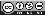1. Introduction
Access to water with sanitary conditions for human consumption is essential for the health of consumers, and their final characteristics will depend on its origin (groundwater or surface water) and the potabilization treatment. Coagulation is a physical-chemical process to reduce the repulsive potential of the electric double layer of colloids using coagulants as metallic, polyelectrolytes and polymers. As a result, colloidal particles begin to develop and then agglomerate into larger particles or flocs (Sillanpàà et al., 2018). The potabilization process consists of treating water for human consumption, which implies low costs, with easily operable installations. Generally, the system has a desander, flocculator, flocculator-sedimentador, filters and storage reserves (Pantoja-Espinosa et al., 2015).
There are different types of coagulants such as metallic salts, natural coagulants from plants rich in polysaccharides, proteins (Shamsnejati et al., 2015) and polyelectrolytes (Donato et al., 2006) (Salehizadeh et al., 2018). Normally, polymeric flocculants and inorganic coagulants have been used; these are are expensive, and generally consist of metal salts of synthetic origin, which affects the bodies of water due sludges are arranged without prior treatment (Kamar, Abdul Aziz and Ramli, et al., 2015). Despite, its effectiveness it has been found large amounts of inorganic ions on effluents, which fall the shelf life-time of the equipment owing to corrosion, fouling and clogging (Gao et al., 2009).
In this sense, starch is a natural coagulant alternative to minimise the harmful impacts caused by traditional coagulants. Starch has a low cost, renewable nature and environmentally friendly (Choy, Prasad and Wu et al., 2016). Gelatinised starch increases the viscosity of aqueous solutions, and it promotes the elimination of turbidity (Zhu, 2015).
Starch and its derivatives have been shown to be very useful for flocculation of ultrafine mineral particles and iron metals (Trujillo et al., 2014). Several researchers have used natural coagulants from alum and rice starch (Teh et al., 2014), calcium alginate (Arcila y Peralta, 2016), modified starches (López-Vidal et al., 2014), starch with alum and poly-aluminium chloride (Choy, Prasad and Wu et al., 2016). They found that the use of coagulants of natural origin is a viable alternative for their use in the clarification of surface waters, which represents a sustainable option for water treatment managers as opposed to inorganic coagulants. Therefore, the objective of the present study was to use plantain starch as a natural coagulant, in order to remove turbidity and colour present in a water sample by varying the agitation rate and concentration of the coagulant.
2. Materials and Methods
2.1. Experimental design
The present investigation used an experimental design of response-surface of central composite type. Two independent variables were evaluated (agitation rate (rpm) and coagulant concentration (mg/L)) with two variation levels (30 and 40 rpm). The dependent variables were pH, colour (Platinum-Cobalt Scale [Pt/Co]) and turbidity (Nephelometric turbidity units [NTU]), and the intervening variables were centrifugation time (min), centrifugation rate (1500 rpm), cooling time (20 h) and sample concentration (0.5 Kg/L). During the experimentation, Merck brand analytical grade reagents were used.
2.2. Starch extraction
The starch extraction was done using 1000 g of plantain, it were peeled and sliced for subsequent washing with distilled water to eliminate impurities. Then 500 g were immersed in a solution of 0.25% NaOH by weight and 500 g in water; both mixtures were cooled to 4 °C for 20 h, and then liquefied for 1min. After the maceration process, starch-rich products were obtained and filtered separately. The filtrates were centrifuged at 1500 rpm for 10 min. The centrifuged material was adjusted to neutral pH using a 2M HCl solution and centrifuged again at the same conditions. Finally, the starch was dried and ground (Maniglia and Tapia, 2016).
2.3. Turbid water preparation
The synthetic turbid water was elaborated by dispersion of 0.3 g of bentonite in 800 mL of deionised water. This solution was mixed for 1 h at 200 rpm, and subsequently was left 21 h at rest for a hydration process. Then, it was diluted in 1.9 L of deionised water, and resulting dispersion has a turbidity of 32.3 NTU (Canepa et al., 2004; López-Vidal et al., 2014).
2.4. Turbidity removal test
The tests to remove turbidity and colour were performed by putting in contact the turbid water sample with the starch extracted in the jar test equipment at 200 rpm for 1-2 min. The agitation rate was reduced to 20-40 rpm for 15 min, the mixture was left to repose, and an aliquot was taken to measure turbidity (NTU), colour (Pt/Co) and pH of the solution.
3. Results and Discussion
3.1. Turbidity Removal Tests
Two samples of starch were obtained from the extraction using NaOH and water. The dry weight of the samples was 38 and 35.5 g respectively. Subsequently, jar tests were performed, and the effect of the coagulant dose and agitation rate on the pH of the raw water samples was evaluated.
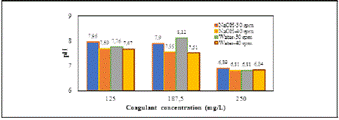
The pH was inversely proportional to the concentration of the coagulant, and the agitation rate does not significantly affect the pH of the samples (Figure 1).
It can be seen that the samples treated at 40 rpm reveal a similar tendency in pH from the beginning (7.67-7.69) until the end (6.84-6.81), for the coagulant extracted with water and NaOH, respectively. On the other hand, the coagulant extracted with water at 30 rpm had a pH from 7.76 to 6.81. At last, the coagulant extracted with NaOH at 30 rpm has the highest initial pH (7.96), and their final pH was similar to the others (6.89). The pH is a main parameter in coagulation-flocculation process, due it can change the surface charge of the coagulant and/or contaminant; then, the starch obtained in the present study and used as natural coagulant is stable over a wide pH range and its use in real waters samples is recomended (Daverey, Tiwari and Dutta et al., 2018; Paredes et al., 2018).
Regarding the colour reduction of plantain starches, the colour value was 10 on the Platinum-Cobalt scale (Table 1) for all the samples. Plantain starch did not affect the colour of water solutions. This result can be considered positive. Starch does not add colour to the raw water sample after the test. These results are within the pH range of 6.0-8.5 established by Shamsnejati et al., (2015) where the effected of pH on colour is not significant for the removal of dye from a textile wastewater model.
TABLE 1 TESTS OF COLOUR ON THE PLATINUM-COBALT SCALE
| Starch | Agitation (rpm) | Coagulant concentration (mg/L) | ||
| 125 | 187.5 | 250 | ||
| Plantain NaOH | 30 | 10 | 10 | 10 |
| 40 | 10 | 10 | 10 | |
| Plantain H2O | 30 | 10 | 10 | 10 |
| 40 | 10 | 10 | 10 | |
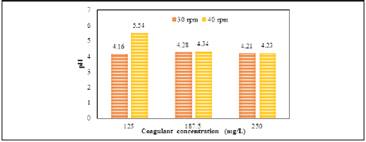
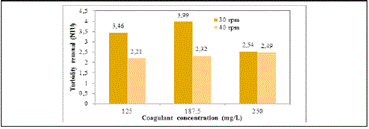
The behaviour of turbidity concerning the coagulant dose and agitation rate during the jar test using plantain starch extracted with NaOH and water ( Figure 2) were evaluated.
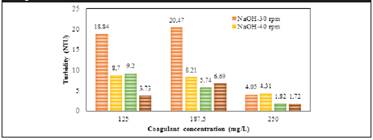
Figure 2 Effect of the agitation rate and concentration of coagulant on the removal of turbidity using plantain starch as coagulant
The coagulant concentration has a significantly positive effect on the removal of turbidity. Starch extracted with water (30 and 40 rpm) exhibits the best results. Turbidity was reduced more effectively, reaching a removal rate of 94.6% using 250 mg/L coagulant concentration and 40 rpm. This behaviour can be explained by the presence of amylopectin in the starch structure due to its electrolytic nature, which is a branched distribution of the proteins that compose it, and would trap in its structure, as was reported in literature (Trujillo et al., 2014).
Al2(S04)3 was used as a synthetic coagulant to compare with the plantain starch extracted with NaOH and water. Figure 4 shows the results obtained from the turbidity measurement of raw water samples after a jug test using aluminium sulphate (Al2(S04)3) as a coagulant.
Comparing the obtained results ( Figure 1,Figure 3 and Figure 4), the residual pH values obtained with the natural coagulants extracted from the plantain were higher than those obtained with the synthetic coagulant. These results are like those reported by Ganjidoust et al., (1997) , a natural coagulant (chitosan) had the better effect than synthetic coagulants (HE and PEL) eliminating up to 90% of the colour and 70% of organic carbon. This phenomenon can be explained due to the high rate of formation of flocs of excellent characteristics during the process, by the electrolytic nature of the natural coagulant tested, which contributes to the appropriate and rapid sedimentation by the consistency and weight of the flocculated particles (Paredes et al., 2018).
Table 2 shows the colour values on Platinum-Cobalt scale using Al2(S04)3 as a coagulant. In all treatment at different concentrations and agitation rates, the colour was constant (10 units). Same results were obtained with the starch. According to Colombian regulation for drinking water, obtained results are admissible because the colour should be lesser than 15 units when a coagulant dosage of 250 mg/L is used (Resolution 2115, 2007).
3.2. Statistical analysis
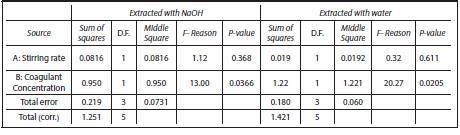
Statistical analysis was performed using an AN0VA analysis of variance, as shown in Table 3, which indicates that the coagulant concentration has a confidence level of 95.0 being significantly influential on the process.
For the analysis of pH variance, it was obtained that the concentration of coagulants extracted with water and NaOH had a P-value lower than 0.05. This fact indicates that this variable affects the pH, on the contrary to the agitation rate (P-value higher than 0.05) in both forms of extraction. The Durbin-Watson statistic (DW) showed that there was no serial autocorrelation since the P-value was higher than 0.05.
Table 4 shows the AN0VA analysis for the percentage of turbidity removal, finding that none of the effects has a P-value less than 0.05 indicating that these variables and their correlations are not statistically incident on the process.
As for the variance of the turbidity percentage, the two variables (agitation rate and coagulant concentration) and the extraction with NaOH had a P-value greater than 0.05. It indicates that none of these effects has an incidence on the turbidity percentage. On the other hand, the agitation rate obtained a P-value lesser than 0.05 for the coagulant extracted with water. The Durbin-Watson statistic (DW) proved that there was no serial autocorrelation since the P-value was higher than 0.05.
4. Conclusion
The plantain starch tested did not contribute odour, flavour and colour to the raw water, it was also highlighted because the pH was maintained in the acceptable range so that the coagulation-flocculation process was given satisfactorily. Under the conditions established when performing the jar test, the percentages of turbidity removal most of the cases were higher than 70%, using doses of coagulants similar to conventional synthetic coagulants. Starch extracted with water being the most efficient for the removal of turbidity, the variable with the highest linear influence being the combination of agitation rate and concentration of coagulant, it is showing better behaviour regarding turbidity removal than the commercial aluminium sulphate coagulant