INTRODUCTION
Mangrove forests are tropical coastal ecosystems with a very high productivity and a complex food web, which support a great variety of organisms (López et al. 2009) and provide many ecosystem services. The aerial roots of the red mangrove, Rhizophora mangle L., provide a hard substrate in an area usually devoid of it, and form a three-dimensional habitat for sessile invertebrates and algae, as well as a refuge for juveniles of mollusks, crustaceans, and fish (Guerra-Castro and Cruz-Motta 2014, Ruiz-Guerrero and López-Portillo 2014).
The food web in this habitat has long been assumed to be based on detritus derived from mangrove litter (Odum and Heald 1972, Lugo and Snedaker 1974, Saifullah and Ahmed 2007). However, it has been proved that the algae on the prop roots may equal or surpass the primary productivity of mangrove trees (Rodriguez and Stoner 1990), and the macroalgal-derived detritus is exported from mangrove forests to adjacent ecosystems.
There is a plethora of studies on macroalgal communities associated with mangrove prop roots (i.e. Coppejans and Gallin 1988, Akil and Jiddawi 2001, Youseef et al. 2004, Schaffelke et al. 2005, Satpati et al. 2013, Shamina et al. 2014) around the world. In the Caribbean, the phyto-cenosis associated with mangrove roots is species-rich (López et al. 2009), and this has been attributed to water transparency and the stability of salinity and temperature year-round (Cordeiro-Marino et al. 1992). At present, there are no studies on the macroalgal flora associated with mangrove roots along the Caribbean coast of Colombia (Cordeiro-Marino et al. 1992), despite the great extension of mangrove forest (54 010 hectares, INVEMAR 2019) the region harbors. We present the first study on the macroalgae epiphytic on aerial roots of Rhizophora mangle L. at Cholón beach, Islas del Rosario National Park, Colombia.
MATERIAL AND METHODS
The National Park of Rosario Islands (10°8'-10° 15'North, 75°40'-75°48'West) is a protected area with an extension of about 120 000 hectares. The protected area harbors the most iconic coastal ecosystems of the Caribbean, including seagrass beds, coral reefs, mangrove forests and rocky shore habitats.
The region faces two rainy periods (May-June and October-November) and two dry periods (December-April and July-September). The mean temperature is 27.7 °C, and the relative humidity reaches 80-85 % (Valle et al. 2011).
The study site is in a small island on the west side of the Baru island (Fig. 1), facing a coastal lagoon, which communicates with the open sea through shallow channels (UAPNN 1998). The area is under the influence of the Dique Channel, which brings part of the freshwater and sediments of the Magdalena River, the main river in Colombia and the Caribbean basin. Salinity ranges between 32.2 and 33.0 ppt.
Three aerial roots of R. mangle were collected in September 2014, less than 50 m apart, and preserved in a 4 % formalin/seawater solution. In the laboratory the roots were analyzed, measured and photographed (Fig. 2a). The algae were removed, and their zonation on the root was recorded, dividing the root in sections of 5 cm (Coppejans and Gallin 1988). Algae were identified using an Olympus BX50 light microscope connected to a Moticampro 282B.

Figure 2 Different aspects of the samples found. a. Aerialroot with epiphytic algae, b-c. Bostrychia montagnei with stichidia (b) and cystocarps (c). Scale bars: 200 um, d-f. Cera-mium brevizonatum var. caraibicum, d. Habit. Scale bar: 2 mm, e. Tetrasporangium. Scale bar: 20 um, f. Inrolled apices. Scale bar: 100 um.
Nomenclature of the algal species follows Wynne (2017) and Guiry and Guiry (c2020). Voucher specimens were deposited in the herbarium of the Biology Department, Universidad Nacional de Colombia.
RESULTS
We report a total of 36 species: 21 species of Rhodophyta, four Phaeophyceae and eleven Chlorophyta (Table 1). The families with the highest species number were Rhodomelaceae with eight and Cladophoraceae with six. The most common taxa were Heterosiphonia crispella var. laxa (Børgesen) M.J.Wynne and Chroodactylon ornatum (C.Agardh) Basson. Of these taxa, 19 species are new records for the islands, and six others are new records for Colombia: Bostrychia montagnei Harvey (Figs 2b-c), Ceramium brevizonatum var. caraibicum H.E.Petersen & Børgesen (Figs 2d-f), Dasya caraibica Børgesen (Figs 3a-b), Melanothamnus pseudovillum (Hollenberg) Díaz-Tapia & Maggs, Parviphycus setaceus (Feldmann) J.Afonso-Carrillo, M.Sanson, C.Sangil & T.Diaz-Villa (Figs 3c-d), and Wittrockiella salina V.J.Chapman (Figs 3e-f). 53 % of the taxa recorded in the present paper are new additions for the local marine flora, while 17 % are new records for Colombia.
Table 1 List of algal species on mangrove roots. * = new record for Rosario islands, ** = new record for Colombia.

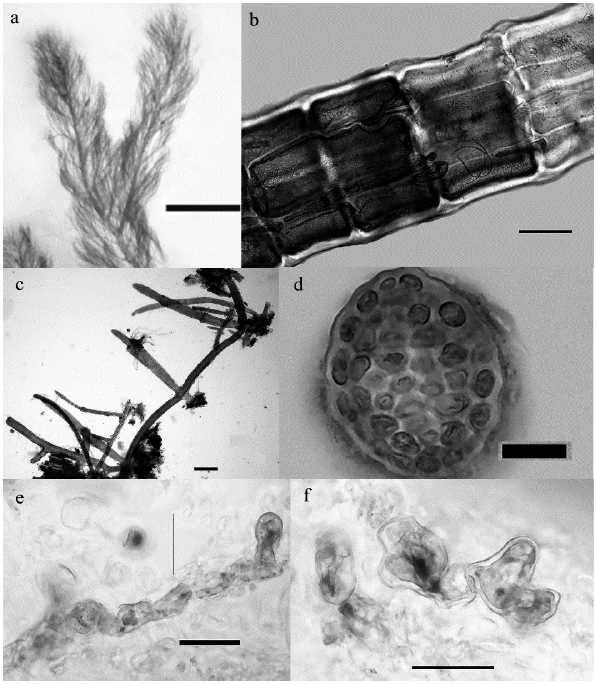
Figure 3 Some of the macroalgae registered in the study area. a-b. Dasya caraibica, a. Habit. Scale bar: 2 mm, b. Detail of partial rizoidal cortication. Scale bar: 100 um, c-d. Parviphycus setaceus, c. Habit. Scale bar: 200 um, d. cross section of thallus. Scale bar: 20 um, e-f. Wittrockiella salina, e. .Habit Scale bar: 5 um, f. detail of cells. Scale bar: 5 um.
We found that some species (Bostrychia montagnei, Ceramium brevizonatum var. caraibicum, Hypnea spinella (C.Agardh) Kützing, Dictyopteris delicatula J.V.Lamouroux, Bryopsis hypnoides J.V.Lamouroux and Bryopsis pennata J.V.Lamouroux) were restricted to the upper portion of the root. In the middle part of the root we observed Antithamnion antillanum Børgesen and Laurencia sp., while Hypnea valentiae (Turner) Montagne and Parviphycus setaceus were restricted to the deeper portion of the root. All the other taxa did not have a clear pattern of distribution; they were observed along the whole length of the root.
DISCUSSION
The number of algae registered in the present study is rather high if compared to previous works: El-Sharouny et al. (2001) reported 23 species of algae along the Red Sea; in the Australian Pacific West et al. (2013) found fifteen species; in Puerto Rico Rodriguez and Stoner (1990) recorded only eight species. Rios et al. (2019) observed fourteen taxa on the Caribbean coast of Panama, while along the Pacific coast of Colombia only 18 species were registered by Peña-Salamanca (2008).
The diversity that we observed is close to that reported from the studies carried out by Moreira et al. (2003), who reported 27 species of mangrove-associated algae in Cuba, and by López et al. (2009), who found 40 species of epiphytic algae along the Peninsula of Paguaná, Venezuela.
It is noteworthy that 25 species, corresponding to 69 % of the identified taxa, are new records, either for the study site or for Colombia. With this study, we increase the diversity of the marine flora in Rosario Island by 14.3 % (Diaz-Pulido and Diaz-Ruiz 2003). All the species observed are rather common in the Caribbean basin, and the new findings reflect the lack of studies, rather than new introductions to the area.
The genus Bostrychia, together with the genera Murrayella, Caloglossa and Catenella, form the Bostrychietum, an association of specialized red algae that grows on roots of different species of mangroves (Seixas et al. 2014). The presence of the species B. montagnei in this environment is therefore not surprising. To date, for the Colombian Caribbean coast, two species of Bostrychia have been reported: B. moritziana (Sonder) J. Agardh and B. tenella (J.V. Lamouroux) J. Agardh (Díaz-Pulido and Díaz-Ruiz 2003). For the Pacific coast of the country, Peña-Salamanca (2017) reported five species: B. calliptera (Montagne) Montagne, B. moritziana, B. radicans (Montagne) Montagne, B. binderi Harvey and B. tenella.
The taxon Ceramium brevizonatum var. caraibicum, originally described for Dominican Republic (Hispaniola island, Børgesen 1924) has been widely reported for all the Caribbean Sea from Bermuda to Brazil as well as at some localities in the Indian Ocean (Guiry and Guiry c2020).
The species is characterized by pseudodichotomous branching, pincer-like apices, 1-2 acropetal rows and one basipetal row of cortical cells and partially covered tetrasporangia (Schneider 2004, Barros-Barreto et al. 2006). Our specimen fits well the description of the species (Figs 2d-f). The genus Ceramium is very species-rich, with 30 taxa and eight varieties reported for the Western Atlantic (Wynne 2017). In Colombia, fourteen species have been registered (Diaz-Pulido and Diaz-Ruiz 2003), and recently a bloom-forming, undescribed species was observed at Cartagena (Gavio 2020).
The genus Dasya C. Agardh (1824) includes red algae with a hairy and delicate appearance and rigid to flaccid texture. The genus currently includes over 90 species distributed in sub-polar, temperate and tropical seas (Guiry and Guiry c2020), nineteen of which are reported for the Western Atlantic (Wynne 2017). Dasya caraibica, originally described by Børgesen (1919), is one of the two species reported for the Caribbean with light rhizoidal cortication (Fig 3b). Dasya rigidula (Kützing) Ardissone, the other lightly corticated taxon, differs mainly in the general aspect of the thallus (compact vs. fluffy in D. caraibica) and the falciform and thick branchlets (Rodriguez-Prieto et al. 2013), which in Dasya caraibica are long and delicate (Fig. 3a).
Melanothamnus pseudovillum, originally described as Polysiphonia pseudovillum by Hollenberg (1968) is a creeping species with closed-connected rhizoids, four pericentral cells, ecorticated, and branches replacing trichoblasts. In the Western Atlantic it has been reported for North Carolina (Schneider and Searles 1991), Florida (Littler and Littler 2000), Mexico (García-García et al. 2020) and Panama (Mamoozadeh and Freshwater 2011). Recently, Díaz-Tapia et al. (2017) reinstated the genus Melanothamnus, where this species was accommodated.
Parviphycus setaceus is a species originally described for the Antilles (Guiry and Guiry c2020) and widely distributed in the Caribbean Sea (as Gelidiella setacea (Feldmann) Feldmann and Hamel, Littler and Littler 2000). It is typically observed on mangrove prop roots (Littler and Littler 2000, Littler et al. 2008), where it grows inconspicuously.
Wittrockiella salina Chapman is a species originally described for New Zealand and distributed along Australia, Tasmania, and Japan, in the Pacific Ocean. In 1984 van den Hoek et al. (1984) reported the species as epiphytic on aerial roots of Rhizophora mangle and on pneumatophores of Avicennia nitida Jacq. on the island of Curacao, Netherland Antilles, in the Southern Caribbean Sea. The alga is small, inconspicuous, grows as an epiphyte on larger algae, so it may go easily undetected. It is difficult to determine if this species has been introduced in the Caribbean Sea, and expanding, or if mangrove aerial roots in the southern Caribbean basin have always been its natural environment.
Although the overall diversity of the biocenosis associated with mangrove prop roots is high (e.g. Riosmena-Rodriguez et al. 2011), single studies seldom report a high number of species. Some authors have regarded mangrove habitats as having a low diversity of epiphytes (Huisman et al. 2015); King (1981) suggested that these habitats are unfavorable due to a lack of stable substratum (excluding the mangroves), mud in suspension, and the physiological stress due to fluctuating salinity. Riosmena-Rodriguez et al. (2011) associated a high diversity of algae on mangrove roots to the presence of other habitats close-by, such as seagrass or rhodoliths beds and hard substrate such as rocky shores. When these habitats are not present, the algal diversity is much lower. In Cholón, the mangroves were adjacent to seagrass beds, and coral reef patches were observed in the area. Furthermore, artificial hard substrate is present close by in the form of spurs.
Considering that we analyzed only three roots, the diversity of this phytocenosis in the Park is grossly underestimated. Most species are microscopic, filamentous, and easily overlooked. Furthermore, for red algae, the reproductive structures are needed to be able to identify the sample to species. More research is needed to understand if stochastic events are structuring the epiphytic flora on single roots, or if other factors are involved. Eston et al. (1992) determined that on mangroves, early algal colonizers are also the final community, with no opportunistic species present and virtually no succession, as it has been reported for seagrass communities (den Hartog 1971, 1987).
Cordeiro-Marino et al. (1992) reported that for the Caribbean basin, the phytocenosis associated with mangroves was dominated by green algae. In our study, 59.4 % of the species belonged to Rhodophyta, 29.7 % were Chlorophyta and only 10.8 % were Phaeophyceae. These percentages correspond to the average composition of the marine flora in the Caribbean (e.g. Taylor and Rhyne 1970, Betancourt-Fernandez and Herrera-Moreno 2001, Suárez 2005). Diaz-Pulido and Díaz-Ruiz (2003) reported, for the Rosario islands, a marine flora dominated by red algae (50.8 %) followed by green algae (39.7 %) and then brown algae (9.3 %). For the mangrove roots, we report a higher percentage of red algae, which also corresponds to the majority of the new records. Recent studies on the seaweeds of another region of the Colombian Caribbean (e.g. Albis-Salas and Gavio 2011, 2015, Ortiz and Gavio 2012, Gavio et al. 2013, 2015, Rincón-Díaz et al. 2014, 2018), registered a high number of red algal species previously overlooked.
The low number of brown algae (Phaeophyceae) observed in this research is in agreement with other studies: López et al. (2009) found only three species of Phaeophyceae out of forty. Billah et al. (2016) reported only one Phaeophyceae of a total of eleven, while Pérez-Estrada et al. (2012) found a single species of brown algae of a total of 68. Rios et al. (2019) did not observe any brown algae on mangrove roots on the Caribbean coast of Panama.
The high diversity observed supports the importance of this community to the food web of mangrove ecosystems. This study by no means pretends to be comprehensive; considering the mangrove cover along the Caribbean coast of Colombia, more research is needed to address algal species diversity, distribution, seasonality and turn-over, to better understand the dynamics shaping these habitats.
















