Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Lasallista de Investigación
Print version ISSN 1794-4449
Rev. Lasallista Investig. vol.5 no.2 Caldas July 2008
Artículo original
Photo degradation of the tartrazine dye by means of photofenton, using an ultra violet light lamp*
Fotodegradación del colorante tartrazina mediante fotofenton empleando lámpara de luz ultravioleta
Foto-degradação do corante tartrazina mediante foto-fenton empregando a luz ultravioleta
Joan Amir Arroyave Rojas1, Luís Fernando Garcés Giraldo2, Álvaro de Jesús Arango Ruiz3, Carlota Marcela Agudelo López4
Línea de investigación: Tratamiento de aguas. Grupo de Investigación GAMA y Semillero SIGMA.
* Investigación financiada con apoyo del Fondo para el Desarrollo de la Investigación de la Corporación Universitaria Lasallista.
1 Ingeniero Sanitario, Candidato a Magíster en Ingeniería Ambiental. Jefe del Programa de Ingeniería Ambiental, Corporación Universitaria Lasallista. Coordinador Semillero de Investigación en Gestión y Medio Ambiente - SIGMA. Grupo de Investigación GAMA.
2 Ingeniero Sanitario, Magíster en Ingeniería Ambiental, Especialista en Cuencas Hidrográficas, y Especialista en Ingeniería Ambiental. Decano Facultad de Ingenierías, Corporación Universitaria Lasallista. Director Grupo de Investigación GAMA. Correspondencia: e-mail: lugarces@lasallista.edu.co.
3 Ingeniero Químico. Magíster en Ingeniería Ambiental de la Universidad Pontificia Bolivariana, profesor de la Facultad de Ingenierías de la Corporación Universitaria Lasallista.
4 Estudiante de Ingeniería Ambiental, Corporación Universitaria Lasallista. Semillero de Investigación en Gestión y Medio Ambiente - SIGMA. Grupo de Investigación GAMA.
Recibido: 10/02/2008; fecha de aprobación: 22/09/2008
Abstract
Introduction. All of the production systems demand natural resources to make goods and services, and therefore requires environmental services as inputs and for the disposal of its waste. Food industry is not the exception to this problem, the generation of waste with dyes has a negative impact on all of the water sources in the country, the presence of color in the water deteriorates the aesthetics of water bodies and also generates environmental misbalances because color is an obstacle to the pass of sunlight in the water surface, avoiding the photo synthesis of algae in rivers and brooks. When photo synthesis is avoided, as it is the first link of the food chains, a general misbalance takes place in the ecosystems, because zooplankton and fish species feed from algae, and the economy of villages and towns located at water shores in the country depend mainly on those fish species. Objetive. To evaluate the degradation of the Tartrazine dye using photo catalysis with iron (iii) and an ultra violet light lamp. Materials and methods. An experimental randomized factorial design was used; for the experimental development, we used a photo reactor system, which consists on an ultra violet light lamp, a glass container to keep the sample to be treated and a submersible pump that allows the re-circulation of the solution through the photo reactor system. The degradation of the Tartrazine dye was determined by the use of ultraviolet/visible espectrophotometry. Results. A good percentage of the Tarzatrine dye removal (100,0%) was obtained for the combination of chemical oxidation by using 0 mg/L of Fe3+ and 0,4 % v/v of hydrogen peroxide as oxidation agent, and also the experimental combination of the photo chemical process of homogeneous photo catalysis and the experimental rehearsal of 50 mg/L of Fe3+ and 0,2 % v/v H2O2; It is also considered that the combinations of 0 mg/L of Fe3+ and 0,2 % v/v H2O2 and 50 mg/L of Fe3+ and 0,4 % v/v H2O2 reach very important removal percentages, above 99,0 %, of degradation. Conclusion. Advanced oxidation processes, as homogeneous photo catalysis, are adequate for the removal and elimination of the Tartrazine dye.
Key words: Photo degradation, Tartrazine, Azo dye, Iron (III), Hydrogen peroxide, Food industries.
Resumen
Introducción. Todo sistema productivo demanda recursos naturales para la manufactura de bienes y servicios, y de esta forma requiere servicios ambientales como insumos, al igual que para los vertimientos de sus desechos; la industria de alimentos no es la excepción a dicha problemática, la generación de vertimientos con colorantes impacta negativamente los recurso hídricos en todo el país, la presencia de color en el agua incurre en un deterioro estético de los cuerpos de agua, y de igual forma provoca desequilibrios ambientales debido a que obstaculiza el paso de la luz solar en los espejos de agua superficiales lo cual evita la generación de los procesos de fotosíntesis que realizan las algas en los causes naturales de los ríos y quebradas, al impedir la fotosíntesis, la cual es el primer eslabón en las cadenas alimenticias o tróficas, se produce un desequilibrio general en los ecosistemas, debido a que a partir de las algas se alimentan el zooplancton y el recurso íctico de las cuencas hidrográficas, de este ultimo depende en gran parte las actividades económicas de las poblaciones asentadas en las riveras de las principales cuencas del país. Objetivo. Evaluar la degradación del colorante Tartrazina empleando fotocatálisis homogénea con hierro (III) y lámpara de luz ultravioleta. Materiales y métodos. Se empleó un diseño experimental factorial aleatorizado; para el desarrollo experimental se utilizó un sistema de fotorreactor el cual consistía en una lámpara de luz ultravioleta, un reservorio de vidrio para el almacenamiento de la muestra a tratar y una bomba sumergible que permitía la recirculación de la solución por el sistema de fotorreactor. La degradación del colorante Tartrazina se determinó mediante espectrofotometría ultravioleta/visible. Resultados. Se obtuvo un buen porcentaje de remoción (100,0 %) del colorante Tartrazina para la combinación de oxidación química mediante el empleo de 0 mg/L de Fe3+ y 0,4 % v/v del agente oxidante peróxido de hidrógeno, además de las combinación experimental del proceso fotoquímico de la fotocatálisis homogénea con el ensayo experimental de 50 mg/L de Fe3+ y 0,2 % v/v H2O2; adicionalmente, se considera que las combinaciones de 0 mg/L de Fe3+ y 0,2 % v/v H2O2 y 50 mg/L de Fe3+ y 0,4 % v/v H2O2 alcanzar porcentajes de remoción muy significativos, superiores al 99,0% de degradación. Conclusión. Los procesos de oxidación avanzados, como es el caso de la fotocatálisis homogénea, son adecuados para la remoción y eliminación del colorante Tartrazina.
Palabras clave: Fotodegradación. Tartrazina. Colorantes azoicos. Hierro (III). Peróxido de hidrógeno. Industria de alimentos.
Resumo
Introdução. Todo sistema produtivo demanda recursos naturais para a manufatura de bens e serviços, e desta forma requer serviços ambientais como insumos, ao igual que para os derrames de seus refugos; a indústria de alimentos não é a exceção a dita problemática, a geração de derrames com corantes impacta negativamente os recurso hídricos em todo o país, a presença de cor na água incorre numa deterioração estética dos corpos de água, e de igual forma provoca desequilíbrios ambientais devido a que põe obstáculos o passo da luz solar nos espelhos de água superficiais o qual evita a geração dos processos de fotossínteses que realizam as algas nos causes naturais dos rios e quebradas, ao impedir a fotossíntese, a qual é o primeiro elo nas correntes alimentícias ou tróficas, produz-se um desequilíbrio geral nos ecossistemas, devido a que a partir das algas se alimentam o zôo-plâncton e o recurso íctico das bacias hidrográficas, deste ultimo depende em grande parte as atividades econômicas das populações assentadas nas beiras das principais bacias do país. Objetivo. Avaliar a degradação do corante Tartrazina empregando foto-catálisis homogênea com ferro (III) e de luz ultravioleta. Materiais e métodos. Empregou-se um desenho experimental fatorial aleatorizado; para o desenvolvimento experimental se utilizou um sistema de foto-reator o qual consistia num de luz ultravioleta, um reservatório de vidro para o armazenamento da mostra a tratar e uma bomba submergível que permitia a recirculação da solução pelo sistema de foto-reator. A degradação do corante Tartrazina se determinou mediante espectrofotometría ultravioleta/visível. Resultados. Obteve-se uma boa percentagem de remoção (100,0 %) do corante Tartrazina para a combinação de oxidação química mediante o emprego de 0 mg/L de Fé3+ e 0,4% v/v do agente oxidante peróxido de hidrogênio, além das combinação experimental do processo fotoquímico da foto-catálisis homogênea com o ensaio experimental de 50 mg/L de Fé3+ e 0,2 % v/v H2Ou2; adicionalmente, considera-se que as combinações de 0 mg/L de Fé3+ e 0,2 % v/v H2Ou2 e 50 mg/L de Fé3+ e 0,4 % v/v H2Ou2 atingir percentagens de remoção muito significativos, superiores ao 99,0% de degradação. Conclusão. Os processos de oxidação avançados, como é o caso da foto-catáliss homogênea, são adequados para a remoção e eliminação do corante Tartrazina.
Palavras chaves: Foto-degradação. Tartrazina. Corantes azoem. Ferro (III). Peróxido de hidrogênio. Indústria de alimento.
Introducción
Dyes are one of the most used raw materials in food, pharmaceutical and textile industries, among others. In the world, there are about 10000 kinds of dyes and estimations say that, ever year, 700.000 tons of dyes are generated. From these, about 15% are expelled into the environment with no treatment at all.
The presence of dyes in water bodies produces very bad environmental consequences: They become an obstacle for the spreading of oxygen and light1 and, at the same time, they give water an unaesthetic aspect. Besides, these compounds, because of their chemical nature, are considered as persistent in the environment, and some of their precursors and sub products are cancer agents.
Azoic dyes have a group (-N=N-) in their chemical structure, with aromatic rings in both extremes. They are the most used group of dyes in industries2,3; Recent research works have shown that these dyes are cancerous. Tartrazine has the E 102 (UE)4 and yellow 5 (FDA-USA)5 and belongs to this dye family. Tartrtazine is widely used in food industry and, though it is innocuous in low concentrations, its ingestion in a high quantity can produce allergic reactions, such as asthma and hives. It is also related to children hyper kinetic behavior and tumors in the thyroid gland. This is why some countries, like Norway and Austria, have forbidden its use.
This way, we can observe a potential risk of water pollution with Tartrazine, which has to be removed from the waste water produced by food industries. Therefore, this project looks for the technological development concerning the advanced oxidation processes (POA"s) in the decontamination of waste water in Colombia. It is then observed that Tartrazine is a synthetic product with a wide spectrum of uses. In recent works, it has been intended to produce substitutes for this dye, looking for a reduction of its use because of its secondary effects when consumed, and this is why it is recommended to the makers of products classified as food, cosmetics and medicines that contain Tarzatrine, to modify the sanitary registration6-9 and must include in the information, the text "Contains Tartrazine". It can produce allergic reactions, angioedema type, asthma, hives and anaphylactic shock. This must be done following the dispositions exposed by the specialized board in medicines and biologic products of the reviewer commission within the concept given in the act No. 1 of 20078,9.
At the moment, there are several Technologies available for the treatment of effluents with dyes, among which we have some physical-chemical methods such as adsorption, coagulationflocculation, advanced oxidation and membrane filtration. These methods can be very effective, though many times they are complex and difficult to be applied in a high scale. Nevertherless, the technological development concerning advanced oxidation technologies seeks the implementation and knowledge, national and internationally, about the application of such technologies, which have been used in the degradation of an endless number of environmental pollutants such as pesticides10,11,14-17,21, dyes18-20 and waste from laboratories21, among others.
The Fenton"s reaction10,11,16,17 is known as one of the most classic and powerful oxidation reactions in water. This reaction takes place without ultra violet light, but its oxidation power can be increased by using ultra violet radiation or ultraviolet/visible (Photo fenton reaction), fact that has generated studies to decompose a great number of refractory compounds10-17. The reactive of Fenton is a mixture of hydrogen peroxide and ferrous ion (Fe2+) which produces the free radical hydroxyl (OH.) and the ferric ion (Fe3+).
The hydroxyl free radical is the primary oxidant species made up by the decomposition of the hydrogen peroxide catalyzed by Fe2+ in the absence of quelating agents of the ferric ion. The hydroxyl free radical is the second oxidation agent after Fluor OH., Eo= -2,8 V vs. Fluor, Eo= -3,0 V), and is able to make not specified reactions of some organic compounds (10-15).
When a sufficient concentration of hydroxyl free radical and other radicals is generated, the oxidation reactions of the organic compounds can achieve, even, a total mineralization10,15,20.
Materials and methods
A photo reactor, made up by an ultra violet light lamp, was used. The lamp has a chamber through which the waste water circulates. There, the irradiation of the contaminated water is done with the ultra violet light, by the use of a mercury lamp. The photo reactor system was complemented by a glass container, which is used as a recipient for keeping the sample to be treated. Besides, there was a submersible pump, to execute the re-circulation of the waste water through the ultra violet light lamp. This system was used with a flow of 0,05 L/s and a pH of the solution of 3,0. An initial concentration of 100 mg/ L of Tartrazine dye was used. Its degradation was determined by the use of UV/visible spectrophotometry, after two hours of experiment.
The concentration of catalyst (iron (III)) and oxidation agent (hydrogen peroxide) was added, corresponding to the concentration shown in table 1. To achieve an appropriate experimental development and the fulfillment of the assumption of residuals, three random replicas of the rehearsals were made.
Results
table 1 and graphic 1 show the experimental results obtained by the use of homogeneous photo catalysis using iron (III) and hydrogen peroxide. According to this, it can be seen that the azo tartrazine dye is a not very photo active dye, because of its low degradation when ultra violet light is applied to it. These rehearsals reach a 3,4% of dye removal for two hours period of irradiation of the solution to be treated at a wave longitude of 254 nm by means of the ultra violet light photo reactor using a mercury lamp. These rehearsals correspond to the experimental combination of 0 mg/L of Fe3+ and 0 %v/v of H2O2.
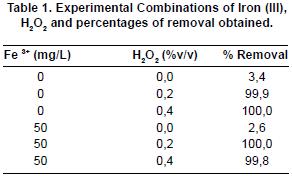
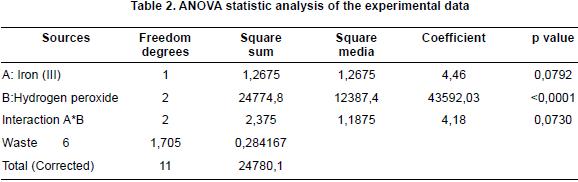
In this same way, it can be obsrerved that the addition of iron (III) as a catalyst in the absence of an oxidation agent (hydrogen peroxide) does not contribute to the dye degradation. For the combination of 50 mg/L of Fe3+ and 0%v/v of H2O2, due to the fact that the removal percentage achieved is 2,6%, a very low one when compared with all of the other combinations, it can be seen that the addition of the catalyst (Fe3+) does not help the degradation reactions of the dye. This result can be corroborated by keeping into account the data that appear in table 2, where the ANOVA for the experimental design given for the study of homogeneous photo catalysis of tartrazine is shown, because the result of p value is higher than 0,05 and, therefore, the presence or addition of the catalyst (Fe3+) does not have a statistic significance in the response variable, which is the removal or degradation of the dye.
On the other hand, in accordance with the results obtained in table 1 and graphic 1, it can be observed that the addition of the oxidation agent hydrogen peroxide (H2O2) generates a high increase in the degradation of the tarzatrine dye. For the four combinations in which hydrogen peroxide was used as oxidation agent, high removal levels were obtained in the two (2) hours treatment of the water sample containing tartrazine dye to be degraded.
The combination of 50 mg/L of Fe3+ and 0,4 v/v of H2O2, reaches the lowest removal percentage among the experiments that use hydrogen peroxide, with a 99,8% of removal, obtaining a good result in the degradation of the tartrazine dye. By this same way, it can be observed that the experimental combination of 0mg/L of Fe3+ and 0,2 %v/v of H2O2, reaches a removal similar to the later combination, around 99,9%. These results are meaningful, because they are high and do not represent big differences in comparison with the highest degradation percentages. These ones correspond to a degradation of a 100% for the experimental combinations of 0 mg/L of Fe3+ and 0,4 %v/v of H2O2 and 50 mg/L of Fe3+ and 0,4 % v/v of H2O2 and 50 mg/L of Fe3+ and 0,2 %v/v of H2O2. Nevertheless, and analyzing the economic factor according to the addition of chemical reactive and the high removal percentages obtained by the advanced homogeneous catalysis oxidation process, the experimental combination of 0 mg/ L of Fe3+ and 0,2 %v/v of H2O2 is defined as the best one.
Besides, a statistic analysis of the experimental data was made, by the use of an ANOVA, as shown in table 2, according to the values obtained of P obtained (p<0,05), it can be seen that the oxidation agent factor (hydrogen peroxide) has a meaningful variation in the change of experimental levels for the response variable (this means the percentage of removal or degradation) while that, for the concentration of the catalyst factor (iron (III)) the value of P equals 0,0792, which is higher than it was expected (p< 0,05) and this means that the variation of the experimental levels does not have a high incidence in the removal percentage of the Tartrazine dye.
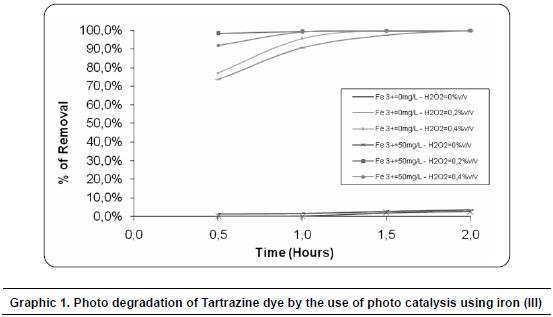
In graphic 2 the method used to identify the meaningful Fisher differences (LSD) can be seen. It is shown that for the interrelationship between the catalyst iron (III) there are no high differences between the medias obtained by experimentation between 0 and 50 mg/L of iron (III). Therefore, it is shown that there are no statistically high differences at a 95,0% level of confidence, between those levels.
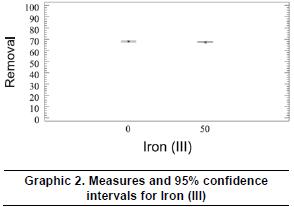
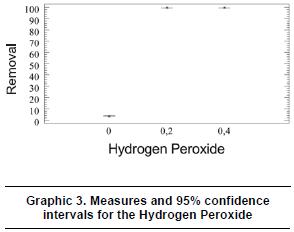
In the same way, graphic 3 shows that for the 95% confidence intervals of the levels of the oxidation agent -hydrogen peroxide, with a level of reliability of 95%, the variations of the media rates between 0,0-0,2% v/v and 0,0-0,5% v/v of hydrogen peroxide, have meaningful differences between the experimental levels and the response variable, this means, the removal of the tartrazine dye, while in the variation between 0,2-0,4 %v/v there is no big difference between the two levels.
Discussion
As it can be seen in graphic 1, when the time of irradiation of waste water containing Tartrazine dye is increased, a higher removal percentage is achieved, because the generation of photo reactions and hydroxyl free radicals is favored, and those free radicals make the degradation of the dye.
In the results obtained from this research work, it can be observed that the Azo dye can be degraded by the use of Chemicals oxidation and homogeneous Photo catalysis by using iron (III), because a dye removal percentage of 100 % is achieved, except for the combination of 0 mg/L of Fe3+ and 0,2 % v/v of H2O2, and 50 mg/L of Fe3+ and 0,2%v/V of H2O2, which is an oxidation by the use of hydrogen peroxide, For this latter combination, there is also a high oxidation percentage, above 99%, thus there is an evidence of the fact that advanced oxidation processes are efficient for the removal of the Tartrazine dye for a 2 hours treatment time.
Conclusion
The implementation of advanced oxidation Technologies (TAO"s), as in the case of homogeneous photo catalysis by the use of iron (III) demonstrates its great number of environmental applications, In this cased, it is used to remove and degrade the Tartrazine dye, commonly used in food industries (2), which is an Azo dye, and this means that its molecule is more stable and difficult to degrade in environmental conditions. We, this way, are advancing in the knowledge and application of one of the TAO"s at a local level, increasing the technological offer to reduce or solve the negative impacts in our immediate natural environment and achieve a sustainable development by the use of environmentally sustainable technologies22, which can be used in cleaner production programs for industries, in this case, for food industries.
References
1. SAWYER, Clair Nathan; McCARTY, Perry L. y PARKIN, Gene F. Chemistry for Environmental Engineering. 4 ed. Estados Unidos: McGraw Hill, 2001. 713 p. [ Links ]
2. RESTREPO GALLEGO, Mauricio. et al. Sustitución de tartrazina por betacaroteno en la elaboración de bebidas no alcohólicas. En: Revista Lasallista de Investigación. Vol. 3, No. 3 (jul - dic 2006); p. 7-12. [ Links ]
3. KAPOR, Marco Andrés. et al. Electroanálise de corante alimentícios: determinacao de indigo carmín e tartazina. En: Eclética Química. Vol. 26, No. 01 (2001); p. 1-20. [ Links ]
4. COMUNIDAD EUROPEA. Decisión No 202, de 17 de marzo de 2005 , relativa a los modelos de formularios necesarios para la aplicación de los Reglamentos (CEE) No 1408/71 y (CEE) No 574/72 del Consejo (E 001, E 101, E 102, E 103, E 104, E 106, E 107, E 108, E 109, E 112, E 115, E 116, E 117, E 118, E 120, E 121, E 123, E 124, E 125, E 126 y E 127) Texto pertinente a efectos del EEE y del Acuerdo UE-Suiza. s.l.: Comunidad Europea, 2006. [ Links ]
5. FOOD AND DRUG ADMINISTRATION. Website of the USA Food and Drug Administration - Center for Food Safety & Applied Nutrition. [online]. Washington: Food and Drug Administration, 2008. [Citado en agosto de 2008]. URL disponible en: <http://www.cfsan.fda.gov/>. [ Links ]
6. RAM, F. S. y ARDERN, K. D. Dietary salt reduction or exclusion for allergic asthma. In: Cochrane database of systematic reviews. No. 3 (2007). [ Links ]
7. COLOMBIA. MINISTERIO DE SALUD. Resolución Número 00580 de 26 Febrero de 1996, Por la cual se modifica el parágrafo primero de la Resolución 10593 de 1985. Bogotá : Ministerio de Salud, 1996. [ Links ]
8. COLOMBIA. MINISTERIO DE LA PROTECCIÓN SOCIAL. Resolución 005109 del 29 de diciembre de 2005, Por la cual se establece el reglamento técnico sobre los requisitos de rotulado o etiquetado que deben cumplir los alimentos envasados y materias primas de alimentos para consumo humano. Bogotá: El Ministerio, 2005. [ Links ]
9. _.Resolución 0670 del 9 de marzo de 2007, Por la cual se establece el reglamento técnico de emergencia sobre los requisitos fisicoquímicos y microbiológicos que deben cumplir los productos de la pesca, en particular pescados, moluscos y crustáceos para el consumo humano. Bogotá: Ministerio de Protección Social, 2007. [ Links ]
10. ARROYAVE ROJAS, Joan Amir y CORREA OCHOA, Adrian Alexis. Fotodegradación de Malatión usando colector solar. Medellín, 104 p. Tesis (Ingeniero Sanitario). Universidad de Antioquia. Facultad de Ingeniería. Ingeniería Sanitaria. [ Links ]
11. ARROYAVE ROJAS, Joan Amir; GARCÉS GIRALDO, Luis Fernando y CRUZ CASTELLANOS, Andrés Felipe. Fotodegradación del Pesticida Mertect empleando Fotofenton con Lámpara de Luz Ultravioleta. En: Revista Lasallista de Investigación. Vol. 3, No. 2 (ene-jun 2007); p. 19 - 24. [ Links ]
12. GARCES GIRALDO, Luís Fernando; MEJÍA FRANCO, Edwin Alejandro. y SANTA MARÍA ARANGO, Jorge Julián. La fotocatálisis como alternativa para el tratamiento de aguas residuales. En: Revista Lasallista de Investigación. Vol. 1, No. 1 (ene-jun 2004); p. 83-92. [ Links ]
13. Red CYTED VIII-G. Eliminación de Contaminantes por Fotocatálisis Heterogénea: Usos de óxidos semiconductores y materiales relacionados para aplicaciones ambientales y ópticas. Brasil: Red CYTED, 2001. 305 p. [ Links ]
14. ARROYAVE ROJAS, Joan Amir; GARCÉS GIRALDO, Luis Fernando y CRUZ CASTELLANOS, Andrés Felipe. Fotodegradación de las aguas residuales con pesticida Mertect en la industria bananera empleando Fotocatálisis con Dióxido de Titanio y Lámpara de Luz Ultravioleta. En: Revista Lasallista de Investigación. Vol 4, No 1 (ene-jun 2007); p. 7-13. [ Links ]
15. _. Empleo de Tecnologías Avanzadas de Oxidación para la degradación del pesticida Mertect. En: 50 CONGRESO ASOCIACIÓN COLOMBIANA DE INGENIERÍA SANITARIA Y AMBIENTAL - ACODAL Y XII BOLIVARIANO DE LA ASOCIACIÓN INTERAMERICANA DE INGENIERÍA SANITARIA Y AMBIENTAL - AIDIS: INTEGRACIÓN, PRODUCTIVIDAD Y RESPONSABILIDAD AMBIENTAL. (12 : Santa Marta : 2007). Santa Marta: ACODAL, 2007. p. 1-8. [ Links ]
16. RED COLOMBIANA DE SEMILLEROS DE INVESTIGACIÓN - NODO ANTIOQUIA. Avances en Investigación Formativa 2006. Medellín: Editorial Artes y letras, 2007. [ Links ]
17. _. Avances en Investigación Formativa 2007. 2 ed. Medellín: Aljara, 2007. [ Links ]
18. GARCES GIRALDO, Luís Fernando. et al. Degradación de aguas residuales de la industria textil por medio de fotocatálisis. En: Revista Lasallista de Investigación. Vol 2, No 1 (ene-jun 2005); p. 15-18. [ Links ]
19. GARCES GIRALDO, Luís Fernando. Cinética de degradación y mineralización del colorante naranja reactivo 84 en aguas. En: Revista Lasallista de Investigación. Vol 2, No 2. (jun-dic 2005); p. 21-25. [ Links ]
20. GARCÉS GIRALDO, Luis Fernando. et al. Fotocatálisis y Electrocoagulación para el tratamiento de aguas residuales: Investigaciones y aplicaciones. Caldas, Antiquia : Corporación Universitaria Lasallista, 2007. 174 p. [ Links ]
21. TELLO RENDÓN, Erick Danilo. Optimización de tecnologías fotocatalíticas de oxidación avanzada aplicada al tratamiento de residuos líquidos de laboratorio. España: Departamento de Química, Universidad de la Palma de Gran Canaria, 2000. 329 p. [ Links ]
22. ARROYAVE ROJAS, Joan Amir y GARCES GIRALDO, Luís Fernando. Tecnologías Ambientalmente Sostenibles. En: Revista de Producción Más Limpia. Vol. 1, No 2 (jul-dic 2006); p. 78-86. [ Links ]














