Remark
| 1) Why was this study conducted? |
| Predominantly Antibody Deficiencies are the most frequent primary immune deficiency disorders affecting adults. Antibody deficiencies with normal IgG levels had been related with recurrent pneumonia and bronchiectasis. The frequency of humoral deficiencies in adults have not been evaluated in Colombia. |
| 2) What were the most relevant results of the study? |
| We find Antibody deficiencies with normal IgG levels in 10% of adult patients evaluated. Although unknown rates of pneumococcal vaccination, protective IgG anti-pneumococcal titles were find in most of the patients. |
| 3) What do these results contribute? |
| Immunological evaluation, including IgG, IgA, IgM, IgE IgG subclasses and IgG anti-polysaccharides could contribute to improve diagnosis and treatment outcomes in adult patients with bronchiectasis or recurrent pneumonia. |
Introduction
Predominantly Antibody Deficiencies are the most frequent primary immune deficiency disorders affecting adults. Predominantly antibody deficiencies clinical presentation is variable, but most patients are susceptible to recurrent infections, autoimmunity, inflammation, allergy or malignancy 1. Although, predominantly antibody deficiencies can manifest at any age, its diagnosis requires a high index of suspicion, but in most cases, its diagnosis is delayed, contributing to the development of complications 2. Antibody Deficiencies spectrum ranges from severe forms such as agammaglobulinemia (X-linked and autosomal recessive) to less severe conditions such as Specific Antibody Deficiency or IgG subclass deficiencies 3,4. Although IgG subclass deficiency or Specific Antibody Deficiency is an important cause of pulmonary complications, its frequency in adults is unknown.
Antibody deficiencies with normal IgG levels are related to infectious and non-infectious complications and lung damage. IgG response against Streptococcus pneumoniae is crucial to control and prevent pneumococcal complications 5. Antibodies are essential for encapsulated microorganism infection control because capsules inhibit macrophages and polymorphonuclear cells phagocytosis 6. Poor antibody response against polysaccharide antigens or Specific Antibody Deficiency increases susceptibility to encapsulated pathogens 7. Normal adults vaccinated with pneumococcal polysaccharide (23 serotypes vaccine) exhibit protective antibody titers against at least 70% of serotypes 8. Streptococcus pneumoniae and Hemophilus influenza infections in subjects with humoral deficiencies are associated with a high risk of morbidity and mortality.
Bronchiectasis is defined as the abnormal and permanent dilatation of the bronchi. Our research is focused on non-cystic fibrosis bronchiectasis. The etiology of bronchiectasis includes respiratory infections sequela, anatomical abnormalities, alpha-1-antitrypsin deficiency, inflammatory diseases, primary ciliary dyskinesia, and primary immunodeficiencies 6. Delayed diagnosis and inadequate management of Predominantly antibody deficiencies patients lead to irreversible lung damage or even death from severe infections. Unfortunately, many patients with Predominantly antibody deficiencies develop bronchiectasis due to delayed diagnosis 9. Previous studies 5,8 have shown that a proportion of patients with bronchiectasis may have a variety of immunodeficiency disorders, mainly subclass deficiency and specific antibodies against polysaccharide antigens. Recurrent pneumonia has been defined as at least two episodes of pneumonia in one year or more than three pneumonia throughout life, with radiographic resolution between episodes 4. Large series reports greater than 50% of the subjects with recurrent pneumonia criteria have disseminated bronchiectasis 9,10. However, the actual frequency of Predominantly antibody deficiencies in subjects with recurrent pneumonia in adults is unknown.
Predominantly Antibody Deficiencies are a neglected reality in Colombia and their frequency is unknown 11. Immune characterization of adults with bronchiectasis or recurrent pneumonia will establish specific therapeutic strategies, improve quality of life, and decrease disease burden. This study aimed to determine the frequency of antibody deficiencies with normal IgG levels in adults with bronchiectasis or recurrent pneumonia.
Materiales and Methods
Study design and participants
This is a cross-sectional study. We enrolled consecutive patients with non-cystic fibrosis bronchiectasis or recurrent pneumonia who were referred by pulmonologists, internists, or allergists from different clinical centers to the Clinical Immunology Service at Universidad del Valle (Cali, Colombia) from January 2nd to December 31st, 2019 for study porpoise.
Participants fulfilling the inclusion criteria were enrolled. Briefly: aged ≥14 and <65 years with bronchiectasis on chest computed tomography (CT) and the clinical syndrome of bronchiectasis (cough, sputum production, or recurrent respiratory infections) or with recurrent pneumonia (at least two cases of pneumonia in 1 year or more than three cases of pneumonia throughout life, with radiographic resolution between episodes) and not the exclusion criteria as an inability to give informed consent, bronchiectasis due to cystic fibrosis, and traction bronchiectasis associated with interstitial lung disease or another respiratory disorder, acquired immune defects or secondary immunodeficiencies (chronic myeloid leukemia, multiple myeloma, Human Immunodeficiency Virus HIV infection, immunosuppression secondary to drugs). Inborn errors of immunity were defined according to European. Society of Immunodeficiency classification and diagnostic criteria 12. Patients older than 65 were excluded from the study because the high frequency of bronchiectasis in this population is related to senescence.
Immunoglobulin quantification (IgG, IgA, IgM and IgE serum levels)
Blood samples were taken to measure serum immunoglobulin levels (IgG, IgA, IgM, and IgE) using nephelometry (Abbott, ARCHITEC c Systems, Germany). Hypogammaglobulinemia (IgG) was defined as serum IgG levels <700 mg/dL (reference value 700-1,600 mg/dL), this relative “high” value pretends increase possible immunodeficiency cases. IgA levels <70 mg/dL (reference value 70-400 mg/dL) or IgM levels <40 mg/dL (reference value of 40-230 mg/dL) 13. Selective Immunoglobulin A (IgA) deficiency was defined as serum IgA levels <7 mg/dL in two separate samples 12. Immunoglobulin deficiency was defined according to literature 14.
IgG subclass quantification
IgG2 subclass concentration was determined using the enzyme-linked immunosorbent assay method using the commercial kit (Human IgG2 ELISA Invitrogen, THERMO FISHER, Catalog number: BMS2093 -96 tests) 15 in all subjects. IgG2 ELISA values lower than 175 mg/dL (ELISA) were confirmed using nephelometry (SIEMENS, BN II System, Germany). In addition, patients with low IgA were evaluated using nephelometry for IgG subclasses (IgG1, IgG2, IgG3, and IgG4).
IgG levels against Streptococcus pneumoniae (anti-pneumococcal antibodies 10 serotypes)
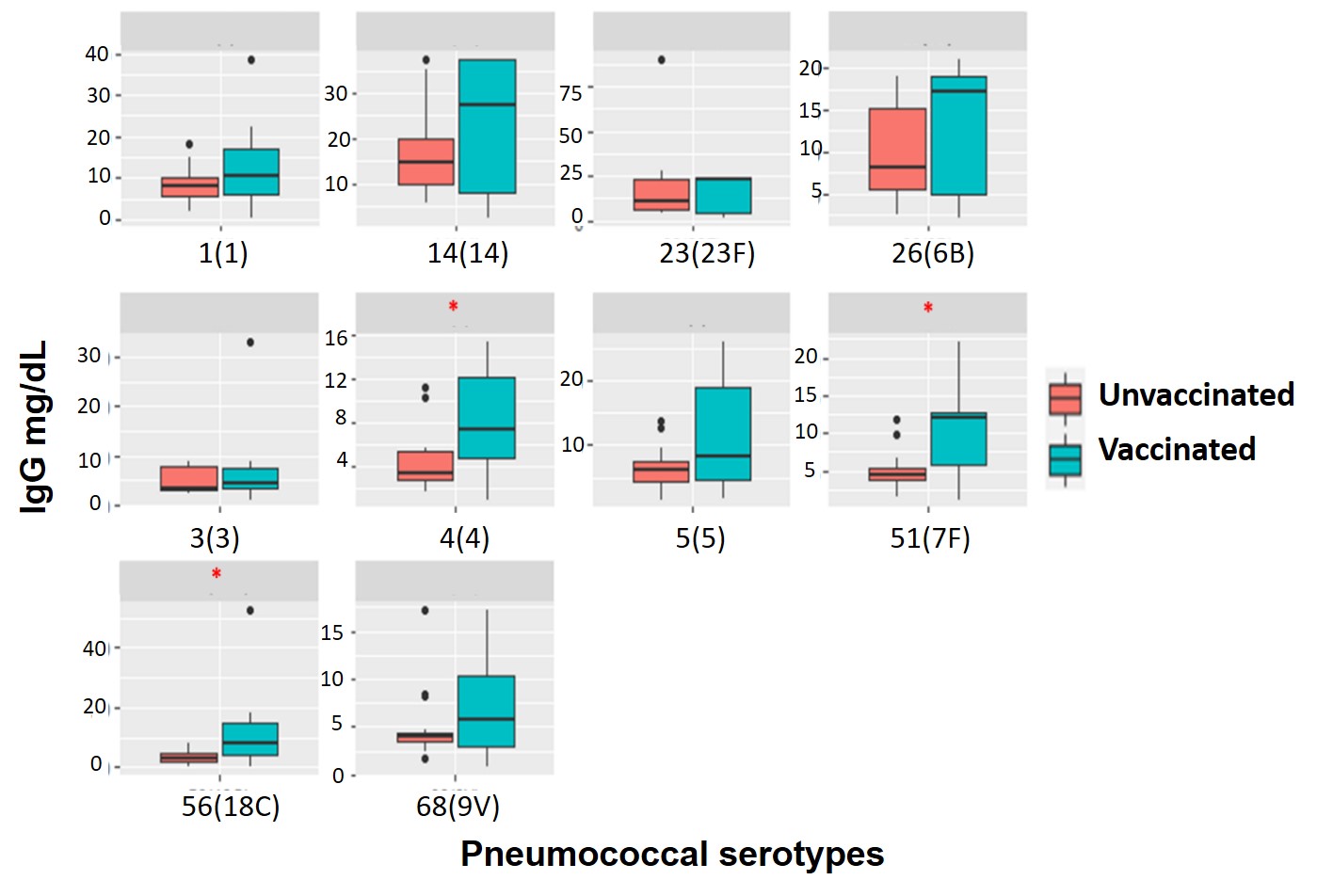
Thirty subjects with suspicious Specific Antibody Deficiency after Clinical Immunologist evaluation (v.g. pneumococcal invasive infection, unknown cause recurrent pneumonia) were evaluated using ELISA for IgG anti-pneumococcal antibodies 16 regardless of their immunization status (non-vaccination, polysaccharide or conjugated pneumococcal vaccines) using an ELISA validated for Colombia 17. Ten pneumococcal serotypes were selected, included in both conjugate and polysaccharide vaccines, serotypes were evaluated were: 1 (1), 3 (3), 4 (4), 5 (5), 14 (14), 23 (23F), 26 (6B), 51 (7F), 56 (18) and 68 (9V). Anti-pneumococcal IgG was considered positive with ≥1.3 µg/mL and protective when antibody titers were positive >70% of serotypes evaluated (7 out of 10) 8. Subjects with less than 70% of serotypes positive were vaccinated with polysaccharide vaccine (indicated by the primary healthcare provider).
Statistical analysis
The sociodemographic, clinical and paraclinical information of each case was extracted from the clinical records provided by each volunteer. Data analysis was performed with either SPSS version 25 and statistical packages in R version 3.5.2. Continuous and categorical variables were subjected to an exploratory analysis to determine central tendency, frequency, and variability measures. Due to the descriptive nature of this study, the presentation of the data is done in terms of frequencies. Pneumococcal antibodies were compared between vaccinated and unvaccinated and their statistical differences were evaluated with the Mann-Whitney U two-tailed test.
Results
One hundred ten volunteers with non-cystic fibrosis bronchiectasis or Recurrent pneumonia or non-cystic fibrosis bronchiectasis + Recurrent Pneumonia were enrolled. As a result, 8 volunteers (7.3%) were diagnosed with IgG hypogammaglobulinemia: Common Variable Immunodeficiency (6 cases), IgG hypogammaglobulinemia and X-Linked Lymphoproliferative Disease (one case each). The remaining 102 patients were eligible for evaluation, looking for predominantly antibody deficiency with normal IgG (Figure 1).
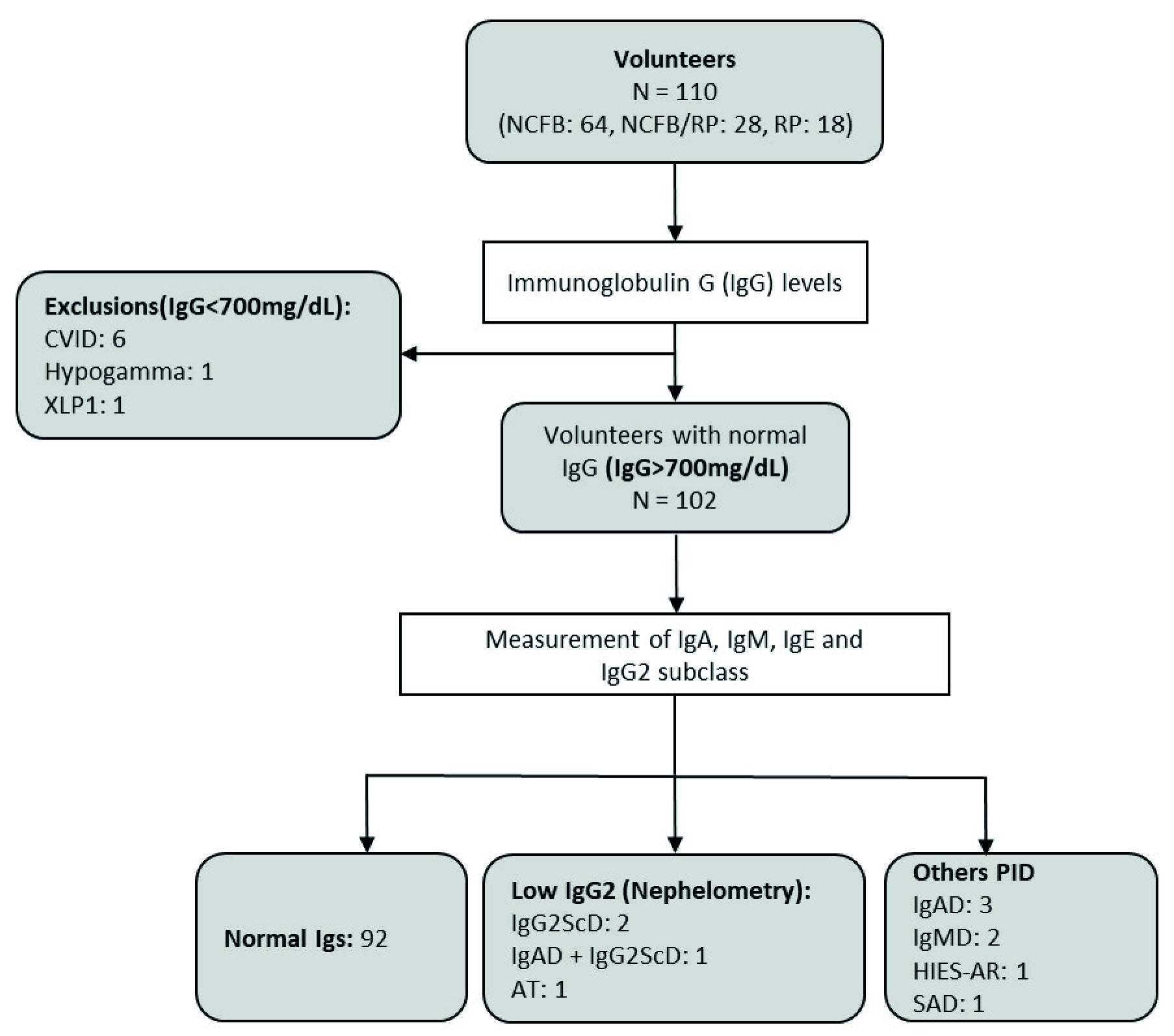
Figure 1 Patients Flowchart. non-cystic fibrosis bronchiectasis, non-cystic fibrosis bronchiectasis; RP, Recurrent Pneumonia; CVID, Common Variable Immunodeficiency; XLP1, X-Linked Lymphoproliferative Disease 1; Igs, Immunoglobulins; IgG2ScD, IgG2 Subclass Deficiency; IgAD, Selective IgA deficiency; IgMD, Selective IgM deficiency; AT, Ataxia Telangiectasia; HIES-AR, Hyper IgE Syndrome Autosomal Recessive; SAD, Specific Antibody Deficiency.
Demographic and clinical characteristics are shown in (Table 1). 102 patients, including 63 females and 39 males, median age of 48 years Interquartile Range (35 - 58), 64 cases (62.7%) were diagnosed with non-cystic fibrosis bronchiectasis, 23 (22.5%) cases with non-cystic fibrosis bronchiectasis plus recurrent pneumonia and 15 patients (14.7%) recurrent pneumonia. In addition, 60 cases (58.8%) had spirometry. Only 25 subjects (24.5%) have received pneumococcal vaccine over a lifetime.
Table 1 Sociodemographic characteristics of participants with non-cystic fibrosis bronchiectasis or recurrent pneumonia with normal IgG levels (N =102).
| Baseline Characteristics | |
|---|---|
| Age*, years | 48 (36-58) |
| Sex, female n (%) | 63 (61.7) |
| Non-cystic fibrosis bronchiectasis, n (%) | 64 (62.7) |
| Recurrent pneumonia, n (%) | 15 (14.7) |
| Non-cystic fibrosis bronchiectasis + recurrent pneumonia, n (%) | 23 (22.5) |
| P. pneumonia Vaccinated, n (%) | 25 (24.5) |
| Ethnicity | |
| Mestizo, n (%) | 73 (71.5) |
| Afro Colombian, n (%) | 24 (23.5) |
| Indigenous, n (%) | 4 (3.9) |
| Others, n (%) | 1 (0.9) |
| Scholarship | |
| Elementary, n (%) | 31 (30.3) |
| High school, n (%) | 49 (48) |
| University, n (%) | 14 (13.7) |
| None, n (%) | 8 (7.8) |
| BMI | |
| <18.5 | 17 (16.6) |
| ≥18.5 | 85 (83.3) |
| Sputum isolation | |
| P. aeruginosa | 7 (6.8) |
| No P. aeruginosa | 8 (7.8) |
| Negative | 27 (26.4) |
| No data | 60 (58.8) |
| Smoking status | |
| Never | 38 (37.2) |
| Former | 21 (20.5) |
| Current | 7 (6.8) |
| Biomass | 36 (35.2) |
| Spirometry n (%) | 60 (58.8) |
| BSI | |
| Mild | 45 (51.7) |
| Moderate | 23 (26.4) |
| Severe | 19 (21.8) |
* Age is expressed in median (years) and the interquartile range. Definition abbreviations; BMI, Body Mass Index; BSI, Bronchiectasis Severity Index.
Antibody deficiencies with normal IgG levels cases are shown in (Table 2). Selective IgA deficiency (3 cases) was the most frequent antibody deficiency with normal IgG followed by IgM deficiency (2 cases). IgG2 deficiency (2 cases) and IgG2+IgA deficiency were detected using ELISA. All cases with IgG2 <175 mg/dL were confirmed by nephelometry. IgG2 levels measured by ELISA were highly concordant with nephelometry (Figure 2).
Table 2 Predominantly Antibody Deficiencies with normal IgG levels
| Patient | Age (years) | Gender | IgG2 mg/dL ELISA | IgG2 mg/dL (nephelometry) | IgG mg/dL | IgA mg/dL | IgM mg/dL | IgE UI/mL | BQ | Type of deficiency |
|---|---|---|---|---|---|---|---|---|---|---|
| BQ007 | 34 | F | 154 | 124 | 1,030 | 3 | 97 | 0 | Yes | IgAD with IgG2ScD |
| BQ013 | 14 | F | 171 | 164 | 2,547 | 1342 | 103 | 331 | Yes | IgG2ScD |
| BQ023 | 29 | F | 122 | 77 | 1,028 | 113 | 65 | 473 | Yes | IgG2ScD |
| BQ054 | 14 | M | 100 | 112 | 1,163 | 7 | 144 | 2 | No | AT |
| BQ021 | 63 | F | 1,232 | 440 | 1,519 | 3 | 82 | 45 | Yes | IgAD |
| BQ072 | 50 | F | 412 | 304 | 1,946 | 10 | 72 | 29 | Yes | IgAD |
| BQ087 | 48 | M | 350 | 323 | 1,394 | 3 | 60 | 0 | No | IgAD |
| BQ090 | 64 | F | 228 | 245 | 1,207 | 406 | 25 | 44 | No | IgMD |
| BQ107 | 64 | F | 243 | 316 | 1,022 | 407 | 26 | 448 | Yes | IgMD |
| BQ052 | 19 | M | 365 | ND | 2,626 | 102 | 34 | 2,001 | Yes | HIES-AR |
| BQ102 | 14 | M | 323 | ND | 1,398 | 211 | 179 | 45 | No | SAD |
BQ: Bronchiectasis; IgG2ScD: IgG2 Subclass Deficiency; IgAD: Selective IgA deficiency; IgMD: Selective IgM deficiency; AT: Ataxia Telangiectasia; HIES-AR: Hyper IgE Syndrome Autosomal Recessive; SAD: Specific Antibody Deficiency; ND: No Data.
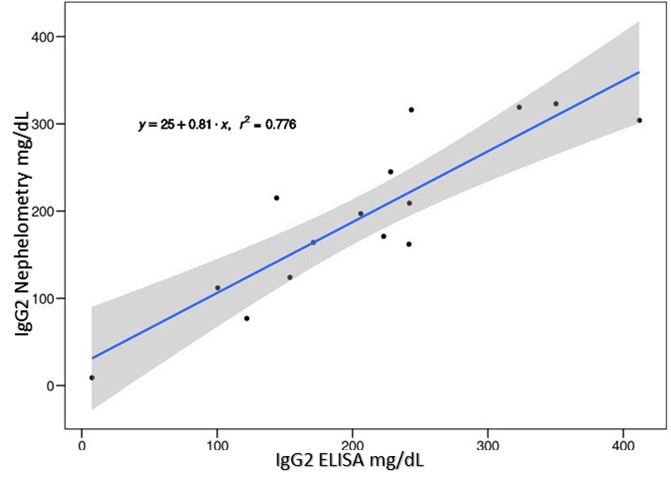
Figure 2 IgG2 levels ELISA vs Nephelometry. Correlation analysis r2 = 0.776. Non-parametric test, Kendall tau.
Two participants (BQ054 and BQ102) had less than 70% of protective anti-pneumococcal antibodies (Table 1S ); both were vaccinated with pneumococcal polysaccharide vaccine without changes in IgG anti-pneumococcal titers (data not shown). BQ054 was finally diagnosed with ataxia-telangiectasia, and BQ102 fulfilled the criteria for Specific Antibody Deficiency 7.
Anti-pneumococcal antibodies according with vaccination status are shown in Figure 3. Interestingly only 3 serotypes exhibited statistically significant difference between vaccinated and unvaccinated subjects: serotypes 4 (p= 0.0282), serotype 7F (p= 0.0357) and serotype 18C (p= 0.0437), unfortunately time from vaccination was not possible to determine.
Discussion
Predominantly antibody deficiencies are frequent in Colombian patients with non-cystic fibrosis bronchiectasis or recurrent pneumonia. We identified 19 cases (17.2%) of Inborn Errors of Immunity. IgG measurement alone was able to identify 8 cases highlighting the importance of serum absolute immunoglobulin evaluation. Our finding is consistent with previous reports of primary immunodeficiency frequency in this conditions (ranging from 2% to 17%) 18-20. In our study, Common Variable Immunodeficiency frequency is slightly higher than reported in other series with recurrent upper/lower respiratory infections 14,21. Therefore, IgG measurement should be mandatory in all patients with bronchiectasis as a frontline test, and it is a standard that has been previously suggested 22.
IgG subclass and complete immunoglobulin workup (IgG, IgA, IgM and IgE determination) are useful strategies to approach the etiology of bronchiectasis and recurrent pneumonia in adults. Serum IgA, IgM and IgE evaluation allow us to identify other Predominantly antibody deficiencies with normal IgG levels previously unrecognized in these patients. Measurement of IgG subclass is a strategy that has been implemented in the evaluation of patients with a history of chronic infections 23. IgG subclasses evaluation has been suggested as a second-line test in patients with recurrent infections 24 and first line in IgA deficiency cases 25. IgG2 subclass measurement by ELISA allowed the diagnosis of four IgG2 deficiencies, two of them with IgA deficiency one patient is currently receiving replacement therapy with human IgG and the other has ataxia-telangiectasia.
IgG2 quantification using ELISA is a useful approach in settings without nephelometry quantification availability. A good correlation of serum IgG2 levels was observed between ELISA and nephelometry, as had been reported previously by Adebajo et al. 26, and Aazami et al 27. ELISA technique is widely used, versatile and sensitive but requires longer processing time than nephelometry. Therefore, ELISA may be a valuable alternative to nephelometry (if this one is not available) to detect samples with low IgG2 levels. However, when it is possible and available, all IgG subclasses should be measured. We recommend carrying out immunoglobulin levels together with IgG subclasses as a first-line approach.
Adults exhibit high IgG anti-pneumococcal titles regardless of their immunization status. IgG antibodies against 10 Pneumococcal serotypes were evaluated regardless of their immunization status (vaccinated or unvaccinated). Interestingly all subjects evaluated (except two cases) exhibited positive and protective IgG anti-pneumococcal antibodies despite low vaccination rates, suggesting a high rate of pneumococcal infection during life. We found that 3 out of 10 serotypes evaluated: 4 (4), 51 (7F) and 56 (18C) presented significantly higher antibody titers in the vaccinated individuals with no statistical difference in the other serotypes evaluated. Our findings suggest high pneumococcal exposure that correlates with the most prevalent serotypes in Colombia 28. Therefore, improve the pneumococcal vaccination rate is mandatory for Colombian patients with bronchiectasis or recurrent pneumonia.
More studies require specific antibody deficiency frequency in adults, including specific IgG evaluation pre- and post-vaccination. However, our study is just an approach that shows a high frequency of positive antibody responses. To the best of our knowledge, this is the first systematic humoral response evaluation in a Colombian population with respiratory complications as bronchiectasis or recurrent pneumonia.
Conclusions
Our results show that Inborn immunity errors, especially predominantly antibody deficiencies with normal IgG serum levels as IgG2 deficiency, IgA deficiency, IgM deficiency or hyper IgE, should be considered an underlying cause of bronchiectasis and recurrent pneumonia in Colombian adults.











 text in
text in