of the remaining area of DTF throughout the tropics is at risk due to one or more factors such as landscape transformation, habitat fragmentation and climate change (Miles et al. 2006). In Colombia, the original area of DTF covered an extent of 88 829 km2, but 90 % has already been transformed into pastures, crops, and urban areas; the remaining area is highly fragmented (González-M et al. 2018). Thus, developing strategies for the conservation of dry forest biodiversity and ecosystem services at country and continental scales is necessary and urgent (Portillo-Quintero et al. 2015, Calvo-Rodríguez et al. 2016). To conserve functional, and therefore viable, ecosystems these strategies should be based on solid knowledge of ecological and evolutionary patterns and processes that may operate at different spatial and temporal scales.
Dryflor et al. (2016) performed an analysis of plant beta diversity, or spatial turnover in species composition, of dry forest for the Americas. This study analyzed patterns of similarity in species composition of 835 sampling sites from Mexico to Argentina, and identified clusters forming twelve floristic provinces, each with 23 % to 73 % exclusive species. At this scale Colombian dry forests fall into two clusters, the northern inter-Andean valleys (Cauca and Magdalena valleys) and a Central America-northern South America group that includes the Caribbean region. Dryflor et al. (2016) recommend considering this spatial differentiation in species composition for designing conservation plans that aim to conserve maximum diversity at a continental scale.
Under the principle of representation, the conservation of dry forest at a continental scale requires creating conservation areas that capture the uniqueness of each of the identified regions (Groves 2003). The patterns of beta diversity, and the processes that generate those patterns vary depending on the spatial scale. Therefore, effective conservation plans require the integration of initiatives at multiple scales, from local to regional, to represent not only the diversity of species but also the ecological and evolutionary processes that generate this diversity. At biogeographical scales, climate, geomorphology, and history are important determinants of differences among regions (Kattan et al. 2004, McKnight et al. 2007, Melo et al. 2009, Barton et al. 2013). At landscape scales, environmental heterogeneity and filtering result in the differentiation of species assemblages (Mac Nally et al. 2004, Veech and Crist 2007, Barton et al. 2013). At local scales, patterns of habitat use and dispersal capacities structure species assemblages in different habitat types (Mac Nally et al. 2004, Veech and Crist 2007, MacGregor-Fors and Schondube 2011, Barton et al. 2013).
Here we explore some of the challenges for drafting conservation plans that take into account beta diversity, through an analysis of the knowledge status and patterns of beta diversity at the scale of Colombia. We had three objectives. First, we evaluated the status of knowledge for Colombian dry forest, because local knowledge of ecological processes that affect ecosystem dynamics such as seed dispersal and forest regeneration is key for the effective design and management of protected areas (Sánchez-Azofeifa et al. 2005). We used species-area relationships and number of studies to assess how well different regions have been sampled. Second, we conducted an analysis of beta diversity of several taxonomic groups for different regions within the country. We aimed to determine how patterns of clustering of different regions changed when we included other taxa and downscaled the analysis to the country level. Third, we characterized patterns of seed dispersal syndromes and disperser diversity. Based on this characterization we explored some implications for forest regeneration
MATERIAL AND METHODS
Literature review
We used Google Scholar to search for publications with the terms "tropical dry forest" and "Colombia", in Spanish and English. Our search included up to October 2018. We also included papers from a local print journal (Cespedesia). Each study was classified into three categories: region, taxonomic group, and the topic of study.
Pizano et al. (2014) define seven regions of DTF in Colombia, but we did not include the Llanos region in our study because currently it has little pristine forest, and is mostly composed of open savannas, shrubby, and secondary vegetation. The six regions included in our study were: 1) the Insular region, composed by the island of Providencia in the Caribbean Sea, 2) the Caribbean region, which includes the Guajira Peninsula, the foothills of the Sierra Nevada de Santa Marta, the lowlands and delta of the Magdalena River valley, and the dry savannas of departments Córdoba, Sucre, and Bolívar, 3) the NorAndean region, in the northern end of the eastern range of the Andes, which includes the Chicamocha River canyon and the foothills of Serranía de Perijá, 4) the Patía River valley in departments Cauca and Nariño, 5) the middle basin of the Cauca River in departments Cauca, Valle del Cauca, and Risaralda, and 6) the mid and upper basin of the Magdalena Valley in departments Tolima and Huila. Studies at the country scale were classified in an additional Colombia category.
Studies were classified according to the taxonomic group at different levels. We used one category for each of plants, lichens and fungi, class for vertebrates (birds, mammals) and order for insects. We also classified studies according to research topic in three main areas: species inventories, ecology, and conservation. We further subdivided the ecology category into plant-animal interactions, seed dispersal, secondary succession, and a general ecology category. The conservation category included the topics of invasive species, management plans, and studies addressing the general conservation status of specific areas.
Data analysis
We used contingency tables (G-tests) to determine whether there were biases in the published literature. We made the following comparisons: the topic of study versus region, the taxonomic group versus region, and the taxonomic group versus study topic. We compiled lists of species for each region from inventories at the region and local patch scales, for the best represented taxonomic groups. Following Dryflor et al. (2016) we used the Simpson index to conduct analyses of similarity among regions for taxonomic groups.
Based on the information available in the online catalog of the Herbario Nacional Colombiano (COL) and in Vargas (2012), we classified plant species in five categories according to dispersal syndrome: water, wind, capsular, insects, and vertebrates. As birds are the main dispersal agent in tropical forests, we classified birds into trophic groups. Frugivorous and some omnivorous birds were considered potential seed dispersers. We further classified birds into body mass intervals. We compared the proportion of potential seed dispersers in different size classes to determine whether there were differences among regions, using G-test. Statistical tests were done in the program PAST v.2.17c (Hammer et al. 2001).
RESULTS
We found a total of 178 published studies on Colombian dry forest, with the first one dating from 1972. The number of studies rapidly increased in recent years, with more than three-fourths (N = 140) published since 2000 (Fig. 1). Almost half of the studies (N = 79) were conducted in the Cauca Valley, with the Caribbean following as the second most-studied region. In contrast, the Patía Valley had two studies. Lichens, Mantodea, Myriapoda and Mollusca were represented by only one study each, and Diptera, Fungi and Odonata had two studies each (Fig. 1).
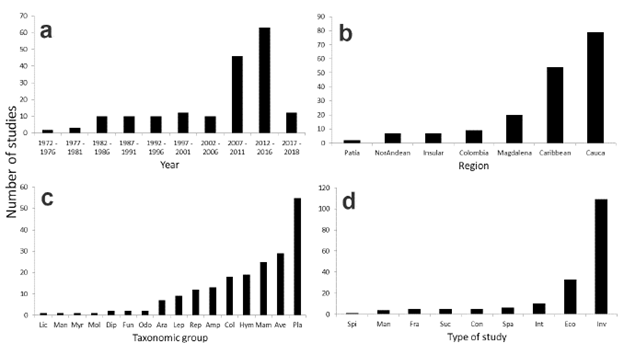
Figure 1 Distribution of number of studies on Colombian dry forests. a. Number of studies published per five-years intervals; b. Number of studies per each region; c. Number of studies by taxonomic group: LIC = Lichens, MAN = Mantodea, MYR = Myriapoda, MOL = Mollusca, DIP = Diptera, FUN = Fungi, ODO = Odonata, ARA = Arachnida, LEP = Lepidoptera, REP = Reptilia, AMP = Amphibia, COL = Coleoptera, HYM = Hymenoptera, MAM = Mammalia, AVE = Aves y PLA=Plants; d. Number of studies by type of study. SPI = Invasive species, MAN = Management plan, FRA = Fragmentation, SUC = Secondary succession, CON = Conservation status, INT = Plant-animal interactions, SPA = Species-area relations, ECO = General ecology, INV = Inventories.
Among insects, Hymenoptera, mostly ants, and Coleoptera had the largest number of studies (19 and 18, respectively), whereas plants, birds, and mammals were the most studied taxa. More than half of the studies (N = 109) were species inventories, mostly of plants, birds and mammals, particularly bats. There were only ten studies on plant-animal interactions and five studies on secondary succession. G-tests showed a dependence in the topic versus region comparison (χ2 = 35.6, P < 0.001, df = 12). There were more studies than expected in the conservation topic for the Cauca Valley and Caribbean regions. There were no biases in the two other comparisons: taxonomic group vs region (χ2 = 76.5, P = 0.84, df = 90) and taxonomic group versus topic (χ2 = 31.6, P = 0.39, df = 30).
There was insufficient information to analyze species richness as a function of area, but some patterns are worth mentioning. For species lists compiled at the regional level, the Caribbean region was atypical in having a small total number of recorded species of birds in relation to the area of remnant forest (Fig. 2). The Cauca Valley, on the other hand, was atypical in having a disproportionately large number of plant species in relation to the area of remnant forest, but this was also the region with the largest number of studies that have been conducted at the scale of forest fragments and the whole region.
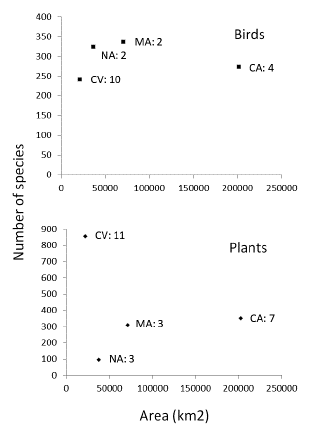
Figure 2 Number of species of birds and plants as a function of the remnant area of dry tropical forest in four regions of Colombia (data for remnant forest taken from García et al. 2014). CV = Cauca Valley, NA = NorAndean region, MA = Magdalena Valley and CA = Caribbean region. The number of inventories per region is shown.
Species inventories were insufficient for conducting rigorous analyses of similarity among regions for most taxonomic groups and regions. We conducted preliminary analyses for birds, bats and plants for different combinations of regions, depending on data availability (Fig. 3). For bats, we obtained counterintuitive clusters that may be an artifact of the small number of reported species and the fact that many species have broad geographic distributions. For both plants and birds, neighboring regions tended to cluster together: the Cauca and Magdalena valleys for plants and birds, the Caribbean and Insular regions for plants. Plants formed two geographically-defined clusters: Andean and Caribbean. Plants exhibited the highest levels of overall dissimilarity among regions, with only 20 % similarity between the two major clusters.
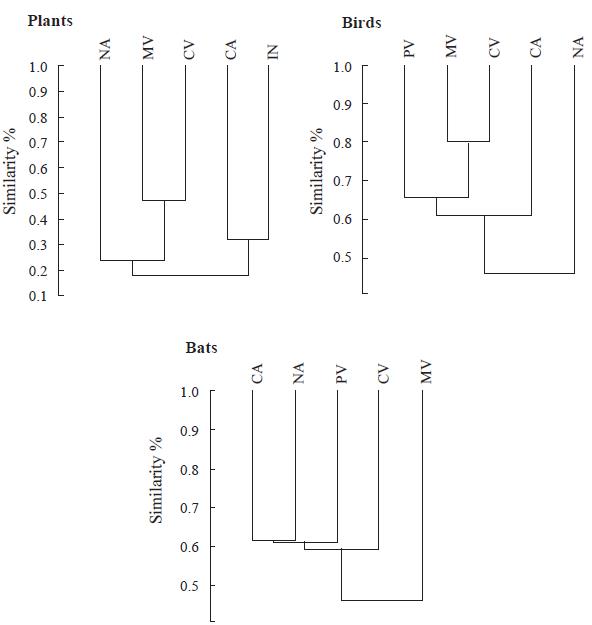
Figure 3 Analysis of similarity in species composition among regions of dry forest in Colombia for plants, birds and bats using the Simpson index. CV= Cauca Valley, MV = Magdalena Valley, PV = Patía Valley, CA = Caribbean, NA = NorAndean and IN = Insular.
We found that almost one-half (46.3 %) of the plant species in the Colombian dry forest are dispersed by birds and mammals (Fig. 4). This was followed by plants with dry capsules (31.3 %) and wind dispersal (20.4 %). Other dispersal mechanisms were represented in small proportions. For 24 species we could not determine their dispersal system. Of a total of 556 recorded bird species for all regions, 140 were potential seed dispersers. The percentage of potential dispersers in relation to the total number of bird species for the five regions varied between 17 % (Caribbean) and 25 % (NorAndean). The distribution of body masses of disperser birds was skewed towards the small sizes (Fig. 5). Only one species larger than 1000 g has been recently reported per region in the Caribbean, Cauca, and Magdalena valleys, two species of Cracids, Ortalis columbiana (Hellmayr, 1906) in the Cauca and Magdalena valleys and O. garrula (Humboldt, 1805) in the Caribbean region. The distribution of body masses was the same for each of these three regions (χ2 = 1.7, P = 0.9, df = 8).
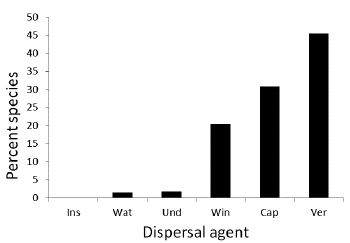
Figure 4 Distribution of plant species according to dispersal agent in Colombian dry forest. INS = Insects, WAT = Water, UND = Undefined, SPP = Spores, WIN = Wind, CAP = Capsular, VER = Vertebrates.
DISCUSSION
In spite of the recent increase of research on dry tropical forests in Colombia, our knowledge of these ecosystems is still insufficient. The species-area relationships that we found reveal the lack of knowledge in most basic aspects such as species inventories, even for commonly studied groups such as plants and birds. For example, the low number of bird species in the Caribbean region most likely reflects the small number of studies, which have been conducted at patch scales.
Research on Neotropical dry forests lags far behind research on wet forests, and most of it originates from a few sites in Mexico and Costa Rica; other countries remain virtually unstudied (Sánchez-Azofeifa et al. 2005). DTF research effort in Colombia has focused on producing species inventories of a few taxonomic groups, which is a critical first step. However, the design and management of protected areas, and indeed the sustainable management of unprotected dry forest remnants in rural landscapes, require adequate knowledge of ecological patterns and processes. Among many critical processes, seed dispersal and forest regeneration are key for maintaining the dynamics and long-term integrity of forest ecosystems (Sánchez-Azofeifa et al. 2005). A general knowledge base of tropical forest regeneration and seed dispersal is available for producing broad management guidelines (e.g., Dalling 2002, Guariguata and Ostertag 2002), but local knowledge is critical for producing site-specific management and restoration plans. This is particularly important given the fragmented nature of remaining forests, as key vertebrate seed dispersers may be absent.
At a continental scale, the patterns of beta diversity identified by Dryflor et al. (2016) are the result of biogeographical processes mostly determined by history, geomorphology, and climate. The main pattern of beta diversity that we found at the country scale, i.e., the differentiation between the inter-Andean valleys and the Caribbean-Insular regions, with species lists for the entire region, not specific sampling sites, agrees with the results obtained by Dryflor et al. (2016). Furthermore, based on climate, soil and vegetation data at the patch scale, González-M et al. (2018) identified three main floristic groups for Colombian DTF: Caribbean region, Orinoquia (Llanos), and inter-Andean valleys (this study included the Llanos but not the insular region). Thus, the same patterns of beta diversity at the country scale are supported by different studies based on different datasets.
Our results also show that for obtaining an adequate representation of beta diversity in conservation initiatives, analyses at smaller spatial scales are required to characterize the uniqueness of different regions within the country. Even between similar regions, such as the Cauca and Magdalena valleys (Kattan et al. 2004), there is 20 % dissimilarity in highly mobile organisms such as birds, and up to 50 % dissimilarity in plants. In addition, regions cluster in different patterns when comparing among taxonomic groups (Kattan et al. 2004).
Among regions within the country, beta diversity is indicative of macroecological and biogeographic processes that result in biotic differentiation. Thus, preserving the uniqueness of regional biotas requires understanding these processes and maintaining the conditions for their conservation. For example, large-scale landscape transformation of the lower Cauca Valley breached an ecological barrier that has facilitated the colonization of bird species from the Caribbean plains to the middle and upper valley (Garcés-Restrepo et al. 2012). This may lead to regional biotic homogenization and loss of beta diversity that for the most part goes undocumented.
Within regions, in contrast, habitat fragmentation may either increase or decrease beta diversity among habitat patches, depending on the interaction among factors such as patch size and isolation, and human disturbance. Habitat fragmentation may increase beta diversity at landscape scales because of sampling effects and differential extirpation of populations of different species in habitat patches (Kattan et al. 2006). Patch isolation and dispersal limitation may increase beta diversity by generating differences in species composition among patches (Jamoneau et al. 2012). However, increasing human disturbance may lead to biotic homogenization of habitat patches and a decrease of beta diversity (Vásquez-Reyes et al. 2017). The functional identity of species lost from habitat patches is in general deterministic, as some groups of species are more vulnerable. For instance, specialist and large-bodied vertebrate species are the first to disappear from tropical forest fragments (Sodhi et al. 2004). Therefore, small fragments mostly contain common, generalist species of small sizes.
The remaining Colombian dry forest is highly fragmented (González-M et al. 2018). Large frugivores are among the first species to disappear when tropical forests are fragmented (Sodhi et al. 2004). In the Cauca Valley, the only species of large mammal that persists in a few forest fragments is the red howler monkey, Alouatta seniculus (Linnaeus, 1766) (Gómez-Posada and Londoño 2012). Other important seed dispersers such as deer (Jara-Guerrero et al. 2018) and large birds such as toucans and cracids have been extirpated in the region (F. C. Lehmann, unpublished data 1960). Trees with large seeds such as Spondias spp., one the most representative taxons of the DTF, are dispersed by large mammals (Sampaio et al. 2018). Large frugivorous birds are probably absent in most remaining Colombian dry forests, and only about 20 % of remaining species, mostly small birds, are potential seed dispersers. Few data on the proportion of frugivores in well-conserved dry forest bird communities are available for comparison. In Bolivian dry forests, 29 % of species are frugi-granivores (Hertzog and Kessler 2002). Frugivorous birds exhibit strong population declines in response to drought conditions (Faaborg 1982). Thus, monitoring population trends of frugivores is a priority for dry forests.
Defaunation is a pervasive global phenomenon that is negatively affecting ecological functions and ecosystem services (Dirzo et al. 2014, Pérez-Méndez et al. 2016). On average, over 70 % of plants have animal-mediated seed-dispersal systems in the Neotropics (Almeida-Neto et al. 2008, Correa et al. 2015), thus, the loss of dispersers may affect a large proportion of plant species. In particular, the loss of large species of birds and mammals has negative consequences for the integrity of seed-dispersal networks, which in turn changes forest composition (Anzures-Dadda et al. 2011, Peres-Méndez et al. 2016). Large frugivores feed on a wide range of fruit sizes and act as hubs in seed dispersal networks and in their absence, networks collapse and plant communities tend to be dominated by fast-growing species with small fruit and seed sizes (Vidal et al. 2013, Palacio et al. 2016). In addition, large frugivores are important long-distance dispersers, which is an important ecological function in fragmented landscapes (Uriarte et al. 2011, Vidal et al. 2013).
CONCLUSIONS
Local basic research on most ecological patterns and processes may be perceived as descriptive and may not be scientifically innovative, but is essential for the successful conservation of DTF and tropical ecosystems in general. A large-scale program of research on DTF is required to generate relevant knowledge in key aspects such as seed dispersal (Sánchez-Azofeifa et al. 2005).
Our results show that patterns of beta diversity vary according to spatial scale, an attribute of the DTF (Dryflor et al. 2016). Thus, conserving this beta diversity, that is, maintaining the uniqueness of species assemblages in different regions, requires taking into account different processes that operate in the regional and local realms. In this context, conservation plans should be conceived to integrate local and national, and even continental, scales. The conservation of dry forests in Colombia requires an integrated plan that takes into account the continuum of processes that operate at the local patch scale (population dynamics, habitat use, local dispersal) through the landscape (environmental heterogeneity, source-sink dynamics) to the inter-regional scale.
A key aspect for the conservation of viable habitat patches is understanding the dynamics of natural disturbance and regeneration mediated by seed dispersal. The alteration of these critical processes affects the ecological integrity of forest fragments. In particular, small fragments may enter a vortex of degradation if regeneration is insufficient to counteract disturbance. Good knowledge of local seed dispersal systems is also crucial for restoration plans. Increasing the area of forest patches could be achieved by passive restoration (natural regeneration), if seed sources and seed dispersers are available. Otherwise, active restoration is required and should be based on solid knowledge of secondary succession (Wunderle 1997).