Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
International Journal of Psychological Research
Print version ISSN 2011-2084
int.j.psychol.res. vol.6 no.spe Medellín Oct. 2013
Genetic and neural relationships underpinning impulsivity
Relaciones neuronales y genéticas que apuntalan la impulsividad
Nichol M. L. Wonga,b and Tatia M. C. Leea,b,c,*
a Laboratory of Neuropsychology, The University of Hong Kong, Hong Kong, China.
b Laboratory of Cognitive Affective Neuroscience, The University of Hong Kong, Hong Kong, China.
c The State Key Laboratory of Brain and Cognitive Sciences, The University of Hong Kong, Hong Kong, China.
* Corresponding author: Prof. Tatia M.C. Lee, Rm 656, Laboratory of Neuropsychology, The University of Hong Kong, Pokfulam Road, Hong Kong, Tel: (852) 3917-8394, Fax: (852) 2819-0978, Email: tmclee@hku.hk
Received: 30-09-2013-Revised: 16-10-2013-Accepted: 22-10-2013
ABSTRACT
Impulsivity refers to acting without forethought. It can be detrimental to daily social functioning and interaction, and is significantly implicated in several clinical conditions, e.g. violence and addiction. Evidence for the neural underpinnings of impulsivity from both healthy and clinical populations, integrated with the findings from genetic studies on the same topic, lend important insight into a neurobehavioral model of impulsivity. In this review, disinhibition and impulsive decision-making in the impulsivity construct are covered. Recent behavioral and imaging-genetic studies on the topic will also be reviewed and discussed. Findings from neuroimaging studies, clinical studies, and genetic studies converge to provide a better understanding of individual differences on the continuum. Future research efforts should continue to focus on the association approach to identify relevant neural-behavioral correlations in order for elucidating the impact from genes through neural to behavioral phenotypes. These potential findings, when being incorporated with physiological and immunological measures, would not only hasten understanding of impulsivity, but guide interventions development for ameliorating maladaptive social/psychological functioning disorders underpinned by it.
Key Words: Impulsivity, disinhibition, decision making, social functioning, risk, imaging-genetic studies, candidate genes.
RESUMEN
La impulsividad se refiere al actuar sin prudencia. Puede ser perjudicial para el funcionamiento y la interacción social, y está significativamente implicada en varias condiciones clínicas, como por ejemplo la violencia y la adicción. Evidencia para el apuntalamiento neuronal de la impulsividad de tanto la población sana como la clínica, integrada con los hallazgos de los estudios genéticos en el mismo tema, dan conocimiento importante dentro de un modelo neuro-conductual de la impulsividad. En esta revisión, la desinhibición y la decisión-acto impulsivo en el constructo de la impulsividad son abordados. Estudios conductuales y de imagen genética sobre el tema serán también revisados y discutidos. Hallazgos en estudios de neuro-imagen, estudios clínicos y estudios genéticos convergen para producir un mejor entendimiento de las diferencias individuales en el continuo. Los esfuerzos de la investigación futura deberían continuar apuntando al enfoque de asociación para identificar correlaciones neuro-conductuales relevantes parar elucidar el impacto de los genes a través de fenotipos desde neurales hasta conductuales. Estos descubrimientos potenciales, cuando son incorporados con medidas psicológicas e inmunológicas, no estarían sólo impulsando el entendimiento de la impulsividad, sino que también guían las intervenciones de desarrollo para mejorar desordenes de funcionamiento social/psicológico inadaptado apuntalados por esta.
Palabras Clave: Impulsividad, desinhibición, toma de decisiones, funcionamiento social, riesgo, estudios de imagen genética, genes candidatos.
1. INTRODUCTION
Social neuroscience is an interdisciplinary field devoted to understanding the biological underpinnings of social cognitive processes and behaviors by using concepts and tools from neuroscience and neuropsychology. Research on the field has been expanding rapidly because of the advent of neuroimaging techniques. Among the many constructs under investigation in the field, impulsivity has been drawing much attention because it is fundamental to adaptive social functioning as well as to psychological health.
Impulsivity generally refers to acting without forethought (Cardinal, 2006). It can be seen as a set of heterogeneous suboptimal behaviors along two dimensions. One dimension reflects disinhibition, such as the inability to inhibit immediate gratification for a delayed larger reward, or to inhibit a prepotent response, distractions, or impulses; whereas the second dimension reflects impulsive decision making, such as making supraoptimal risky decisions without sufficient consideration (Bevilacqua & Goldman 2013). Research on the impulsivity construct has important implications, as impulsive behaviors greatly influence one’s social life; its adverse outcomes can include delinquency, antisocial behaviors, suicide attempts, aggression, and crimes (Flory et al., 2006; John, Caspi, Robins, Moffitt, & Stouthamer-Loeber, 1994; Luengo, Carrillo-de-la-Pena, Otero, & Romero, 1994; Tremblay, Phil, Vitaro, & Dobkin, 1994).
The discovery of the relationship between the brain and impulsivity can be dated back to the incident of Phineas Gage, ‘the American Crowbar Case,’ in the 19th century, when Phineas Gage was a railroad worker and a large steel rod penetrated through his head in an accident. His left frontal lobe was damaged and he was reported to have significant changes in personality. As described by his co-workers and friends, Phineas had changed profoundly from a polite gentleman to a very irritable and impulsive person after the crowbar accident. This dramatic change in his personality and increased impulsiveness which accompanied his frontal brain injury provoked scientists to start questioning whether a linkage exists between the frontal region of the brain and our impulsivity construct. With on-going improvement in neuroimaging technology, scientists have been investigating dysfunctional impulse regulation in relation to brain lesions and other clinical conditions characterized by high impulsivity. The significance of impulsivity in psychopathology catalyzes the increased research in how variations in impulse control in healthy individuals are related to their brain. As it will be described below, evidence from people with traumatic brain injury (TBI) and disorders such as substance dependence converge with the findings from the healthy population, suggesting that high impulsiveness is mainly associated with our frontal region, in particular the dorsolateral prefrontal cortex (DLPFC), orbitofrontal cortex (OFC), anterior cingulate cortex (ACC), and insula. On the other side, recent genetic research efforts have started to use the imaging-genetic approach. Scientists have started to investigate associations between genotype and the neuroanatomical characteristics and MRI activation patterns of impulsiveness as endophenotypes, apart from investigating the genotype-behavior associations. The imaging-genetic approach is informative in how genetic variation mediates neural mechanisms on individual differences in impulsiveness and their clinical risks of having impulsivity-characterized disorders, bringing benign implication on therapeutic purposes.
This review paper summarizes the findings on disinhibition and impulsive decision-making collected from lesion and behavioral studies on clinical populations, as well as neuropsychological and neuroimaging studies on healthy populations. The goal is to address the converging evidences on how different areas in the frontal region are responsible for our impulsivity. Genetic studies on the topic were reviewed to explore the commonly associated genetic variants reported. Lastly, some recent imaging-genetic studies on impulsivity were reviewed, and the benefits of using this approach in social cognitive neuroscience were discussed.
2. NEUROPSYCHOLOGICAL AND NEUROIMAGING EVIDENCES
2.1. Lesions and psychiatric disorders
Maladaptive risk-taking and impulse control problems are consistently observed in a wide range of clinical conditions. In particular, patients suffering from TBI are often reported to be highly impulsive, leading to slow progression in rehabilitation (McAllister, 2008). Studies conducted in TBI patients involved using a temporal discounting paradigm in which patients were asked to choose between immediate smaller reward and larger reward received later, so that their subjective value of temporal rewards could be inferred from their choices. In general, TBI patients’ reward-choice behaviors were different from the control as they were discounting more (Rochat et al., 2010; Rochat, Beni, Annoni, Vuadens, & Van der Linden, 2013; Wood & McHugh, 2013) and Newcombe and colleagues (2011) have also found that TBI patients tended to place more early bets, and such impulsive behaviors were associated with their abnormalities in bilateral OFC, insula, and caudate.
Patients, who fall into some psychiatric disorder categories including Substance Use Disorders, are also reported to have high impulsivity. The temporal discounting paradigm was used in Heil, Johnson, Higgins, & Bickel’s study (2006) to measure cocaine users’ impulsiveness, and in another study (Ohmura, Takahashi, & Kitamura, 2005) to measure cigarettes smokers. In both studies, substance users were found to have higher discounting rate for reward in comparison to healthy controls, suggesting that they had higher preferences for immediate gratification as a consequence of high impulsiveness. Heroin users also had difficulties in controlling their impulses and were disinhibited in comparison with matched healthy controls (Lee et al., 2005; Lee & Pau, 2002). By using event-related fMRI scanning, abstinent heroin users were also found to exhibit lower activation in ventral lateral frontal regions, left ACC, and left parietal region than controls during inhibition of prepotent responses (Lee et al., 2005), which was attributed to be associated with their weak inhibitory control.
Impulse Control Disorders, as they are called, are also characterized by maladaptive control of impulses. In one of our studies, pathological gamblers’ trait and state impulsivity were assessed (Lai, Ip, & Lee, 2011). Trait impulsivity refers to an enduring impulsive personality profile across situations, and state impulsivity refers to situational, short-term impulsiveness. Their trait impulsivity was measured using Barratt Impulsiveness Scale (BIS; Patton, Stanford, & Barratt, 1995), and their state impulsivity in the cognitive domain and emotional domain were both measured using Stroop test (Lee & Chan, 2000) and Emotional Conflict Task (ECT; Etkin, Egner, Peraza, Kandel, & Hirsch, 2006) respectively. Gamblers were only found to score higher in BIS, suggesting that they have higher trait but not state impulsivity and our findings on emotional state impulsivity were also supported by another study (Fishbein et al., 2005). The fMRI literature on the topic has generally agreed that pathological habits in gambling were associated with ventral striatum and posterior OFC, showing an abnormal sensitivity to reward (e.g. Balodis et al., 2012; Sescousse, Barbalat, Domenech, & Dreher, 2013). There were also other studies suggesting that gambling habits were partly due to disinhibition, associated with their DLPFC, inferior frontal, and ACC (e.g. van Holst, van Holstein, van den Brink, Veltman, & Goudriaan, 2012).
Schizophrenics are characterized by the irresistibility of immediate gratification over delayed larger reward (Heerey, Bell-Warren, Gold, 2008). Our laboratory has also conducted a study on schizophrenic males to assess their risky decision-making qualities (Cheng, Tang, Li, Lau, & Lee, 2012). Results pointed to the notion that safer options were more preferable by schizophrenics in conjunction with their intact sensitivity to punishment. Their suboptimal risky choices could be interpreted as difficulties in delaying the immediate smaller reward or as deficiency in utilizing reward information on later decisions. Studies have suggested that ACC and OFC (Schiffer et al., 2010) were mediating impulsivity in schizophrenia, with their frontostriatal system often appeared abnormal (Barch & Dowd, 2010).
From the abovementioned studies, abnormalities in frontal regions were reported in lesions and disorders with a strong sign of impulsivity. Specifically, DLPFC, OFC, and ACC were reported to have structural and functional deviations from healthy controls, where OFC and insula were more associated with impulsive risky decisions with respect to reward, and DLPFC, and ACC associated with impairments in inhibitory control.
2.2. Individual differences
Impulsivity is not an all-or-none concept but one that lies on a continuum of intensity. Indeed, there are large individual variations within the healthy population. As in below, we will present neuropsychological and neuroimaging findings on healthy individuals and summarize factors that can contribute to the diversity of impulsiveness.
Neural activities underlying risk-taking behavioral patterns were observed in one of our fMRI studies focusing on age-difference. Previous literature has suggested that elderly needs more time to make decisions and is often more conservative (Deakin, Aitken, Robbins, & Sahakian, 2004). Such differential behavioral patterns between age group may be due to underlying differences in the associated neural correlates. Indeed, OFC were consistently found to be involved in risky decision-making, and the tendency of the elderly to make more conservative choices could be attributed to their stronger activation in right insula (Lee, Leung, Fox, Gao, & Chan, 2008).
Brain activation differences between healthy volunteers of high and of low trait impulsivity were also observed (Lee, Chan et al., 2008). Participants of higher trait impulsiveness had greater activation in the right insula, left OFC, and bilateral parietal regions during risk-taking, confirming the association between impulsivity and OFC and insula. Findings also suggested that impulsive people would recruit parietal regions to enhance their impulse control. In another voxel-based morphometry study, the delay discounting paradigm was used to distinguish individuals with higher impulsivity, and their regional brain volumes were compared with that of lower impulsivity (Yu, 2012). It was found that impulsive individuals had smaller white matter (WM) volumes in the prefrontal cortex, but larger WM volumes in the right parahippocampus and hippocampus. The involvement of the parahippocampus and hippocampus suggested that impulsive decisions can be due to the weaker ability to prospect (Luhmann, Chun, Yi, Lee, & Wang, 2008).
ACC and DLPFC were also found to associate with risky decision-making in other studies (De Martino, Kumaran, Seymour, & Dolan, 2006; McClure, Laibson, Loewenstein, & Cohen, 2004) and could be related to mood. People in an induced negative (or depressed) mood were more conservative in making risky-decisions (Yuen & Lee, 2003), and this effect was related to the reduction of brain activities in ACC and DLPFC (Drevets & Raichle, 1998). DLPFC was also suggested to be crucial in controlling impulses towards reward gratification in other studies (e.g. McClure et al., 2004), and a traditional Chinese medicine recipe that promotes advantageous risk-taking was reported to facilitate stronger activation in DLPFC in improved risk-takers (Lee, Guo, et al., 2009).
The frontostriatal pathway (mainly the right) was consistently reported to underlie behavioral inhibition, and striatum was also suggested to influence impulsive decision-making by facilitating learning from reward and punishment processes (Dobryakova & Tricomi, 2013). It is also noted that inferior and medial prefrontal cortices and subthalamic nucli have also been reported to be activated in impulsive tasks (Congdon & Canli, 2008; Paulus, Rogalsky, Simmons, Feinstein, & Stein, 2003, Xue et al., 2009). In particular, the inferior frontal cortices were suggested to control behavioral inhibition whereas the subthalamic nucleus was responsible for inhibiting motor responses.
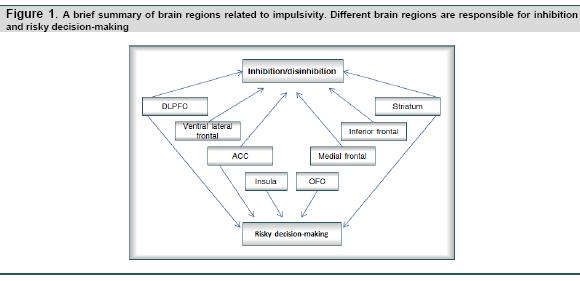
Neuroimaging evidences in the healthy population are suggesting that OFC and insula are crucial in contributing to impulsiveness, probably through their function on evaluating potential risk with reference to goals, whereas DLPFC helps by inhibiting impulses towards immediate gratification. Other regions including ACC, striatum, hippocampal, and parietal have also been reported to be involved in impulsiveness. Findings in healthy individuals generally converge to findings from lesion studies and clinical populations (see Figure 1 for summary of brain regions related to impulsivity); though the interpretation of neuroimaging results should be done with caution because of the complications brought by the multidimensional characteristic of impulsivity. For instance, findings in Horn, Dolan, Elliott, Deakin, & Woodruff’s study (2003) have revealed that impulsivity captured by different self-report measures could correlate positively with activations in different regions using the same inhibition paradigm; and Brown, Manuck, Flory, & Hariri (2006) have also revealed that impulsivity captured by the same self-report measure could correlate with different regions if different inhibition paradigms were used. These complications arise from the divergence of measurements on a multidimensional impulsivity construct and would complicate our interpretation of data.
2.3. Gender differences
Males were viewed as more impulsive by public and indeed previous literature has suggested that males tend to engage in more frequent risk-taking behaviors and sensation seeking than females (Rosenblitt, Soler, Johnson, & Quadagno, 2001). In one of our studies, females were also found to choose more safe choices after losing points in a risky decision-making context, with stronger activations in bilateral OFC and right insula (Lee, Chan, Leung, Fox, & Gao, 2009). Correlation analyses revealed a significant positive relationship between the insula and risky (and riskier-then-risky) responses in females, with the rate of risky responses after feedback associating with the OFC. The more activated right insula in conservative females was attributed to insula’s function of risk anticipation (Sanfey, Rilling, Aronson, Nystrom, & Cohen, 2003), whereas OFC activities could reflect the regulation of responses with updated information (Wallis, 2007). Gender differences in the neural correlates of inhibition were also observed in other fMRI studies. Greater activations in brain regions including bilateral medial frontal and cingulate cortices, globus pallidus, thalamus, and parahippocampal gyrus were observed in male, implying more neural resources were required for male to inhibit (Li, Huang, Constable, & Sinha, 2006). Successful inhibits in males also activated frontal regions including the right inferior frontal cortex, and another fMRI study (Liu, Zubieta, & Heitzeg, 2012) has also found that males exhibited stronger activations in ACC but less activation in middle temporal region during inhibition.
3. IMPULSIVITY AND GENES
Within the past decade, genetic studies have grown enormously and have drawn a lot of attention from scholars and the public. In any individual, there are variations in the nucleotides of the genomes that can cause individual differences in terms of traits, phenotypes, or risk of developing certain diseases. Inheritance studies, genetic association studies, and genetic knockout animal studies used to be the common methodologies until the genome-wide association study (GWAS) approach became popular around the declaration of completion of Human Genome Project at the start of 21st century. The ability to scan the entire genome would allow hypothesis-free identification of loci in relation to disorders and other phenotypes. GWA would detect small effect sizes of associations between independent single-nucleotide polymorphisms (SNPs) and major diseases by comparing the DNA of a large sample of clinical cases with controls to determine whether any particular type of genetic variants is more prevalent in the clinical sample. Associations between SNP genotyping and behavioral phenotypes were vigorously investigated.
On the other hand, more recent advances in genetics and neuroimaging have facilitated collaboration between the two disciplines. By marrying the genetic information and neuroimaging data of same subjects, there is another imaging-genetic approach looking at how genetic variation mediates neural system and related to individual differences in complex behaviors. In general, this genetic association analytic approach involves the identification of a candidate gene which is hypothesised to be associated with a particular behavioral phenotype at first. The SNP genotyping of the candidate genes is then compared to the endophenotypes. In imaging-genetics, the endophenotypes will usually be neuroanatomical characteristics and activation pattern of a particular region or network.
Below we will discuss several candidate genes that were suggested to associate with impulsivity through its implication in neurotransmitters, in particular serotonin and dopamine. From genetic studies and neuroimaging approaches, we will discover that genetic variance mediates our impulsiveness through its influence in brain regions and circuitries.
3.1. TPH2
Trytophan hydoxylase (TPH) is an enzyme that facilitates the rate-limiting synthesis of neurotransmitter serotonin (5-HT). Lower serotonin levels were consistently reported to relate to high impulsiveness (e.g. Pattij & Vanderschuren, 2008) and activations of 5-HT receptor can inhibit 5-HT release, leading to reduced serotonergic neurotransmission and impulsive behaviors (Winstanley, Theobald, Dalley, Cardinal, & Robbins, 2005). Depletion of tryptophan was also reported to associate with more impulsive choices (Rogers et al., 2003), hence, genetic variations in the TPH gene could influence impulsivity through its regulation of 5-HT. Juhasz and co-workers (2010) have recruited a sample of more than 1000 participants to conduct an association-analysis between their TPH2 gene and their performance on gambling trials, in which a choice between a higher chance of a small ‘win’ and a lower chance of bigger ‘win’ has to be made in each trial. Within the SNP genotypes, they have revealed that the minor alleles in two of the SNPs (rs6582078, rs1352250) were associated with more risky choices. In another study, Stoltenberg and colleagues (2006) have also examined the association between TPH2 and impulsiveness in the form of response inhibition. The Stop-signal Task (SST) was delivered to participants carrying different allelic combinations of TPH2 (T/T, T/C, and C/C). By comparing participants’ indices calculated from the inhibition task, they have found that the T/T genotype was associated with less inhibitory control in males and C/C associated with higher inhibitory control in females, indicating influence from the TPH2 genotypes can be different between genders.
Moreover, manipulation of 5-HT within the brain by TPH have suggested that serotonergic neurotransmission could modulate the prefrontal regions that were highly related to inhibitory control (Morgan et al., 2007). One recent study has adopted the imaging-genetics approach to investigate two common TPH2 6-locus haplotypes (i.e. sets of associated SNPs) in relation to inhibition and decision-making (Kennedy et al., 2012). Their findings revealed that increased number of copies of TPH2 yin-haplotype could be associated with inefficient and abnormal responses to cognitive control demand from brain regions including dorsal ACC and right inferior frontal gyrus. In addition, their functional connectivities with other regulatory regions would be compromised.
3.2. SLC6A transporter
SLC6A3 is a dopamine transporter gene responsible for the transfer of dopamine back to presynaptic neurons, which dopamine was involved in the regulation of responses to reward, and an abnormal dopaminergic system could result in impulsive behaviors because of the reduction of sensitivity to reward and punishment (Bevilacqua & Goldman, 2013). About 400 healthy adults were recruited and the association between SNPs genotyping of SLC6A3 and the participants’ inhibitory control were examined (Cummins et al., 2012); which the authors reported that allelic variations in rs37020 and rs460000 were related to participants’ inhibitory behaviors. In addition, the SLC6A3-3’untranslated region-variable numbers of tandem repeats (VNTR) alleles with 10 repeats were also reported to predict pathological gambling (Comings et al., 2001). In another study, Roiser, Roger, Cook, & Sahakian (2006) have investigated the decision-making of participants with respect to their 5-HT linked polymorphic region (5-HTTLPR) genotypes (L/L, L/S, S/S) in the SLC6A4 gene. They found that both S/S and L/S carriers of healthy individuals attended more to the differences between high and low probabilities of winning and S/S carriers chose fewer risky choices in the risk-choice task because of greater reflection. In contrast, Walderhaug, Herman, Magnusson, Morgan, & Landrø (2010) have measured participants’ attentional impulsiveness using Continuous Performance Task, and found that individuals with S/S genotype were more attentionally impulsive (i.e. could not sustain their attention for long) than those with L/S and L/L genotype. Sonuga-Barke and colleagues (2011) have also suggested that S-allele carriers were more avoidant to delayed reward compared to immediate gratification. Hence, SLC6A4 is also attributed to be associated with different aspects of impulsiveness through its impact on serotonin, yet how the allelic combinations related to the impulsive behavioral phenotype was not fully uncovered.
Whelan and co-workers (2012) have sought to link the inhibitory control in early adolescence with the inhibitory networks, and to determine the influence of genetics on these brain networks. They have identified a number of independent functioning networks (e.g. bilateral frontal, basal ganglia, OFC) corresponding to their inhibitory control and have observed apparent phenotypic differences in activation patterns. As per genetic findings, variation in the SLC6A2 gene was found to be related to the right frontal network when participants inhibited their responses. Furthermore, activity in anterior and superior frontal regions and superior medial gyrus during inhibition was suggested to associate with SLC6A3 (Cummins et al., 2012). This is in line with the genetic findings, which suggested that the SLC6A transporter gene family mediated impulsivity through the frontal regions.
3.3. DRD2
DRD2 gene in the dopamine receptor gene family was reported to be associated with impulsivity through the encoding of D2 dopamine receptors responsible for dopamine levels. A study conducted by Colzato, van den Wildenberg, & Hommel (2010) has investigated the association between SNP rs6277 of DRD2 gene in healthy subjects and their performance in SST. They have found that C/C carriers were better than T-allele carriers (T/T, T/C) in inhibiting their response, and this was due to the higher density of D2 receptors in extrastriatal regions in C/C carriers. T-allele carriers would have weakened inhibitory pathways because of higher levels of striatal dopamine, and therefore, would have reduced control over inhibition.
The preferences for immediate smaller over delayed larger rewards in individuals are also studied using an imaging-genetic approach. DRD2 genotypes including 141C deletion carriers (Ins/Del) and non-carriers (Ins/Ins), and DRD4 VNTR with 7-repeat and VNTR with other repeats were examined by Forbes and colleagues (2009). They have found an association between DRD2 (Ins/Del) genotype and greater ventral striatum activation, and between DRD4 7-repeat and greater ventral striatum activation during their participation in a monetary reward task, suggesting a relationship between DRD4 gene, ventral striatum, and their rate of temporal discounting.
3.4. MAOA
In extreme cases, impulsivity can express as aggression and violent behavior. Monoamine oxidases A (MAOA) involves in the oxidation of several biogenic amines including serotonin and contributes to manipulating the serotonin levels. The VNTR of MAOA gene was studied for association with impulsive-aggressive traits and violent behaviors. MAOA-VNTR alleles with 3 and 4 repeats are more common in the normal population, and it is suggested that males with MAOA-VNTR with 4 repeats have higher activity of MAOA, hence, are less violent (Kim-Cohen et al., 2006); whereas carriers of MAOA-VNTR with 2 repeats were reported to have a two-fold increased probability of committing violent crimes due to more aggressive (Guo, Ou, Roettger, & Shih, 2008).
Functional imaging findings of MAOA-VNTR also resembled these impulsivity studies. Decreased activation in the ACC and decreased ventral prefrontal engagement were observed in MAOA-L carriers, and morphometric changes in OFC and amygdala were observed in women with partial deletions of the MAOA gene (Buckholtz & Meyer-Lindenberg, 2008). Such distinctive gender-specific relationship aligns with the notion that male is of greater vulnerable to MAOA genetic variation. In addition, connectivity analyses revealed that connections between the ventral medial prefrontal cortex and the amygdala were also associated with the MAOA-L allele, and this network predicted increased harm avoidance and decreased reward dependence, further validating the notion that the genetic influence of MAOA on neuroanatomical changes was associated with impulsive aggression.
3.5. COMT
Catechol-O-methyl transferase (COMT) is an enzyme that degrades dopamine and involves in dopamine clearance from synapses, implying its close association with the dopamine levels. Kulikova et al. (2008) in one study has used an aggression inventory which evaluates different dimensions of aggression and violence and found higher physical aggression in COMT H/H carriers than L/L and L/H carriers, but another study conducted on children has found that L-allele carriers were more direct and physically aggressive (Albaugh et al., 2010). In addition, L/L carriers in schizophrenia had a higher risk (Strous, Bark, Parsia, Volavka, & Lachman, 1997) and higher rate of aggressive behaviors (Han, Park, Na, Kee, & Lee, 2004), with more history of violent behavior in the past (Lachman, Nolan, Mohr, Saito, & Volavka, 1997). The reasons for obtaining such contradicting findings regarding the association between the COMT gene’s allelic combinations and aggressive behavior (whether L- or H-allele is the risk allele) were still unclear, but COMT was generally reported to be related to aggression towards others, which such behavior being highly coupled with impulsivity.
Shehzad, DeYoung, Kang, Grigorenko, & Gray (2012) have used a gene-trait interaction approach which might help explain the association between the genetic variation of COMT and impulsiveness. They measured brain activities of a male sample during a multi-source interference task (Bush & Shin, 2006) and found stronger activation in CC in high externalising H/H carriers and low externalising L/L carriers. Their study not only suggested that the externalising trait was mediating the association between COMT gene and the brain, leading to conflicting findings on impulsive-aggression, but more importantly, their study also suggested that the gene-brain-behavior relationship was also mediated by other factors.
The recent imaging-genetic studies have attempted to bridge the link between the major genetic variants in candidate genes and constructs of impulsivity. Findings have suggested that TPH2 gene is related to dorsal ACC, right inferior frontal gyrus and other regulatory brain regions, whereas the SLC6A transporter gene family influences inhibitory control through frontal regions. MAOA genotypes are suggested to cast impact on OFC, ACC, and amygdala with gender-specificity and COMT allelic combinations appear to be more complicated and are suggested to affect prefrontal activities by interacting with other trait factors.
It is also clear that the imaging-genetic approach is a powerful tool to study social cognitive neuroscience. With the advances in neuroimaging, research discoveries have led to a better understanding of the neural mechanisms of social cognitive behaviors. Genetic studies have also highlighted evidence of associations between social cognitive traits and SNPs genotyping. Taking together, social and cognitive behaviors were influenced by the genetic impact on synapse structure and transmission at a genetic level and on network and connectivity (both structural and functional) at neural level. Studies using the imaging-genetic approach are hence suggested to link the genetic, neural, and social cognitive behavioral variations and will definitely benefit our research discoveries and implications.
4. CONCLUSIONS
From the literature reviewed above, it is clear that “impulsivity” is a multifaceted construct underpinned by the influence of both genetic and neural factors. Findings from clinical studies, neuroimaging studies, and genetic studies converge to provide a better understanding of individual differences on the continuum of the intensity of impulsivity presentation. The imaging-genetic approach increases our understanding of the linkage of gene and impulsivity using brain measures as intermediate phenotypes. These studies have begun to characterize networks and pathways conferring individual differences in different aspects of impulsivity. Also, studies have started to investigate common genetic variations conferring risk for symptoms of impulse control disorders and other clinical conditions such as schizophrenia through influence of neural circuitries. The momentum of this approach of research recently increased. Future research efforts should continue to focus on the association approach to identify relevant neural-behavioral correlations, and should include more large-sample studies to elucidate the impact of genes through neural to behavioral phenotypes. In addition, environmental and developmental factors should also be considered. Gender, as one of the factors, are rarely addressed and it is seemingly that male typically more impulsive than female may have basis on both genetic (e.g. TPH2, MAOA) and neural levels, though there is in need of further justification. Future research should address the gender difference in different levels because it may be relevant to the prevalence of impulsive-related psychiatric disorders.
Candidate genes related to impulsivity was suggested to be those that are related to serotonin and dopamine levels, and indeed there could be other internal physiological factors related to different facets of impulsivity. As such, the potential findings, when incorporated with physiological and immunological measures, would lead to the development of a neurobehavioral model of impulsivity that would not only hasten understanding of this construct, but guide intervention development for ameliorating maladaptive social functioning/psychological disorders underpinned by impulsivity.
5. ACKNOWLEDGEMENT
We would like to thank the funding support from the Research Grant Council General Research Fund (HKU747612H). There are no conflicts of interest including any financial, personal or other relationships with persons or organizations for any author related to the work described in this article.
6. REFERENCES
Albaugh, M. D., Harder, V. S., Althoff, R. R., Rettew, D. C., Ehli, E. A., Lengyel-Nelson, T., ... Hudziak, J. J. (2010). COMT Val158Met genotype as a risk factor for problem behaviors in youth. Journal of American Academy of Child & Adolescent Psychiatry, 49(8), 841-849. [ Links ]
Balodis, I. M., Kober, H., Worhunsky, P. D., Stevens, M. C., Pearlson, G. D., & Potenza, M. N. (2012). Diminished frontostriatal activity during processing of monetary rewards and losses in pathological gambling. Biological Psychiatry, 71(8), 749-757. [ Links ]
Barch, D. M., & Dowd, E. C. (2010). Goal representations and motivational drive in schizophrenia: the role of prefrontal-striatal Interactions. Schizophrenia Bulletin, 36(5), 919-934. [ Links ]
Bevilacqua, L. & Goldman, D. (2013). Genetics of impulsive behaviour. Philosophical Transactions of the Royal Society B, 368(1615), 20120380. [ Links ]
Brown, S. M., Manuck, S. B., Flory, J. D., & Hariri, A. R. (2006). Neural basis of individual differences in impulsivity: contributions of corticolimbic circuits for behavioural arousal and control. Emotion, 6(2), 239-245. [ Links ]
Buckholtz, J. W., & Meyer-Lindenberg, A. (2008). MAOA and the neurogenetic architecture of human aggression. Trends in Neurosciences, 31(3), 120-129. [ Links ]
Bush, G., & Shin, L. M. (2006). The Multi-Source Interference Task: an fMRI task that reliably activates the cingulo-frontal-parietal cognitive/attention network. Nature Protocols, 1(1), 308-313. [ Links ]
Cardinal, R. N. (2006). Neural systems implicated in delayed and probabilistic reinforcement. Neural Networks, 19(8), 1277-1301. [ Links ]
Cheng, G. L. F., Tang, J. C. Y., Li, F. W. S., Lau, E. Y. Y., & Lee, T. M. C. (2012). Schizophrenia and risk-taking: impaired reward but preserved punishment processing. Schizophrenia Research, 136(1-3), 122-127. [ Links ]
Colzato, L. S., van den Wildenberg, W. P. M., & Hommel, B. (2010). The genetic impact (C957T-DRD2) on inhibitory control is magnified by aging. Neuropsychologia. Advance online publication. doi: 10.1016/j.neuropsychologia.2013.01.014. [ Links ]
Comings, D. E., Gade-Andavolu, R., Gonzalez, N., Wu, S., Muhleman, D., Chen, C., ... Rosenthal, R. J. (2001). The additive effect of neurotransmitter genes in pathological gambling. Clinical Genetics, 60(2), 107-116. [ Links ]
Congdon, E., & Canli, T. (2008). A Neurogenetic Approach to Impulsivity. Journal of Personality, 76(6), 1447-1484. [ Links ]
Cummins, T. D. R., Hawi, Z., Hocking, J., Strudwick, M., Hester, R., Garavan, H., ... Bellgrove, M. A. (2012). Dopamine transporter genotype predicts behavioural and neural measures of response inhibition. Molecular Psychiatry, 17(11), 1086-1092. [ Links ]
Deakin, J., Aitken, M., Robbins, T., & Sahakian, B. J. (2004). Risk taking during decision-making in normal volunteers changes with age. Journal of the International Neuropsychological Society, 10(4), 590-598. [ Links ]
De Martino, B., Kumaran, D., Seymour, B., & Dolan, R. J. (2006). Frames, biases, and rational decision-making in the human brain. Science, 313(5787), 684-687. [ Links ]
Dobryakova, E., & Tricomi, E. (2013). Basal ganglia engagement during feedback processing after a substantial delay. Cognitive, Affective, & Behavioral Neuroscience. Advance online publication. doi: 10.3758/s13415-013-0182-6. [ Links ]
Drevets, W. C., & Raichle, M. E. (1998). Suppression of regional cerebral blood during emotional versus higher cognitive implications for interactions between emotion and cognition. Cognition & Emotion, 12(3), 353-385. [ Links ]
Etkin, A., Egner, T., Peraza, D. M., Kandel, E. R., & Hirsch, J. (2006). Resolving emotional conflict: a role for the rostral anterior cingulate cortex in modulating activity in the amygdala. Neuron, 51(6), 871-882. [ Links ]
Fishbein, D. H., Eldreth, D. L., Hyde, C., Matochik, J. A., London, E. D., Contoreggi, C., ... Gran, S. (2005). Risky decision making and the anterior cingulate cortex in abstinent drug abusers and nonusers. Cognitive Brain Research, 23(1), 119-136. [ Links ]
Flory, J. D., Harvey, P. D., Mitropoulou, V., New, A. S., Silverman, J. M., Siever, L. J., & Manuck, S. B. (2006). Dispositional impulsivity in normal and abnormal samples. Journal of Psychiatric Research, 40(5), 438-447. [ Links ]
Forbes, E. E., Brown, S. M., Kimak, M., Ferrell, R. E., Manuck, S. B., Hariri, A. R. (2009). Genetic variation in components of dopamine neurotransmission impacts ventral striatal reactivity associated with impulsivity. Molecular Psychiatry, 14(1), 60-70. [ Links ]
Guo, G., Ou, X. M., Roettger, M., & Shih, J. C. (2008). The VTNR 2 repeat in MAOA and delinquent behaviour in adolescence and young adulthood: associations and MAOA promoter activity. European Journal of Human Genetics, 16(5), 626-634. [ Links ]
Han, D. H., Park, D. B., Na, C., Kee, B. S., & Lee, Y. S. (2004). Association of aggressive behaviour in Korean male schizophrenic patients with polymorphisms in the serotonin transporter promoter and catecholamine-O-methyltransferase genes. Psychiatry Research, 129(1), 29-37. [ Links ]
Heerey, E. A., Bell-Warren, K. R., & Gold, J. M. (2008). Decision-making impairments in the context of intact reward sensitivity in schizophrenia. Biological Psychiatry, 64(1), 62-69. [ Links ]
Heil, S. H., Johnson, M. W., Higgins, S. T., & Bickel, W. K. (2006). Delay discounting in currently using and currently abstinent cocaine-dependent outpatients and non-drug-using matched controls. Addictive Behaviors, 31(7), 1290-1294. [ Links ]
Horn, N. R., Dolan, M., Elliott, R., Deakin, J. F., & Woodruff, P. W. (2003). Response inhibition and impulsivity: an fMRI study. Neuropsychologia, 41(14), 1959-1966. [ Links ]
John, O. P., Caspi, A., Robins, R. W., Moffitt, T. E., & Stouthamer-Loeber, R. (1994). The little five: exploring the nomological network of the five factor model of personality in adolescent boys. Child Development, 65(1), 160-178. [ Links ]
Juhasz, G., Downey, D., Hinvest, N., Thomas, E., Chase, D., Toth, Z. G., ... Anderson, I. M. (2010). Risk-taking behavior in a gambling task associated with variations in the tryptophan hydroxylase 2 gene: relevance to psychiatric disorders. Neuropsychopharmacology, 35(5), 1109-1119. [ Links ]
Kennedy, A. P., Binder, E. B., Bowman, D., Harenski, K., Ely, T., Cisler, J. M., ... Kilts, C. D. (2012). A common TPH2 haplotype regulates the neural processing of a cognitive control demand. American Journal Medical Genetics Part B: Neuropsychiatric Genetics, 159B(7), 829-840. [ Links ]
Kim-Cohen, J., Caspi, A., Taylor, A., Williams, B., Newcombe, R., Craig, I. W., & Moffitt, T. E. (2006). MAOA, maltreatment, and gene-environment interaction predicting children's mental health: new evidence and a meta-analysis. Molecular Psychiatry, 11(10), 903-913. [ Links ]
Kulikova, M. A., Maluchenko, N. V., Timofeeva, M. A., Shlepzova, V. A., Schegolkova, J. V., Sysoeva, O. V., ... Tonevitsky, A. G. (2008). Effect of functional catechol-O-methyltransferase Val158Met polymorphism on physical aggression. Bulletin of Experimental Biology and Medicine, 145(1), 62-64. [ Links ]
Lachman, H. M., Nolan, K. A., Mohr, P., Saito, T., & Volavka, J. (1997). Association between catechol O-methyltransferase genotype and violence in schizophrenia and schizoaffective disorder. The American Journal of Psychiatry, 155(6), 835,837. [ Links ]
Lai, F. D. M., Ip, A. K .Y., & Lee, T. M. C. (2011). Impulsivity and pathological gambling among Chinese: Is it a state or a trait problem? BMC Research Notes, 4, 492. [ Links ]
Lee, T. M. C., & Chan, C. C. H. (2000). Stroop Interference in Chinese and English. Journal of Clinical and Experimental Neuropsychology, 22(4), 465-471. [ Links ]
Lee, T. M. C., & Pau, C. W. H. (2002). Impulse control differences between abstinent heroin users and matched controls. Brain Injury, 16(10), 885-889. [ Links ]
Lee, T. M. C., Chan, C. C. H., Leung, A. W. S., Fox, P. T., & Gao, J.-H. (2009). Sex-related differences in neural activity during risk taking: an fMRI study. Cerebral Cortex, 19(6), 1303-1312. [ Links ]
Lee, T. M. C., Leung, A. W. S., Fox, P. T., Gao, J.-H., Chan, C. C. H. (2008). Age-related differences in neural activities during risk taking as revealed by functional MRI. Social Cognitive & Affective Neuroscience, 3(1), 7-15. [ Links ]
Lee, T. M. C., Chan, C. C. H., Han, S.-h., Leung, A. W. S., Fox, P. T., & Gao, J.-H. (2008). An event-related fMRI study on risk taking by healthy individuals of high or low impulsiveness. Neuroscience Letters, 438(2), 138-141. [ Links ]
Lee, T. M. C., Zhou, W.-h., Luo, X.-j., Yuen, K. S. L., Ruan, X.-z., & Wen, X.-c. (2005). Neural activity associated with cognitive regulation in heroin users: a fMRI study. Neuroscience Letters, 382(3), 211-216. [ Links ]
Lee, T. M. Y., Guo, L.-g., Shi, H.-z., Li, Y.-z., Luo, Y.-j., Sung, C. Y. Y., ... Lee, T. M. C. (2009). Neural correlates of Traditional Chinese Medicine induced advantageous risk-taking decision making. Brain and Cognition, 71(3), 354-361. [ Links ]
Li, C.-s. R., Huang, C., Constable, R. T., & Sinha, R. (2006). Gender differences in the neural correlates of response inhibition during a stop signal task. NeuroImage, 32(4), 1918-1929. [ Links ]
Liu, J., Zubieta, J.-K., & Heitzeg, M. (2012). Sex differences in anterior cingulate cortex activation during impulse inhibition and behavioural correlates. Psychiatry Research, 201(1), 54-62. [ Links ]
Luengo, M. A., Carrillo-de-la-Pena, M. T., Otero, J. M., & Romero, E. (1994). A short-term longitudinal study of impulsivity and antisocial behavior. Journal of Personality and Social Psychology, 66(3), 542-548. [ Links ]
Luhmann, C. C., Chun, M. M., Yi, D. J., Lee, D., & Wang, X. J. (2008). Neural dissociation of delay and uncertainty in intertemporal choice. The Journal of Neuroscience, 28(53), 14459-14466. [ Links ]
McAllister, T. W. (2008) Neurobehavioral sequelae of traumatic brain injury: Evaluation and management. World Psychiatry, 7(1), 3-10. [ Links ]
McClure, S. M., Laibson, D. I., Loewenstein, G., & Cohen, J. D. (2004). Separate neural systems value immediate and delayed monetary rewards. Science, 306(5695), 503-507. [ Links ]
Morgan, R., Parry, A., Arida, R., Matthews, P., Daview, B., & Castell, L. (2007). Effects of elevated plasma tryptophan on brain activation associated with the Stroop task. Psychopharmacology, 190(3), 383-389. [ Links ]
Newcombe, V. F. J., Outtrim, J. G., Chatfield, D. A., Manktelow, A., Hutchinson, P. J., Coles, J. P., ... Menon, D. K. (2011) Parcellating the neuroanatomical basis of impaired decision-making in traumatic brain injury. Brain, 134(3), 759-768. [ Links ]
Ohmura, Y., Takahashi, T., & Kitamura, N. (2005). Discounting delayed and probabilistic monetary gains and losses by smokers of cigarettes. Psychopharmacology, 182, 508-515. [ Links ]
Pattij, T., & Vanderschuren, L. J. M. J. (2008). The neuropharmacology of impulsive behaviour. Trends in Pharmacological Sciences, 29(4), 192-199. [ Links ]
Patton, J. H., Stanford, M. S., & Barratt, E. S. (1995). Factor structure of the Barratt impulsiveness scale. Journal of Clinical Psychology, 51(6), 768-774. [ Links ]
Paulus, M. P., Rogalsky, C., Simmons, A., Feinstein, J. S., & Stein, M. B. (2003). Increased activation in the right insula during risk-taking decision making is related to harm avoidance and neuroticism. Neuroimage, 19(4), 1439-1448. [ Links ]
Rochat, L., Beni, C., Annoni, J.-M., Vuadens, P., & Van der Linden, M. (2013). How Inhibition Relates to Impulsivity after Moderate to Severe Traumatic Brain Injury. Journal of the International Neuropsychological Society, 19(8), 890-898. [ Links ]
Rochat, L., Beni, C., Billieux, J., Azouvi, P., Annoni, J. M., & Van der Linden, M. (2010). Assessment of impulsivity after moderate to severe traumatic brain injury. Neuropsychological Rehabilitation, 20(5), 778-797. [ Links ]
Rogers, R. D., Tunbridge, E. M., Bhagwagar, Z., Drevets, W. C., Sahakian, B. J., & Carter, C. S. (2003). Tryptophan depletion alters the decision-making of healthy volunteers through altered processing of reward cues. Neuropsychopharmacology, 28(1), 153-162. [ Links ]
Roiser, J. P., Roger, R. D., Cook, L. J., & Sahakian, B. J. (2006). The effect of polymorphism at the serotonin transporter gene on decision-making, memory and executive function in ecstasy users and control. Psychopharmacology (Berlin, Germany), 188(2), 213-227. [ Links ]
Rosenblitt, J. C., Soler, H., Johnson, S. E., & Quadagno, D. M. (2001). Sensation seeking and hormones in men and women: exploring the link. Hormones and Behavior, 40(3), 396-402. [ Links ]
Sanfey, A. G., Rilling, J. K., Aronson, J. A., Nystrom, L. E., & Cohen, J. D. (2003). The neural basis of economic decision-making in the Ultimatum Game. Science, 300(5626), 1755-1758. [ Links ]
Schiffer, B., Muller, B. W., Scherbaum, M., Forsting, M., Wiltfang, J., Leygraf, N., & Gizewski, E. R. (2010). Impulsivity-related brain volume deficits in schizophrenia-addiction comorbidity. Brain, 133(1), 3093-3103. [ Links ]
Sescousse, G., Barbalat, G., Domenech, P., & Dreher, J.-C. (2013). Imbalance in the sensitivity of different types of rewards in pathological gambling. Brain. Advance online publication. doi:10.1093/brain/awt126. [ Links ]
Shehzad, Z., DeYoung, C. G., Kang, Y., Grigorenko, E. L., & Gray, J. R. (2012). Interaction of COMT Val158Met and externalizing behaviour: Relation to prefrontal brain activity and behavioural performance. NeuroImage, 60(4), 2158-2168. [ Links ]
Sonuga-Barke, E. J. S., Kumsta, R., Scholtz, W., Lasky-Su, J., Marco, R., Miranda, A., ... Farone, S. V. (2011). A functional variant of the serotonin transporter gene (SLC6A4) moderates impulsive choice in ADHD boys and siblings. Biological Psychiatry, 70(3), 230-236. [ Links ]
Stoltenberg, S. F., Glass, J. M., Chermack, S. T., Flynn, H. A., Li, S., Weston, M. E., & Burmeister, M. (2006). Possible association between response inhibition and a variant in the brain-expressed tryptophan hydroxylase-2 gene. Psychiatric Genetics, 16(1), 35-38. [ Links ]
Strous, R. D., Bark, N., Parsia, S. S., Volavka, J., & Lachman, H. M. (1997). Analysis of a functional catechol-O-methyl transferase gene polymorphism in schizophrenia: evidence for association with aggressive and antisocial behavior. Psychiatry Research, 69(2-3), 71-77. [ Links ]
Tremblay, R. E., Phil, R. O., Vitaro, F., & Dobkin, P. L. (1994). Predicting early onset of male antisocial behavior from preschool behavior. Archives of General Psychiatry, 51(9), 732-739. [ Links ]
van Holst, R. J., van Holstein, M., van den Brink, W., Veltman, D. J., & Goudriaan, A. E. (2012). Response inhibition during cure reactivity in problem gamblers: an fMRI study. PLoS ONE, 7(3), e30909. [ Links ]
Walderhaug, E., Herman, A. I., Magnusson, A., Morgan, M. J., & Landrø, N. I. (2010). The short (S) allele of the serotonin transporter polymorphism and acute tryptophan depletion both increase impulsivity in men. Neuroscience Letters, 473(3), 208-211. [ Links ]
Wallis, J. D. (2007). Orbitofrontal cortex and its contribution to decision-making. Annual Review of Neuroscience, 30, 31-56. [ Links ]
Whelan, R., Conrod, P. J., Poline, J.-B., Lourdusamy, A., Banaschewski, T., Barker, G. J., ... the IMAGEN Consortium. (2012). Adolescent impulsivity phenotypes characterized by distinct brain networks. Nature Neuroscience, 15(6), 920-927. [ Links ]
Winstanley, C. A., Theobald, D. E., Dalley, J. W., Cardinal, R. N., & Robbins, T. W. (2005). Double dissociation between serotonergic and dopaminergic modulation of medial prefrontal and orbitofrontal cortex during a test of impulsive choice. Cerebral Cortex, 16(1), 106-114. [ Links ]
Wood, R. L., & McHugh, L. (2013). Decision Making after Traumatic Brain Injury: A Temporal Discounting Paradigm. Journal of the International Neuropsychological Society, 19(2), 181-188. [ Links ]
Xue, G., Lu, Z., Levin, I. P., Weller, J. A., Li, X., & Bechara, A. (2009). Functional dissociations of risk and reward processing in the medial prefrontal cortex. Cerebral Cortex, 19(5), 1019-1027. [ Links ]
Yu, R. (2012). Regional White Matter Volumes Correlate with Delay Discounting. PLoS One, 7(2), e32595. [ Links ]
Yuen, K. S. L., & Lee, T. M. C. (2003). Could mood state affect risk-taking decisions? Journal of Affective Disorders, 75(1), 11-18. [ Links ]