Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Producción + Limpia
Print version ISSN 1909-0455
Rev. P+L vol.8 no.1 Caldas Jan./June 2013
Evaluation of the biocidal potential of hydrogen peroxide and copper sulphate for biofilm removal in drinking water systems*
Evaluación del potencial biocida del peróxido de hidrógeno y el sulfato de cobre en la remoción de biofilm en sistemas de agua potable
Avaliação do potencial biocida do peróxido de hidrogênio e o sulfato de cobre na remoção de biofilme em sistemas de água potável
Diana Catalina Rodríguez**, Gustavo Peñuela**, Jordi Morató***
*Article derived from the research project "Control of biofilm in drinking water distribution networks by the use of hydrogen peroxide and copper sulfate" financed by the Contamination Diagnostic and Control group (GDCON), Universidad de Antioquia, Medellín, and developed between August 2006 and June 2011.
**Diagnostics and Pollution Control (GDCON), University of Antioquia, Medellin-Colombia.
***Lab. Health & Environmental Microbiology Polytechnical University of Catalonia,Terrassa-08222. Barcelona-Spain.
Mail: Diana Catalina Rodríguez L. e-mail: catalinarodriguez@udea.edu.co
Article received: 22/04/2012; Article approved: 01/06/2013
Abstract
Introduction. The biofilms are communities of microorganisms which adhere to surfaces, thus avoiding the rapid and effective biocidal action and causing deterioration of the quality of the water in the distribution networks. Objective. To evaluate the efficiency of removal of biofilms in distribution networks of drinking water, as biocides using hydrogen peroxide and copper sulfate at different concentrations. Materials and methods. Biocides such as hydrogen peroxide (H2O2, 200 mg/L, 1000 mg/L and 12000 mg/L) and copper sulfate (CuSO4, 0.2 mg/L, 0.6 mg/L and 1.0 mg/L) were used. They were applied to a pilot system used for the formation of biofilm in the distribution system of Medellin, Colombia. The time taken for the adhesion of microorganisms to the pilot was 20 days. Results. A biofilm removal of 99% was obtained, using a concentration of 12000 mg/L of H2O2 in a contact time of 60 minutes, consuming 27% of the initial H2O2. On the other hand, the use of CuSO4 reached the maximum removal, 67%, using a concentration of 1.0 mg/L in a contact time of 60 minutes, consuming 41% of the initial CuSO4 and evidencing the efficiency in the removal of hydrogen peroxide. Conclusion. The H2O2 has shown to be a biocide which acts strongly on the bacterial cells since in the three concentrations used it was observed a significant decrease in heterotrophic bacterial counts, compared with the three tested concentrations of CuSO4.
Key words: biofilm, biocide, hydrogen peroxide, copper sulfate, heterotrophic bacteria.
Resumen
Introducción. Los biofilms son comunidades de microorganismos que se adhieren a las superficies, evitando así la rápida y efectiva acción biocida y causando deterioro de la calidad del agua en las redes de distribución. Objetivo. Evaluar la eficiencia de remoción de las biopeliculas en las redes de distribución de agua potable, empleando como biocidas el peróxido de hidrogeno y sulfato de cobre a diferentes concentraciones. Materiales y métodos. Los biocidas utilizados fueron peróxido de hidrógeno (H2O2, 200 mg/L, 1000 mg/L y 12000 mg/L) y sulfato de cobre (CuSO4, 0.2 mg/L, 0.6 mg/L y 1.0 mg/L), aplicados a un sistema piloto utilizado para la formación de biofilm en la red de distribución de Medellín, Colombia. El tiempo para la adhesión de microorganismos al piloto fue de 20 días. Resultados. Una remoción de biofilm de 99% se obtuvo utilizando una concentración de 12000 mg/L de H2O2 en un tiempo de contacto de 60 minutos. Se consumió 27% del H2O2 inicial. Por otra parte, el uso de CuSO4 alcanzó una tasa máxima de remoción de 67% utilizando una concentración de 1.0 mg/L en un tiempo de contacto de 60 minutos, consumiendo 41% del CuSO4 inicial, evidenciando la eficiencia en la remoción de peróxido de hidrógeno. Conclusión. El H2O2 demostró, ser un biocida que actúa fuertemente sobre las células bacterianas, dado que en las tres concentraciones utilizadas se observó una disminución significativa en el conteo de bacterias heterotróficas en comparación con las concentraciones de CuSO4 evaluadas.
Palabras clave: biofilm, biocida, peróxido de hidrógeno, sulfato de cobre, bacterias heterotróficas.
Resumo
Introdução. Os biofilmes são comunidades de microrganismos que se aderem às superfícies, evitando assim a rápida e efetiva ação biocida e causando deterioração da qualidade do água nas redes de distribuição. Objetivo. Avaliar a eficiência de remoção de biofilmes nas redes de distribuição de água potável, como biocidas utilizando peróxido de hidrogênio e sulfato de cobre em diferentes concentrações. Materiais e métodos. Os biocidas utilizados foram peróxido de hidrogênio (H2Ou2, 200 mg/L, 1000 mg/L e 12000 mg/L) e sulfato de cobre (CuSO4, 0.2 mg/L, 0.6 mg/L e 1.0 mg/L), aplicados a um sistema piloto utilizado para a formação de biofilme na rede de distribuição de Medellín, Colômbia. O tempo para a adesão de microrganismos ao piloto foi de 20 dias. Resultados e discussão. Uma remoção de biofilme de 99% se obteve utilizando uma concentração de 12000 mg/L de H2Ou2 num tempo de contato de 60 minutos. Consumiu-se 27% do H2Ou2 inicial. Por outra parte, o uso de CuSO4 atingiu uma taxa máxima de remoção de 67% utilizando uma concentração de 1.0 mg/L num tempo de contato de 60 minutos, consumindo 41% do CuSO4 inicial, evidenciando a eficiência na remoção de peróxido de hidrogênio. Conclusão. O H2Ou2 demonstrou,ser um biocida que atua fortemente sobre as células bacterianas, dado que nas três concentrações utilizadas se observou uma diminuição significativa na contagem de bactérias heterotróficas em comparação com as concentrações de CuSO4 avaliadas.
Palavras importantes: biofilme, biocida, peróxido de hidrogênio, sulfato de cobre, bactérias heterotróficas.
Introduction
Biocides can be added to other materials to protect them against biological infestation and growth. Biocides are prepared containing one or more active substances, and are made up in the form in which they are supplied to the user. They are intended to destroy, deter, render harmless, prevent the action of or otherwise exert a controlling effect on any harmful organism by chemical or biological means. A Biofilm is a complex community of microorganisms fixed to a surface which is either alive or inert (Costerton; et al., 1995). It is adapted to conditions of scarcity of nutrients and high levels of chlorine (Codony; et al., 2002) and is surrounded by a layer of extracellular polymeric substances (EPS) that it secreted (Costerton; et al., 1995) these substances facilitate the accession of new cells and particulate matter, allowing the absorption of nutrients and preventing the access of biocides (Codony; et al., 2002; Stopforth; et al., 2002). Overall the EPS act as protectors of a variety of environmental conditions of stress such as ultraviolet rays, changes in pH and drying (Flemming, 1993). The consequences of biofilm growth are associated with problems regarding the obstruction and corrosion of drinking water systems and effects on water taste and smell. Biofilm can also promote the presence of pathogenic organisms that can grow within it, or occasionally disguise pollutants (Costerton; et al., 1995), which prevents a proper disinfection and causes an increase of these pollutants in drinking water systems (Lechevallier, Cawthon & Lee, 1988; Camper, 1994). In drinking water distribution systems, there are a number of factors that can have a significant impact on the formation of biofilm, these are: concentration of biodegradable organic matter (BOM), concentration of residual disinfectant, temperature, corrosion (Ndiongue, Huck, & Slawson, 2005), material and age of the pipe, flow regime and retention time of water inside the pipes (Delanoue; et al., 1997). One of the ways to remove this biofilm in the drinking water systems is through the use of biocides. There are several biocides that cannot be used in aqueduct networks because of their toxicity to humans and lack of effectiveness in the removal of biofilms in distribution systems. In this study the efficiency of hydrogen peroxide (H2O2) and copper sulfate (CuSO4) was evaluated in the removal of biofilm. It should be taken into account that both biocides can be used as disinfection systems for a few hours in cases where the use of a disinfectant other than chlorine is required. There are several laboratory methods that are used for studies of biofilm formation in distribution systems. In this investigation a system of packed beds was employed which consisted of a cylinder containing glass spheres (Codony, Domenico & Mas, 2003) used as a porous medium of support for the adherence of the biofilm(Morató, 2001). It wasdesigned to obtain sufficient quantities of samples for analysis in the minimum space possible. Using this method the biofilm disinfection was measured and evaluated.
Work was carried out at the GDCON Group laboratory of the University of Antioquia (Medellín-Colombia), where a packed-bed system was connected to the potable water distribution system for a time of 20 days to allow the formation of biofilm. A shock treatment was then applied for an hour using different concentrations of hydrogen peroxide and copper sulfate. The efficiency of both biocides was assessed by performing a heterotrophic plate count (HPC) and analyzing the residual material of each biocide.
Materials and methods
Biofilm formation system
The pilot system used for biofilm development contained 3 different units, each with 10 packed bead cartridges filled with sterile glass spheres. Each cartridge was constructed with PVC high pressure pipes (RDE - 21) ½ " diameter. In each system, the water flow was controlled by 2 ½" valves and regulated by means of a ball valve (figure 1). Table 1 shows the characteristics and the specifications of the experimental set up and the specifications the 3 beds. The water used to develop the biofilm came from the potable water distribution network (Medellín, Colombia).
Biocide application system
The biocide application was carried out in an independent unit (figure 2) designed to assemble the pipes once the biofilm had formed. By means of a pump the biocide suspension was recirculated throughout the bed from the biocide solution storage tank. A butterfly valve was used to regulate the biocide flow in order to work under the same hydraulic conditions as the system worked when the biofilm was formed.
Reagents and Analytical methods
The following reagents were used to conduct assays for biofilm removal; Hydrogen peroxide (H2O2,Protokimica35%), Copper sulphate (CuSO4, Merck 249.68 g/mol), Hydrochloric acid (Protokimica 1.0 M) and Sodium hydroxide (Protokimica1.0 M).
For planting microbiological was used an ultrasound scanner (Elma, Ultrasonic LC 60 H). The determination of hydrogen peroxide and copper were performed using the according to the APHA(AWWA - APHA, 1998), 1998 by iodometric method and atomic absorption equipment (BBC 932 plus), respectively (Clesceri; et al., 1998).
Experimental design
• Biofilm formation
Tap water flowed into the system for 20 days, until the formation of a stable biofilm.
• Biocide application
After 20 days, each bed was dismantle done by one. Pipes were taken to the GDCON laboratory and were placed in the module bed of the biocide recirculation system. In each experiment two of the reactors were removed and were used for biofilm determination at time 0.Each biocide solution was freshly prepared in the recirculation system storage tank; biocides were used in three different concentrations (Schmida; et al., 2004).: 200, 1000 and 12000 mg/L for H2O2, and 0.2, 0.6, 1.0 mg/L for the CuSO4. Experiments were carried out in triplicate. Hydrogen peroxide and copper sulfate were selected as biocides because they have been used commercially as disinfectants and in water purification treatments as well as because the former is an oxidizing biocide and the latter is a non-oxidizing biocide. After the solution was prepared, the pH was adjusted to a value between 6.5 -7.0 with either Hydrochloric acid or Sodium hydroxidein order to have a pH similar to that in the potable water distribution system. The biocide solution was recirculated for 60 minutes.Every 15 minutes a water sample was taken to analyze the biocide decay and the 2 reactors were removed for biofilm analysis. After removing the pipes, the spheres were washed (NaCl 0.9%) to remove planktonic cells(Tachikawa & Tezuka, 2005) that could interfere with the results by contributing to a greater amount of bacteria. A container with 15 mL of NaCl 0.9% and the washed spheres were placed in an ultrasound scanner for 3 minutes in order to allow biofilm detachment, in accordance with the procedure of the European Biofilm Workgroup.
Results and discussion
Hydrogen peroxide results
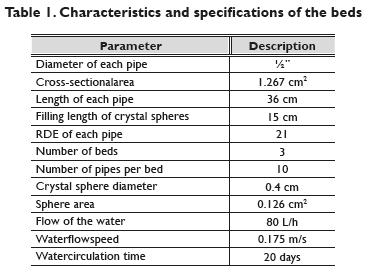
Figure 3 shows that decay in heterotrophic levels increases as contact time with the 3 hydrogen peroxide concentrations of 200, 1000 and 12000 mg/L increases. The heterotrophic bacteria count decreased from 1.28 x 104 CFU/cm2 to 4.40 x 103 CFU/cm2 (68.4% of removal) with 200 mg/L H2O2. It diminished to 1.03 x 103 CFU/cm2 (91.6% of removal) with 1000 mg/LH2O2 and went to 2.33 x 102 CFU/cm2 (98.1% of removal) with 12000 mg/LH2O2. These decreases were achieved after an average time of 60 minutes. The 12000 mg/L concentration significantly lowered the colony forming units without exceeding the recommended hydrogen peroxide concentration for human health (< 30000 mg/L according to the EPA) (Grant, 1986, Ellenhorn; et al., 1997). However, for cost and efficiency reasons 1000 mg/LH2O2 would be the best option. In addition, the objective is to decrease the biofilm, not to remove it completely. Even though with 200 mg/L the removal percentage is a little low, it can still used effectively in drinking water distribution networks and at a lower cost.
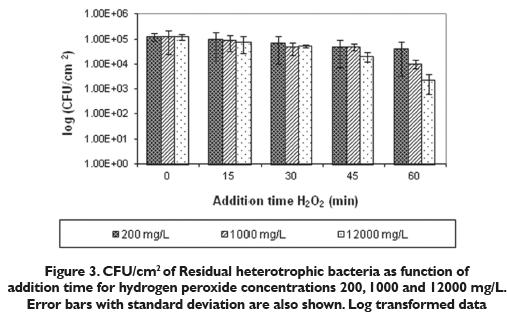
In 60 minutes average decays of 179, 862 and 8972 mg/L H2O2 were achieved (figure 4) from initial average concentrations of 200,1000 and 12000 mg/L respectively. The high residual disinfectant level indicated ensuresa good degree of complementary disinfections despite the release of attached microorganisms. This is an important issue because all biofilm treatments need to be effective against detached cells in order to prevent a secondary colonization downstream.
A positive correlation exists between microbial counts and H2O2 decay, as seen with the 12000 mg/L H2O2, concentration, which removed more heterotrophic bacteria and, where higher hydrogen peroxide consumption was detected. Nevertheless, it is important to clarify that it is not possible to say that all the H2O2 consumption in this experiment was as a result of its reaction with bacterial cells. H2O2 can react with all the organic substances in the system. The H2O2 percentage that disappeared due to the reaction with the other components is not known.
Previous to the beginning of each experiment the pH was measured. Values lower than 6.5 can cause the corrosion of tanks and pipes, and further more do not abide by most legal regulations. With 1000 and 12000 mg/L it was necessary to perform an initial adjustment of pH, where as with 200 mg/L no adjustment was necessary.
Copper sulfate results
Although hydraulic and experimental conditions were equivalent, each colonization and biofilm event was carried out at different times. In the environment in this study tap water did not present considerable variations in chemical and bacteriological quality. Nevertheless, the water flowing during each colonization event was not identical and the qualitative composition of the biofilm may have been different.
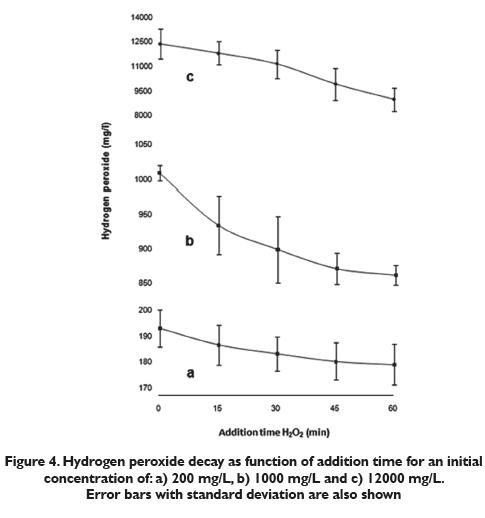
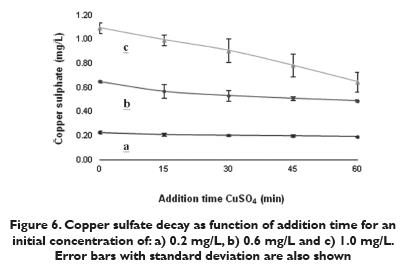
Figure 5 shows a diminution of heterotrophic levels as the contact time increases with 0.2, 0.6 and 1.0 mg/L of copper sulfate concentrations. After 60 min the heterotrophic bacteria count decreased from 1.85 x 105 CFU/cm2 to 1.22 x 105 CFU/cm2 (34.2% of removal) with 0.2 mg/L CuSO4. It diminished to 1.07 x 105 CFU/cm2 (45.8% of removal) with 0.6 mg/L CuSO4 and went to 8.79 x104CFU/ cm2 (50.8% of removal) with 1.0 mg/L CuSO4. Although the count was reduced, the results were not as good as those for the hydrogen peroxide. This is probably a result of the non oxidizing nature of the biocide. According to the Colombian normativity 2115 of 2007, the maximum amount of copper (II) that can be present in potable water is 1.0 mg/L, indicating that the maximum amount of copper sulfate is 2.0mg/L. The fact that residual copper (II) remain sunchanged in water supply networks, while hydrogen peroxideis rapidly destroyed suggests that the latter can be usedin higher concentrations for the removal of biofilm. In 60 minutes an average CuSO4 decay of 0.19mg/L,0.45mg/L and 0.65 mg/L (figure 6) occurred using the initial concentrations 0.20 mg/L, 0.60 mg/L and 1.00mg/L respectively. With the three copper sulfate concentrations, a large amount of residual material remained, as it did with the hydrogen peroxide. Never the less, the decay of hydrogen peroxide was much greater in spite of the smaller CFU/cm2 decay.
Biofilm removal percentages (35%, 43%, and 53%) with the three concentrations (0.2, 0.6 and 1.0 mg/L) of copper sulfate were much lower compared with those obtained using hydrogen peroxide. These results show the lack of efficiency of copper sulfate at eliminating heterotrophic bacteria with the three concentrations used in the short contact time evaluated. When using 0.2 mg/L of copper sulfate, it was not necessary to adjust the pH value. Nevertheless, with 0.6 and 1.0 mg/L of copper sulfate the pH was adjusted to 6.5 so all the experiments would be done with a similar pH and the influence of this important variable would be prevented. The results show that the optimum strategy for biofilm removal in drinking water pipes should be based on the use of oxidizing biocides, such as H2O2. The contact time should be short, but no shorter than 1 hour. Although the use of copper may be useful in some circumstances, such as Legionella control in hot water distribution systems in big buildings, its use not is appropriate for biofilm removal in drinking water pipe systems.
Conclusions
- It was demonstrated that hydrogen peroxide is a biocidal agent that acts strongly on bacterial cells, seen by the fact that in the three concentrations used the heterotrophic bacteria count decreased.
- The best biofilm removal results were obtained with an average concentration of 12000 mg/L for hydrogen peroxide and 1.0 mg/L for copper sulfate, in 60 minutes of contact time.
- In a contact time of 60 minutes the heterotrophic bacteria count decreased to 2.33 x 102 UFC/cm2 when using 12000 mg/L of hydrogen peroxide and to 8.79 x 105 UFC/cm2 when using 1.0 mg/L of copper sulfate.
- When using H2O2 the largest removal percentage was 98% and the smallest percentage was 68%, with average concentrations of 12000 mg/L and 200 mg/L respectively. Such large removals were not found using copper sulfate whose maximum removal percentage was 53% and the minimum was 35% with concentrations of 1.0 and 0.2 mg/L respectively.
Acknowledgements
The authors wish to thank to Andreas Nocker and the Center for Biofilm Engineering (CBE) at Montana State University-Bozeman for their valuable contributions in the revision of this article.
References
Costerton, J. W.; et al. (1995). Microbial Biofilms. Annual Reviews of Microbiology, 49, 711-745. [ Links ]
Codony, F.; et al. (2002). Effect of chlorine, biodegradable dissolved organic carbon and suspended bacteria on biofilm development in drinking water systems. Basic Microbiology, 42(5), 311-319. [ Links ]
Flemming, H. C. (2002). Biofouling in water systems-cases, causes and countermeasures. Applied Microbiolology Biotechnology, 59(6), 659. [ Links ]
Stopforth, J. D.; et al. (2002). Biofilm formation by acid-adapted and nonadapted Listeria monocytogenes in fresh beef decontamination washings and its subsequent inactivation. Journal of Food Protection, 65(11), 1717-1727. [ Links ]
Flemming, H.C. (1993). Biofilms and environmental protection. Water Science and Technology, 27, 1-10. [ Links ]
Lechevallier, M. W.; Cawthon, C. D. & Lee, R. G. (1988). Factors promoting survival of bacteria in chlorinated water supplies. Applied Environmental Microbiology, 54, 649-654. [ Links ]
Camper, A. K. (1994). Coliform regrowth and biofilm accumulation in drinking water systems. In: Geesey G. G, Lewandowski Z, Flemming H. C. (Eds.) Biofouling and Biocorrosion in Industrial Water Systems. 91-105. [ Links ]
Ndiongue, S.; Huck, P. M. & Slawson, R. M. (2005). Effects of temperature and biodegradable organic matter on control of biofilms by free chlorine in a model drinking water distribution system. Water Research. 39(6), p. 953-964. [ Links ]
Delanoue, A.; et al. (1997). Effect of pipe materials on biofilm growth and deposit formation in water distribution systems. Procceedings of WQT. Washington: AWWA. p. 399-409. [ Links ]
Codony, F.; Domenico, P. & Mas, J. (2003). Assessment of bismuth thiols and conventional disinfectants on drinking water biofilms. Journal of Applied Microbiology, 95, 288-293. [ Links ]
Morató, J. (2001). Biofilm formation and health risks in water distribution systems. Ph. D.thesis. Autonomous University of Barcelona: Bellaterra. [ Links ]
American Public Health Association - Apha. American Water Works Association - AWWA. (1998). Standard methods for examination of water and wastewater. 16th ed. Washington: APHA-AWWA. [ Links ]
Clesceri, S.; et al. (1998). Standard Methods for the Examination of Water and Waste-Water. 20th ed. Washington, D.C.:American Public Health Association. [ Links ]
Schmida, T.; et al. (2004). Investigation of biocide efficacy by photoacoustic biofilm monitoring. Water Research, 38, 1189-1196. [ Links ]
Tachikawa, M. & Tezuka, M. (2005). Evaluation of some halogen biocides using a microbial biofilm system. Water Research, 39, 4126-4132. [ Links ]
Grant, W. M. (1986). Toxicology of the Eye. 3rd ed. Springfield, IL: Charles C. Thomas Publisher, 493p. [ Links ]
Ellenhorn, M. J.; et al. (1997). Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore: Williams & Wilkins, 2047p. [ Links ]