Introduction
Helicobacter pylori is accepted to be the causative agent of acute and chronic gastritis, and a major predisposing factor for peptic ulcer disease, gastric carcinoma, gastric lymphoma and mucosa-associated lymphoid tissue (MALT) 1,2. This microorganism is a spiral-shaped Gram-negative bacterium endowed with a very powerful urease activity and with polar flagella. Urease activity buffers the pH at the cell surface, allowing bacterial survival in the stomach lumen until it enters the mucus layer by means of their flagella, which is a protective barrier against the high proton concentration 3. To colonize the host stomach, H. pylori expresses several virulence factors that can play a role in pathogenesis, being the most important the vacuolating VacA, a cytotoxin associated with gene A (CagA), the neutrophil-activating protein (NAP), and the urease activity 4.
Peptic ulcer disease and other H. pylori-associated disorders usually regress or even heal completely after treatment with antimicrobials. The most effective and best tolerated combinations consist of a triple therapy which includes bismuth salts or proton pump inhibitors with two antibiotics (clarithromycin and amoxicillin, clarithromycin and metronidazole, or amoxicillin and metronidazole), and a quadruple therapy, including a proton pump inhibitor plus a bismuth salt and the antibiotics metronidazole and tetracycline 5,6. However, the available antimicrobial therapies for H. pylori infection have several shortcomings: (i) limited efficacy in vivo of the antimicrobials due to both inability of drugs to achieve appropriate levels in the gastric mucus layer, and/or inactivation of drugs at low pH 7; and (ii) the development of antimicrobial resistance for H. pylori8-10. Consequently, an optimal therapy against H. pylori infection is not available yet, therefore, novel approaches are needed, including vaccine development 11, use of antimicrobial peptides 12, and utilization of naturally occurring compounds with antimicrobial activity, such as porphyrins, essential oils and plant polyphenols 13-15.
Several epidemiological studies have demonstrated that the modest consumption of alcoholic drinks may influence spontaneous eradication of H. pylori16,17. Alcoholic beverages, mainly wine, have a strong antimicrobial activity, as has been shown both in vitro and in vivo18,19; however, other conditions depending on the beverage type, dose, host physiological conditions, to name a few, affect to the H. pylori mechanisms of action and may explain the inconsistent results obtained by several authors 19,20. Marimon et al 21 carried out an in vitro analysis of the bactericidal action of red wine, HCl solution (pH 3.5), a solution containing 12% ethanol, and 12% ethanol at pH 3.5 on H. pylori. Red wine exerted a higher bactericidal effect upon H. pylori than that other observed for the other solutions. However, no explanation was found for the mechanisms by which wine exerts an in vitro bactericidal effect independently of its alcohol concentration, nor have the substances responsible been identified to date.
In the present study, the antibacterial effect of different wines from the Douro region (Iberian Peninsule) and their main components (proanthocyanidins, pH, alcohol content and four antioxidant compounds) on H. pylori strains (isolated from gastric biopsies) has been evaluated. In addition, the potential interactions between amoxicillin or metronidazole and the antioxidant components of the wine have also been evaluated.
Materials and methods
Bacterial strains, culture and identification
Eleven clinical strains of H. pylori isolated from gastric biopsies and a reference strain (CCUG 15813) from the Culture Collection of the University of Gothenburg (Sweden) have been used in this study. Strains were subcultured on Columbia Agar (Oxoid, Hampshire, UK) supplemented with 10% of defibrinated horse blood (Oxoid) (CAB) and in CAB with Dent's supplement (SR 147, Oxoid). Subcultures were incubated at 36 ± 1°C under microaerophilic conditions using jars with Gas generating kits for Campylobacter (Oxoid) for 48-72 h 22. All isolates were identified as H. pylori using the phenotypic and biochemical tests described previously 23. The urease activity was performed using Urea Broth Medium (Oxoid) with the SR20 20K supplement (Oxoid).
Antibiotic susceptibility assays and PCR-based genotyping
Disk diffusion testing was performed by standard CLSI methods 24 using CBA for the evaluation in vitro of the antibiotic susceptibility of H. pylori strains against the following antimicrobials, used frequently for treatment of H. pylori infection, supplied by Oxoid: metronidazole, clarithromycin, erythromycin, amoxicillin, ciprofloxacin, moxifloxacin, levofloxacin, rifabutin, furazolidone, and tetracycline. Inocula were prepared from two agar plates of a 2-day fresh growth on CBA, which were scraped and suspended in 5 ml of Columbia broth (CB) (Oxoid) to achieve the turbidity equivalent to a McFarland standard of 3 to 4. Serial dilutions of the bacterial suspensions were performed to yield a colony count of about 4 x 108 CFU/mL. After incubation period (48-72 h) at 36 ± 1°C under a microaerophilic atmosphere, inhibition zone diameters (in mm) were read with the aid of callipers. Bacterial isolates were classified as resistant, sensitive or intermediate according to the National Committee for Clinical Laboratory Standards breakpoints 25. Genomic DNA of H. pylori isolates was extracted as described by Oleastro et al 26. Genotyping of the virulence factors vacuolating gene (vacA) and cytotoxin-associated gene (cagA) was performed by PCR in accordance to protocols and primers described previously 27,28.
Determination of the Minimal Inhibitory Concentrations (MICs)
E-test strips supplied by AB Biodisk (Solna, Sweden) were used to determine the MICs of amoxicillin and metronidazole. CBA plates were allowed to dry for 10 min and the strips were kept on the bench for 1015 min to attain room temperature. Colonies from pure cultures of H. pylori were transferred using a sterile loop into a sterile tube containing 4.5 ml of CB and adjusted to standard 2 of the MacFarland scale. The solution was flooded over the plate (CBA) and excess fluid was removed with a pipette. After drying for 15-20 min, an E-test strip was placed on each plate and then incubated under microaerophilic conditions at 36 ± 1°C for 48-72 h. Results from E-test were interpreted by recording the point of intersection of the growth elliptic margin zone with the MIC scale value on the E-test strip. MIC breakpoints of >2 μg/ml and >8 μg/ml were used for amoxicillin and metronidazole, respectively.
Susceptibility assays of the wines and their components
Seven different wines of Douro region, 5 reds and 2 whites, were used to determine the susceptibility of H. pylori strains. Four red wines were product of monocastes each one named Touriga Nacional, Touriga Brasileira, Tinta Roiz, and Tinta Barroca. The fifth one was a mixture of castes Negra mole, Castelao and Trincadeira. The wines were previously filtered through membrane filters of 0.45-μm pore-size (Millipore Co., Belford, MA, USA), and several concentrations of each filtered wine were added to culture media in aseptic conditions. Four different assay media were prepared; all of them were composed of 39 g/L of CAB and variable percentages of filtered wine (9% for medium 1, 22.5% for medium 2, 45% for medium 3, and 67.5% for medium 4).
To control the effect of alcohol graduation in the growth of H. pylori, several compositions of CAB supplemented with ethanol were performed. The final proportion of alcohol in media tested were 12.69° (v/v), 13° (v/v), 14.55° (v/v) and 14.8° (v/v). In addition, H. pylori was grown in CAB plates at low pH values (from 4.49 to 4.89) by adding of lactic acid (10%).
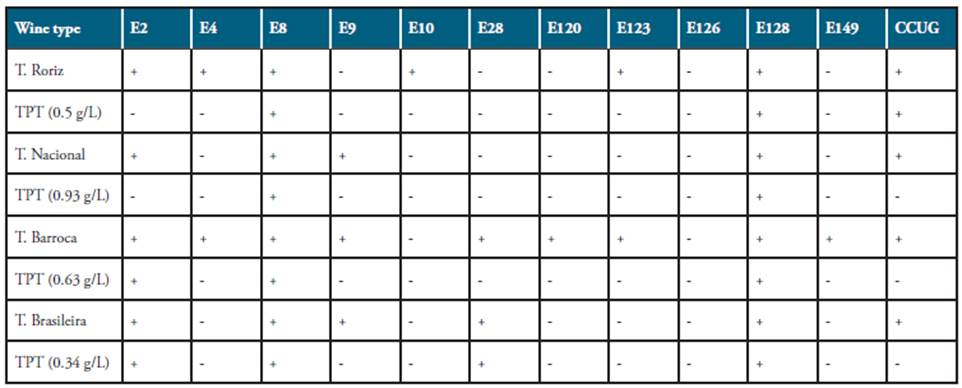
Proanthocyanidins were isolated from several red wine grapes originating from the Douro region. The pips were carefully removed from 150 frozen grapes and then lyophilized. The dry extract was stored at -20°C until use. Proanthocyanidins of the extract were separated and identified by HPLC following the protocol described by Roggero et al 29. The detection of proanthocyanidins was performed by the monitorization of the absorbance detected at 535 nm in a DAD (Merck, Madrid, Spain). The proanthocyanidin concentrations were calculated attending to the calibration line obtained by the use of maldivin as pattern. Different concentrations of proanthocyanidins in distilled water according to the values of HPLC of the tested wines (0.93 g/L for Touriga Nacional, 0.63 g/L for Tinta Barroca, 0.50 g/L for Tinta Roriz, and 0.34 g/L for Touriga Brasileira) were added to the CAB to achieve a concentration of 67.5% (v/v). Inoculated plates were incubated at 36 ± 1°C under a microaerophilic atmosphere for 72 h.
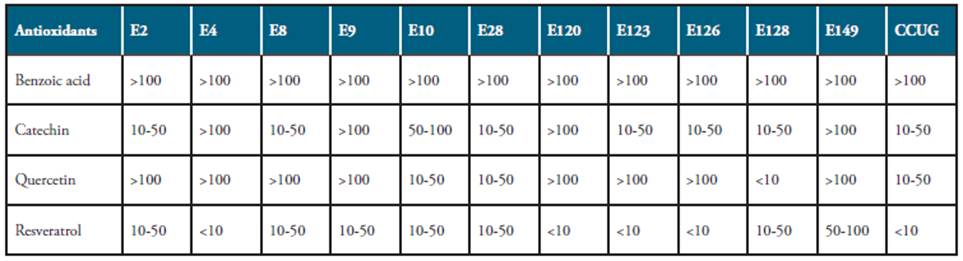
Considering the total phenol index as the equivalent to gallic acid units calculated by the Folin-Cicocalten's method. The total phenolic compounds of the wines were determined by the absorbance values at 725 nm of different concentrations of gallic acid. The agar dilution method 30 was used to determine the MICs of the antioxidant compounds present in the wines. The antioxidants used were benzoic acid (Sigma-Aldrich Química, S.A., Madrid, Spain), catechin (Sigma-Aldrich), quercetin (Sigma-Aldrich) and resveratrol (Sigma-Aldrich), and they were prepared in deionised water. Media consisted of CBA with the antioxidant compounds tested at concentrations of 0.01 to 0.1 mg/mL in doubling dilutions. Inocula of 10 μL of bacterial suspension of each H. pylori strain (about 4 x 108 CFU/mL) were applied on the surface of the growth medium. Plates were incubated at 36 ± 1°C under a microaerophilic atmosphere for 72 h. The lowest concentration of antioxidant compound showing no growth was recorded as the MIC. All the susceptibility assays were three-times replicated.
To determine the possible interaction between amoxicillin and/or metronidazole and the wine antioxidants, 10 μg of each antioxidant agent tested was added to each aqueous antibiotic solution containing amoxicillin (25 μg) or metronidazole (5 μg). Later, the antibiotic solutions supplemented with the antioxidants were tested according to the broth microdilution method 25. A t-student test was applied to determine the statistical differences of the susceptibility of the bacterial strains to the antimicrobials and to the interactions anti-microbial-antioxidant agents.
Results
Characterization of the H. pylori strains
All the isolates collected from endoscopic biopsies were confirmed as H. pylori according to 23 phenotypic features tested specified by Megrauf and Lee 23 (data not shown). The molecular characterization of the H. pylori isolates using a PCR-based method allowed the discrimination of the isolates in three groups on the basis of the presence/absence of the virulence factors CagA and VacA (Table 1). Group 1 (Cag A +/Vac A s1/m1, urease +) composed to 7 strains; group 2 (Cag A +/Vac A s1/m2, urease +), 3 strains; group 3 (Cag A -/Vac A s2/m2, urease +), 2 strains. Overall, ten strains (83.3%) presented the cytotoxin CagA, whereas all the strains showed the vacuolating factor VacA and the urease activity (Table 1). Antibiotic susceptibility of H. pylori showed that all the strains were susceptible to 10 antimicrobials tested, but only 4 strains were resistant to the concentration tested (50 μg) of metronidazole (Table 1).
Table 1 Distribution of Helicobacter pylori genotypes and antibiotic susceptibility using the disk diffusion method.
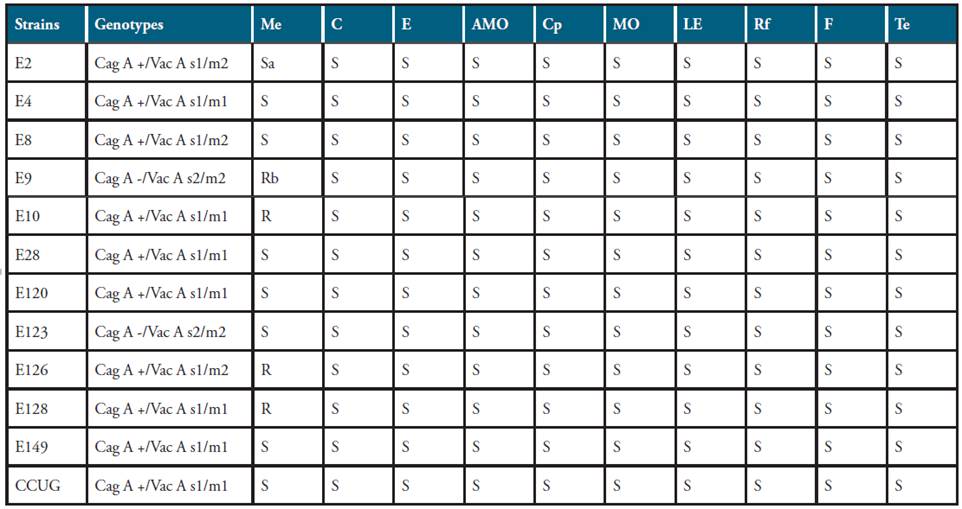
Me: metronidazole; C: clarithromycin; E: erythromycin; AMO: amoxicllin; Cp: ciprofloxacin; MO: moxifloxacin; LE: levofloxacin; Rf: rifatubin; F: furazolide; Te: tetracycline. aS: sensitivity; bR: resistance
Source: Authors.
Effect of the types and concentrations of wine, pH, alcohol concentration and proanthocyanidins on H. pylori strains
All strains tested showed growth in medium 1 composed of CAB supplemented with 9% of all the wines assayed. On the contrary, none of the strains grew in media 3 and 4 (45% and 67.5% of wine, respectively). In the case of the medium 2, variable results were obtained depending on the type of wine tested, for example, only 7 strains (E2, E4, E8, E28, E123, E126 and E149) showed growth at 22.5% of Siria white wine, and 3 strains (E120, E128 and CCUG 15813) grew at 22.5% of Negra-Castalao red wine.
All H. pylori strains tested grew in media without wine at pH values ranging 4.49 to 4.89. Similarly, all the strains showed an optimal growth at the ethanol concentrations tested (12.69°, 13°, 14.55° and 14.8° v/v).
Similarly, all the H. pylori strains showed growth in CAB supplemented with the concentration of proanthocyanidins present in the different types of wines (Table 2). Urease test was used to study the changes of H. pylori strains after growth with wine (9%) and proanthocyanidins (Table 2). Only the strain E126 loss the urease activity after grown in CAB supplemented with wine or proanthocyanidins; on the contrary, E8 and E128 strains maintained their urease activity for all the conditions assayed. An inverse but significant relationship was obtained between the response of the urease activity and the total phenol index of the red wine tested [y = -48 x + 33, r2 = 0.914, p < 0.001, where y is the absorbance at 750 nm and x is the concentration (mg/mL) of gallic acid].
Effect of antioxidant agents on H. pylori growth
The inhibitory activity exerted by the antioxidant compounds tested on the H pylori growth depending both of the strain and the concentration of the antioxidants. The benzoic acid did not demonstrate to possess any effect on the tested strains at any assayed concentrations. On the contrary, the resveratrol showed the highest growth inhibition rates for all the tested strains, even at the lower concentration assayed (0.1 mg/mL), and 91.7% of the strains possessed a (MIC) lower than 50 μg (Table 3). On the other hand, only 33.3% of the tested strains presented a MIC lower than 50 μg for quercetin, and 58.3% of them have MIC lower than 50 μg for catechin (Table 3).
Interaction between antimicrobial agents and antioxidants tested
Amoxicillin and metronidazole were choose to perform the interaction effects with antioxidant compounds on the basis of their susceptibility patterns on H. pylori strains tested (Table 1). Table 4 presents the results of potentiation or antagonism that produced the combination of antimicrobials and the antioxidant agents. The results obtained were very variable, depending both of the combination and the strains tested.
Table 4 Interactions between antimicrobials and wine antioxidants on the susceptibility of H. pylori strains.
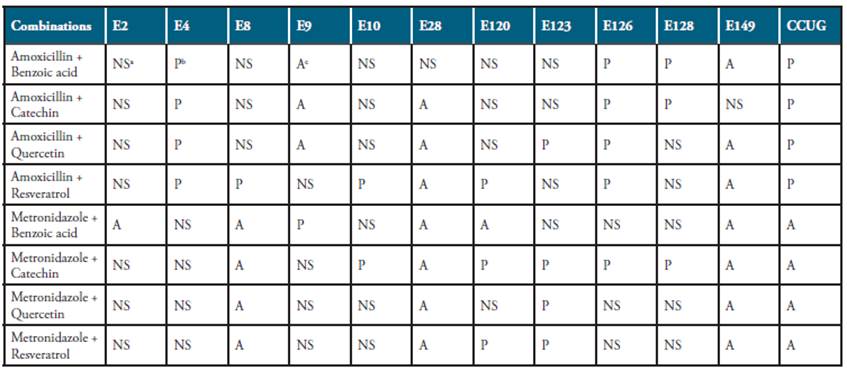
aNS: No significant effect; bP: Potentiation (significant at p<0.05 level, t-student test); cA: Antagonism (significant at p<0.05 level, t-student test)
Source: Authors.
In the case of amoxicillin, resveratrol was the antioxidant that provoked a higher number of potentiation of the antimicrobial agent, since 50% of the H. pylori strains (E4, E8, E10, E120, E126 and CCUG) increased their susceptibility with the combination antimicrobial-antioxidant. However, two strains (E28 and E149) decrease their sensitivity with the combination compared to the effect of the antimicrobial alone. For metronidazole, the antibiotic-resistant strains E9, E10, E126 and E128 showed susceptibility to the antibiotic supplemented with catechin or benzoic acid. For the other strains tested, the combination metronidazole and antioxidant compounds only provoke potentiation effect in strains E120 and E123 with the combinations metronidazole-catechin, metronidazole-quercetin and metronidazole-resveratrol. On the contrary, in the strains E8, E28, E149 and CCUG an antagonism effect was recorded with all the combinations tested (Table 4).
Discussion
The frequent used of a limited number of antibiotics for anti-H. pylori therapy has resulted in the development of resistance mechanisms in H. pylori strains 1,8,31. Therefore, it is necessary to study other approaches to control the H. pylori-related chronic inflammatory processes and mediators responsible for carcinogenesis. Phytoceuticals is a term for plant products that are active on biological systems. Some of them, present in red ginseng, green tea, or red wine, are known to inhibit H. pylori colonization, decrease gastric inflammation by inhibiting cytokine and chemokine release, and repress precancerous changes by inhibiting nuclear factor-kappa B DNA binding, inducing profuse levels of apoptosis and inhibiting mutagenesis 31,32.
The results obtained in the present study show that red wines from the Douro region, at concentration of 22.5%, inhibited the in vitro growth of all the H. pylori strains tested. However, the two white wines tested allowed the growth of some H. pylori strains at this concentration (22.5%) (data not shown). The inhibitory effect of wines on the growth of several enteropathogens, including to H. pylori, has been previously described 33-35. Marimon et al 21 demonstrated that red wine exerted a marked bactericidal effect upon H. pylori, and this effect could not be attributed to the acid pH of wine or at its alcohol concentration. Similar results have been obtained in our study, since none of the strains tested were inhibited by the four ethanol concentration tested (from 12.69° to 14.8° v/v) and for the pH range (from 4.49 to 4.89). Our results suggested that the viability of the H. pylori strains grown in wine might depend on the antioxidant and other phenolic components of the wine. A relationship between the phenotypic characteristics and virulence factors of the strains might be established with the susceptibility to 22.5% of wine. However, no convincing explanation has yet been found for the mechanism by which wine exerts and in vitro bactericidal effect independently of its alcohol concentration. In vivo mechanisms that could contribute to the protective effect of wine might be the rise in acid secretion, and the increase in intestinal motility 18.
Polyphenols are plant secondary metabolites, which have. Wine, particularly red, contains a variety of polyphenols derived from the skin of the grape, including flavonols (quercetin), stylbenes (resveratrol), flavanols (catechin), gallic acid, condensed tannins (catechin polymers), and polymeric anthocyanins 36. These polyphenolic compounds potential health benefits on the human organism, mainly as antioxidants, anti-allergics, anti-inflammatories, anticarcinogenics, decrease platelet aggregation and endothelial adhesion, and decrease the level of high-density lipoprotein cholesterol 37-39. Several authors have demonstrated that the main active phenolic compound present in the red wine against H. pylori is the resveratrol 33,40,41. Resveratrol exhibits a number of biological activities, including anti-inflammatory, antioxidant, platelet antiaggregatory and anticarcinogenic properties, and modulation of lipoprotein metabolism 42. The results obtained in the present study for resveratrol assays showed higher inhibitory effects on H. pylori strains tested that those obtained for the other phenolic compounds tested, catechin and quercetin (Table 3). Similar results have been reported by Mahady et al 43, who obtained a MIC of 12.5 μg/mL (range of 6.25-25 μg/mL) for H. pylori VacA and CagA +. However, in our study a no significant difference on the resveratrol effect on the sensitivity of H. pylori CagA + and CagA - strains was obtained. The mechanisms of action of resveratrol are very wide 44, and related to H. pylori, Tombola et al 15 suggested that this phenolic compound potently inhibit the VacA, a cytotoxin that plays an important role in the H. pylori colonization and survival in the stomach, and, in addition, it causes epithelial damage 45,46.
H. pylori is susceptible to many antibiotics in vitro, although only a few of them can be used in vivo to treat the microbial infection. Several authors have suggested that several phytoceutical substances can be used either as monotherapy or in combination with antimicrobials 8,12. In the present work, we have studied the potential interaction between two antimicrobials frequently used in the H. pylori therapy and the antioxidants present in red wine. The combination of amoxicillin with resveratrol showed the higher inhibitory effect; on the contrary, for metrodizadole only a significant effect was obtained in the resistant strains for the catechin. Similar potentiation effects between antioxidants and antibiotics have been reported previuosly 47,48. However, alcoholic drinks cannot be consumed under metronidazole treatment because of a disulfiram-like reaction.
In short, the results obtained in this study demonstrate that red wine and resveratrol inhibit the growth of H. pylori strains in vitro and may support their role as chemo-preventive agents of the bacterial infection or the insurgence in H. pylori infected individuals. However, due to the clinical guidelines for the treatment of dyspepsia recommend the stopping of ingestion of alcoholic beverages, further studies based on the effect of the ingestion of antioxidant substances, such as resveratrol, which has shown an inhibitory effect on in vitro growth of H. pylori strains, could be necessary for the development of clinical treatments using resveratrol as chemopreventive agent.