1 Introduction
For synthetic polymers, obtained from petroleum, the degradation is a process that occurs slowly and its speed depends on the environmental conditions and the composition of these polymers. This longer life cycle causes accumulation of these materials in the environment after disposal, if not recycled. Thus, biodegradable polymers such as PHB are seen as a viable alternative for the management of these wastes. The objective of this work is to review the literature on the process and conditions for degradation of PHB.
2 Degradation of PHB
The degradation of a polymer can be characterized as a process that results in the breakage of a large and complex molecule into smaller molecules [1].
It can also be understood as the process of changing in chemical properties or physical of the polymer [2]. The degradation, loss of functionality of the polymer [2], in general, occurs by division of the polymer chain and breakage of the structure in the crystalline reticulum [3]. The speed of the degradation varies according to the chemical structure and the degree of crystallinity of the polymer [3]. The chemical structure, the molecular weight, the morphology and the crystallinity of the material influence the degradation rate [4],[5]. Some reflections of degradation in the polymer can be seen as changes in the mechanical characteristics, electrical or optical, crazing, stress cracking, erosion, discoloration, phase separation, delamination or formation of a new functional group [2]. Environmental factors (light, air, heat and humidity), chemical, mechanical, biological and processing are responsible for triggering the degradation of polymers [3]. Depending on the process of initiation of reactions, the degradation may be called thermodegradation, thermo-mechanical degradation, abiotic degradation and biodegradation [6].
The following shall be described in the different forms of degradation and also described its respective particularities that each one has in relation to the factors that influence the process, the by-products, the mechanism and the assessment methods of degradation.
2.1 Thermal and chemical degradation
The degradation caused by chemical agents apparently would happen only if the polymer was exposed to an aggressive chemical agent specific [6]. However, this does not match reality, because all polymer produced on an industrial scale can contain contaminants that can promote the start of degradation by chemical attack.
Cases of this type of degradation are polymers used in contact with other materials (metal, for example) are apparently inert, but that can be chemically activated by heating. These materials are called internal chemical agents, such as: contamination from the polymerization process, residues of catalytic converter, additives, mixture with product off-grade, loads, agents of color and components of the formulation of the polymer [6]. This type of degradation could be observed in the study of KAWALEC et al. [7], which intermingled the PHB with alkaline metal salts (Cs, K, Li) and with metal salts bivalents (Ca, Mg, Zn) with the objective of obtaining the PHB macro monomers, through thermal degradation at a moderate temperature (150 ◦C to 170 ◦C). The degradation was perceived by the loss of mass of the polymer, in percentage terms, and the by-products found of this process were the E-crotonic acid (greater quantity) and Z-crotonic acid (lower quantity). It was concluded that the salts of alkali metals are more effective to accelerate the degradation of PHB in mild conditions, temperature, even not being able to catalyze the degradation process. Now the salts of Ca, Mg and Zn showed less active, however, were also able to decrease the thermal stability of PHB, accelerating the degradation. The mechanism of degradation occurs by anionic route (known as E1cB mechanism, caused by deprotonation of PHB). In this type of degradation of one of the factors that influence the speed of the process is the dimension of the radius of the cation that, the greater it will be, the greater the rate of degradation, by promoting the fission of the polymer chain more quickly. Other factors are the content of salt, the type of cation and the nature of the anion. The conclusion that the presence of chemical elements in the PHB decreases the thermal stability of the material, accelerating the process of degradation, was also evidenced by BORDES et al. [8] which added surfactants of ammonia in polymer and noticed the more pronounced degradation of PHB when mixed with surfactant.
2.2 Thermodegradation
This process can be explained in a simple way as being the breakage of the long polymer chain in the minor monomeric units through heat. It is also known as an oxidative reaction term, since it is necessary the presence of oxygen [1]. Some factors affect the temperature at which the thermal decomposition of the polymer initiates. The inter and intra molecular interaction is one of these factors, as it relates to the way in which the heat is transported by polymeric mass, i.e., the thermal conductivity of the material. This is because a more effective transport of heat will favor the thermal degradation, while the opposite will cause superficial degradation [6]. Another factor that influences the thermal stability of the polymer is the mobility of macromolecules, which in turn depend on the temperature of the phase transition of first and second order of the material, the degree of crystallinity and the location of the crystalline phase in the polymeric material after processing [6].
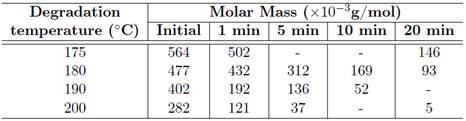
The degree of the folding polymeric chains also affect the dissipation of thermal energy in the material [6]. Thus, a greater folding generates greater restriction of macromolecular movements and a lower thermal power dissipation, increasing the likelihood of breakage of chemical bonds. The temperature of thermal degradation also depends on the power of connections that constitute the polymer. The reactions involved in this process leads to changes in physical properties and optical drive from PHB, and an example of this is the shift in the molecular mass of the material [2], which causes a greater mobility of its polymeric chains [9]. This finding is in agreement with the study of CHEN et al. [10] that indicate a reduction of molar mass of PHB when submitted to temperatures above the melting range, due to thermal degradation. The increase in the exposure time and temperature influence the reduction of molar mass of PHB [10],[11]. This can be seen in Table 1 which shows the dependence of the molar mass of PHB on various temperatures.
The smaller the initial molar mass of PHB the lower will be its thermal stability and the faster will be its degradation [8]. The thermodegradation may cause other changes in the PHB as the reduction of ductility, embrittlement, changes in coloration, stress cracking and the reduction in other mechanical properties [2]. The tension and the rupture can be reduced by 30 % of its initial value when the PHB is subjected to a temperature of 180 ◦C for approximately 10 minutes [12].
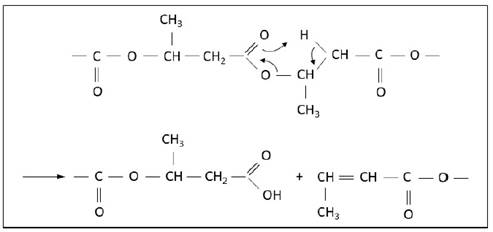
When the PHB undergoes thermal degradation its molar mass initially decreases, is replaced by a slight increase and returns to decrease. This slight increase of molar mass occurs by the reaction of the molecules which have separated and it occurs change of the characteristics of the polymer. These reactions are called polycondensation and they occur between the hydroxyl terminals of PHB original and existent carboxylic acids groups and newly-formed in the degradation process [2]. The proposed mechanism for thermal degradation of PHB is driven almost exclusively by random fission ester group to temperatures above 200 ◦C [9]. This mechanism facilitates the division of the groups next to the end of the chain, fact that generates carboxylic and vinyl groups (crotonate) [9],[13]. In addition, the increase of temperature causes the degradation process evolves, implies in the complete polymer volatization (between 250 ◦C and 300 ◦C) and entails the formation of dimers and trimers crotonic acid as final products [13]. The mechanism of thermal degradation of PHB can be seen in Figure 1.
The study of ARIFFIN et al. [14] deals with the possibility of other degradation mechanisms taking place, besides random fission of PHB. This question is based on the broad range of variation found in literature for the activation energy of degradation (between 110 kj/mol to 380 kj/mol). In this context, the study of ARIFFIN et al. [14] seeks to characterize the products of pyrolysis that occur during the process of thermodegradation. Thus, crotonic anhydrides and its oligomers were found in smaller quantity, which led the authors to associate the occurrence of dehydration as a lesser reaction within the process of thermal degradation. According to the authors, the formations of these final products happen by condensation reaction between carboxylic groups. This way, this study concludes that the pyrolysis of PHB has other reactions (such as dehydration) besides random fission, and this indicates the complexity of the process of thermodegradation of this material.
CHEN et al. [10] led their work to understand which is the fact that the insulated heating causes in the molecular mass and in the crystallinity of PHB, because the process of an insulated treatment is one of the stages of production of PHB, the drying, and that the majority of studies of thermodegradation made with this material use not isotherms techniques. According to these authors, the main chain of PHB is cleaved in a one-step process, the decomposition temperature is between 190 ◦C to 300 ◦C and that the temperature of the beginning of decomposition is 240 ◦C. Another finding of CHEN et al. [10] was the constant rate of degradation isothermal for the PHB, indicated in Table 2.
Still in accordance with these authors, crystallinity of PHB increased along with the temperature that ranged between 130 ◦C to 150 ◦C and dropped quickly from 180 ◦C (PHB merges near this temperature). In face of these findings, the authors concluded that the properties of PHB do not change with heating below 60 ◦C, however, higher temperatures potentiate the degradation of this material and therefore the PHB must pass by drying at temperatures below 60◦C to minimize the degradation of their properties [10].
2.3 Thermo-mechanical degradation
It is the process of breakage of chemical links of the polymer when these are in their softened state (with greater fluidity) or above Tm, caused by mechanical effect associated with the temperature. This process occurs mainly during processing by extrusion, injection, calendering or wiring [6]. In thermodegradation in the presence or not of oxygen influences in the form that the degradation occurs. When there is a limitation in the diffusion of oxygen, the oxidative processes do not compete with the breach of chemical bonds and reticulations. However, in processes with availability of oxygen the favoring of oxidative processes occurs that pass to compete with the formation of vinylic connections and reticulations [6]. This process can also be explained having as base the search of PACHEKOSKI et al. [9] that had as objective to characterize the degradation caused in the PHB by common industrial processes (extrusion and injection). The majority of studies found in literature takes into account only the thermal degradation of PHB without that this material to pass through any processing technique.
To get to the research objectives PACHEKOSKI et al. [9] made a comparison between characteristics of not processed and processed PHB. One of these comparisons were made between the molar mass theoretical reduction and real reduction of molar mass after processing. First, the authors took into consideration only the thermal degradation and calculated its value as a function of kinetic parameters, such as constant thermal degradation and the processing time of the material. For the second, they submitted the PHB to temperatures above 170 ◦C and the shearing tension and evaluated reduction of molar mass.
It was observed that the processing of the material caused no change in the degree of crystallinity and in chemical structure of PHB, did not affect the relaxation of the polymer and nor modified the transition temperature and the melting temperature. The processing affected the cold crystallization temperature (Tcc) of the polymer, which presented decrease of its value and the melt flow index (MFI), which increased causing better fluidity of the material. If the thermal degradation was the only factor of degradation of the material involved in processing, the variation between the reduction of theoretical molar mass and the reduction of real molar mass real should be around 3 %. However, the difference between both was 29 %. The reduction of Tcc, the increase in MFI and the reduction of molar mass greater than expected led the authors to conclude that the thermal degradation of the material is not the main factor for the reduction of molar mass of PHB [9]. The mechanical forces involved in the extrusion process and injection are able to reduce the molar mass of the material through the shearing of the polymer chain. Therefore, it should be taken into account the shearing forces, present in the injection and extrusion, when processing the PHB industrial scale, because this material is susceptible to thermo-mechanical degradation.
2.4 Abiotic degradation
This degradation process occurs without the presence of living beings and one of the most known reactions is the hydrolysis. In this type of degradation, the crystallinity of the polymer is a determining factor, as the crystalline regions of the material are impermeable to water [3]. The polyesters, as the PHB, are more susceptible to hydrolysis due to the presence of the group - COO - [3], and can have their ester connections cleaved through the hydrolytic process, under natural conditions [15].
SALGUERO et al. [16] submitted the PHB to a hydrolytic degradation in vitro, in a saline solution of phosphate with pH 7, at a temperature of 37 C during 40 days. After this period and in these conditions, the authors verified that the contact angle of the PHB decreased by about 10 % because the fact of hydrolysis generating hydrophilic chains and, thus, the material was able to absorb higher amount of water. This study also showed that the PHB lost little mass during the incubation period, a value that was between 1 % and 1.5 % [16]. This small percentage may be explained due to micro porosity of polymer that reduced the rate of water adsorption into the polymeric matrix and at the same time represented a barrier to the flow of the products of degradation to the solution. Another observation made by the same authors, through DSC, indicated a decrease in the value of the Tg and, consequently, an increase in the mobility of the polymeric chains in amorphous region. In addition, the crystallinity increased with the hydrolytic degradation.
2.5 Photodegradation
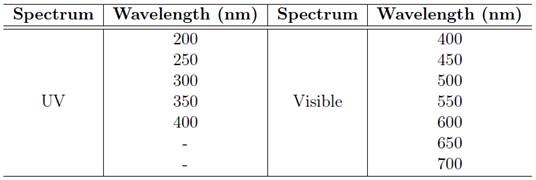
The photodegradation is a process by which particles of high intensity photons are used to degrade polymers in smaller particles. It happens when high radiation intensity (high energy) react with groups of the polymer [17], activating the electrons of these chemical groups bringing their respective reactivity, which causes oxidation, breaking the chains or other degradations [2],[3]. DE PAOLI [6] defines this process through off photochemical reactions that occur with the participation of a molecule or chemical species in an excited electronic state which is generated by absorption of light in bands of wave length ranging from ultraviolet (200 nm to 400 nm) to visible (400 nm to 700 nm). Since a photochemical reaction needs a excited state, that is generated by the incidence of the light on the polymer and, taking into account that the sun is the most important source of light for the Earth [18], the photodegradation can also be defined as the ability of the polymer in absorbing the harmful part of tropospheric solar radiation [2]. Thus, the solar light after being filtered by the ozone layer, it can reach the Earth as UV-B radiation (295 nm to 315 nm) and UV-A (315 nm to 400 nm), which are directly responsible for the photodegradation. It can reach with wavelength referring to the visible part of sun light (400 nm to 700 nm), which accelerates the polymeric degradation by heating. It can also come to Earth as infrared radiation (above 700 nm), which accelerates the thermal degradation [2],[6]. Table 3 shows the spectrum of light and the wavelength.
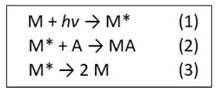
The photochemical degradation occurs by the light that is absorbed by the polymer. The energy that is not absorbed in the form of electronic excitation by system will not cause photochemical effect [6]. The diagram shown in Figure 2 represents a type of photodegradation reaction. The stage "1" represents the absorption of light by the chromophore group of the polymer (photochemical group that absorbs light), which causes an excited state M*. In stage "2", this excited state reacts with a molecule A, that generates a molecule MA. In stage "3", a chemical bond of the chromophore group suffers a homolitic breakage, generating two macro free radicals M, which in the case of PHB the reticulation begins [6], although it is not the prevailing response [19]. It is worth remembering that "hv" refers to the lightening energy.
The carbonyl groups of polymers, due to its characteristic of absorption of light, are responsible for the start of the majority of the photochemical processes. This makes the material photochemically unstable, since it is this group that initiates auto catalytic reactive processes [6]. In the case of PHB, the photochemical reaction that connection C = O can suffer is the Norrish I [19]. In this reaction, there is the formation of free radicals that launch reticulations (not predominant) [19],[6] and the reactions of division (predominant) [20]. An important characteristic of the photodegradation is that this process occurs on the surface of the polymer [19] and the depth of penetration of light, of the order of only some µm of thickness, depends on the wavelength [6]. At the photodegradation, the efficiency depends, mostly of the emission intensity of UV radiation [21]. The photodegradation presents the formation of additional chromophores in propagation step that as a consequence triggers new chain reactions and accelerates even more the degradation process [6]. In the study by SADI et al. [19], in which the PHB was submitted to photodegradation through artificial UV radiation, it was found that this process creates greater quantity of carbonyl. According to these authors, the index of formation of carbonyl increased according to the time of exposure to radiation of PHB, as it can be seen in Table 4.
PHB used for the experiment went through processing and that made the material brown coloring. After a comparison between the PHB in solution before processing, PHB in solution after processing and the PHB solid after processing, it was verified a difference in the material transmittance. The brown color of PHB was a factor that has blocked the UV radiation, leaving the material little conveyor. This has caused a significant impact in the profile of the degradation material since the intensity of UV radiation incident on layers of PHB were widely weakened, when these were directed to the inside of the material [19]. Both the study done by SADI et al. [19] as the one conducted by CARASCHI et al. [22] noted the same changes in the PHB caused by photodegradation: change in coloration of the material, becoming more opaque and whitish; increase of crystallinity; surface became rougher by presenting leveling on the surface, cracks and fissures. The PHB also suffers reduction of molar mass, in the melting temperature and mechanical properties [19].
The biodegradation of PHB is impaired when the material is replaced by a prior photodegradation [19]. The explanation for this is that the UV irradiation causes a superficial increase of the crystallinity of PHB and it is known that the enzymes responsible for hydrolysis this polymer prefer, in the initial stages, the amorphous region of the material. Therefore, this initial delay caused by the photodegradation, does not prevent the decomposition of PHB, since after the consumption of the superficial layer of the material occurs the acceleration of biodegradation through the consumption of degraded molecules, below the first layer, which could not be reorganized in crystals. ROSA et al. [23] examined the biodegradability of PHB and PCL in soil compostage at pH 7, 9 and 11 before and after submission of the polymers to UV-B irradiation for 192, 425 and 600 h. They concluded that UV-B aging did not produce chemical changes that improved the rate of biodegradation, except when PHB was irradiated for 425 h and buried in soil compostage at pH 11.
TERTYSHNAYA and SHIBRYAEVA [24] conducted experiments to study the degradation of PHB films and their blends with ethylene-propylene rubber (EPDM) during treatment with water and UV radiation at 20 and 90 ◦C. They observed that pure PHB showed the highest weight loss (10%) during degradation in distilled water at 20 ◦C. In the case of blends, the most significant changes in component structures are observed for the 50:50 sample (% wt). In addition, water affects the structure of both PHB and EPDM, as confirmed by IR spectroscopy. Based on the DSC and IR data, the mechanism controlling the combined effect of UV and water radiation on PHB and its blends with EPDM has been suggested.
IVANOV et al. [25] observed that PHB is able to considerably increase the rate of photodegradation and photooxidation of polyethylene (PE), from petroleum, in blends of PE with PHB. According to the authors, this fact occurs due to the presence of products formed as a result of the thermal oxidation of PHB during the blending of the polymers and the formation of films.
2.6 Biodegradation
The literature presents two routes to the process of biodegradation of PHB: enzymatic and microbiological. Each of these routes will be explained separately in the next sections with the objective to describe in more detail the particularities of such routes.
2.6.1 Enzyme degradation This type of degradation is similar to biodegradation concerning the fact of being catalyzed by the action of enzymes of micro-organisms, however, it differs from the moment that the enzymes used in this process have already been previously isolated (biocatalysis in vitro) [26] It is used in tests where it is desired to evaluate the biodegradation of determined polymer in experimental shorter times, to obtain results quickly. These tests based on enzyme degradation are shown important once that the conventional are more time-consuming and the enzymatic tests may give the result within a period of hours, days or weeks if a test system controlled and defined is used [2]. This process happens through the hydrolysis of connections esters of polymer [26],[15]. First, the enzyme reaches the polymer and then hydrolytic catalysis [2].
During the degradation enzymes break the long polymeric chain, for- ming oligomers and after some time, these oligomers may be hydrolyzed to monomeric units of the polymer [26]. Two categories of enzymes are actively involved in the degradation of PHB: the intra and extracellular matrix [2]. As the PHB is a polyester, this polymer is susceptible to the action of stereolithic enzymes, as the esterases, lipases and proteases [26]. However, the literature reports that the use of specific enzymes to degrade the PHB, is the case of PHB depolymerase, that can be secreted by various microorganisms and have an important role in the metabolism of PHB in the environment [2].
Some of these PHB depolymerase were isolated and purified of microorganism species Alcaligenes, Comamonas, Pseudomonas and it was noticed that the optimal actuation of these enzymes occur at pH between
and 9.8 [22]. The exceptions are the PHB depolymerases from Pseudomonas picketti and Penicillium funiculosum that have better performance in pH between 5.5 and 7.0 [26],[2]. There are also other species that are able to excrete depolymerase PHB, as Bacillus thuringiensis, Streptomyces sp., Alcaligenes faecalis, Pseudomonas fluorescens [26], Acidovo- rax facilis, Aspergillus fumigatus, Pseudomonas lemoignei, Acinetobacter johsonni, Comamonas testosteroni, Flavobacterium johnsoniae, Pseudoal- teromonas haloplanktis, Vibrio ordalii, Zooglea ramigera and Pseudomonas stutzerii [27-30].
SHAH et al. [2] report that a primary analysis revealed the structure of depolymerases PHB. These are composed by the field of connection to the substrate, by catalytic domain and by region of connection, which unites the two preceding domains. The connection domain to the substrate links the enzyme to PHB. Now the catalyst domain is the responsible for the break of connections.
2.6.2 Biotic degradation It is the type of degradation that involves biological activities and can be defined as an irreversible process and important for a considerable change in the physico-chemical properties of the materia [31], in which a series of reactions break the connections of the main chain and form other chains in polymer. It can also be called biological degradation, because organic substances are broken down by living organisms [2], are converted into simpler compounds, mineralised and redistributed through elementary cycles as carbon, for example [32].
This type of degradation should only occur when the polymer and the micro-organism coexist in a same rational system [26] and when this system is a biologically active environment. When comparing with other chemical routes, the biotic degradation is a slower process and usually happens in milder conditions of temperature and pH [26]. The biodegradation occurs in a natural way, has no initiator reaction and begins by the microorganisms action [33]. These micro-organisms are found in the environment and can be bacteria, fungi and algae [2],[34]. In addition to the microorganisms reported, in enzyme degradation section, as being able to excrete enzymes capable to metabolize the PHB, still there are others that are also able to degrade this polymer. These are belonging to different fungi species: Acremonium, Cladosporium, Debaryomyces, Emericellopsis, Eu- penicillium, Fusarium, Mucor, Paicilomyces, Penicillium, Pullularia, Rhodosporidium and Verticillium [1]. So that the process of biodegradation occurs is necessary that the micro-organisms produce enzymes responsible for breaking the chains and to make such polymeric chains can be processed and digested by micro-organisms [34],[35]. The microorganisms are an excellent source of enzyme due to their biochemistry diversity [36]. There are two categories of enzymes responsible for the process of biodegradation, the intracellular (located inside the cell) and the extracellular (synthesized and excreted in the middle) [2],[37].
Each of these categories of enzymes participates in one of the steps of biodegradation. First the microorganisms colonize the surface of polymers, forming the biofilm (complex mixture of micro-organisms, water and ex- tracellular polymeric substances), and this step is called biodeterioration [38],[39]. As the majority of polymers are too large to pass by the cellular membrane of micro-organisms, it is necessary that the chains of materials are broken, forming smaller compounds, and thus the cells are able to absorb and then degrade the polymer [2].
It is in this context that the extracellular enzymes come into action. They are responsible for digesting the polymeric chains in smaller units (oligomers, dimers and monomers), through the hydrolysis, for which these compounds pass by the semipermeable membrane of the micro-organisms [2],[32]. This step of the process can be called depolymerisation or biofragmentation [2]. In the next step, when the polymer is assimilated by the cells, the intracellular enzymes metabolize the material and use it as a source of nutrients and carbon [2],[37]. This last step is known as mineralization [2]. It is worth remembering that the polymeric degradation rarely reaches 100 %, since part of the carbon of the polymer will be embedded into the microbial biomass [26]. The biodegradation process described above is affected by an external factor, the presence of oxygen, that will establish the conditions of aerobic or anaerobic conditions [26]. What differentiates a condition of another is the presence or not of oxygen and the final products of the process [2]. In aerobic conditions are produced CO2, water and microbial biomass. Meanwhile, in anaerobic conditions are pro- duced CO2, H2O, CH4 and microbial biomass [31],[40],[41]. Usually, the aerobic conditions occur in nature, the anaerobic conditions in sediments and landfill and the conditions partially aerobic and partially anaerobic in composting and in soil [19].
The biotic degradation generates some losses of the polymer properties such as: a consequent change in chemical structure [42], a significant reduction of average numeric molar mass (Mn), change in the surface roughness, emergence of puncture and craters, structural and mechanical deformations, discoloration or color change, fragmentation, fragility and formation of biofilms on surface [2]. Often biodegradation in different ecosystems is affected by microbiological and environmental factors and material characteristics, being these interdependent. As microbiological factors there are the distribution, abundance, diversity, activity and adaptation of the microbiota. The adaptation of the microbiota can increase the degradation, since it may occur a change in the genotype of the species, which induces the production of extracellular enzymes, or can happen a change in the structure of the community through the increase of species of microorganisms involved in biodegradation [43]. So that there is a breach of the polymeric chains, several microbial populations should be involved [2].
The environmental factors that affect the biotic degradation are: the pH, temperature [26], the type of microbial population present in the environment and the supplementation of nutrients [44]. Now the factors relating to the characteristics of the polymer are: the porosity of the polymeric matrix since the biodegradation happens by the accession of the microorganisms in the material, the nature of the pre-treatment, the melting temperature, the surface area, the type of group also in the main chain, the types of connections, the conformational flexibility, the tacticity, the de- gree of crystallinity, the presence of additives and plasticizers [43]. Other characteristics of the material which influence the speed of the degradation are: morphology, which includes the occurrence and frequency of crystalline phase, size, shape and number of crystallites, seen that regions amorphous are more susceptible to hydrolysis than the crystalline regions, the hydrophilicity and the chirality of polymer [26].
3 Final considerations
Degradation is a physical, chemical or even biological process that leads to the loss of functionality of the polymer and the loss of its molar mass. This change in the properties of a polymer can occur in different ways according to the process of initiation of the reactions. For PHB, which is a material susceptible to degradation may occur thermodegradation, thermomechanical degradation abiotic degradation, photodegradation and biodegradation.
The thermodegradation and thermomechanical degradation characteristics of the PHB require additional care mainly at the time of processing, since the drying temperature of the material to its injection or extrusion, given its narrow processing window. In relation to abiotic degradation is observed that the more permeable is the material most subject it will be hydrolysis. This is a property that can be advantageous to dispose of the material, but the high crystallinity of PHB reduces that possibility. One of the photodegradation characteristics of the material is at the same time increases the degree of crystallinity, reducing its permeability, eventually making it more fragile. Finally, it is clear that the degradation is not always a bad thing. The biodegradation of PHB allows products produced with this material are degraded at the end of its life cycle, favoring its disposal sustainably