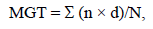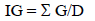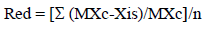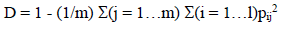Introduction
Salinity is one of the most serious problems affecting soils in arid and semi-arid areas around the world, affecting more than half the world's irrigated land and 20% of the cultivated land (Hasegawa and Bressan 2000). Peng et al. (2008) suggest that the widespread increases in saline soils and losses of arable lands, especially in the arid and semi-arid areas of most countries, could be the result of climate change, overgrazing, mowing and inappropriate farming systems. Salinization in soils affects crop growth as well as livestock production in pastoral regions (Peng et al. 2008). As the area affected by salinity is increasing, there is an urgent need to develop cultivars of forage species with an improved tolerance to salinity.
Saline stress involves both osmotic stress and ion toxicity (Munns 2002; De Lacerda et al. 2003), which interferes with ion homeostasis in plants (Huang and Redmann 1995; Huh et al. 2002), reducing growth by decreasing the rate of photosynthesis and leaf elongation (Pittaro et al. 2015). Salinity reduces and delays germination and emergence due to abnormal morphological, physiological and biochemical changes. It has been suggested that salt tolerance at a given growth stage is not strictly correlated with tolerance at other stages (Tobe et al. 2000) and it is generally accepted that plants are often more susceptible to salinity during germination and the seedling stage than as adults (Tober et al. 2007). Moreover, in order to survive saline conditions as an adult plant, pasture plants first have to overcome the inhibition of germination.
It is well known that the first step in establishing a breeding program is to demonstrate that there is enough variability for the target characteristic in the available germplasm collection (Vogel and Burson 2004). Morpho-physiological traits have been widely used to evaluate the genetic diversity for salt tolerance in crop species and over recent decades, molecular markers have been used extensively to study genetic diversity (Shahzad et al. 2012). In addition to advantages such as stability, reproducibility, high polymorphism and reliability, molecular markers are not influenced by the environment and many individuals can be screened at the same time. Markers such as Inter-simple sequence repeats (ISSR) have been utilized widely to investigate the genetic variations among populations in several species (Ganopoulos et al. 2015), especially grasses (Jurgenson 2005), using a primer designed from dinucleotide or tri-nucleotide simple repeats.
Panicum coloratum, a warm-season C4 perennial grass, native to South Africa, is a cross-pollinated species and appears to be adapted to a wide range of soil conditions that makes it attractive for forage production in marginal areas. In particular, P. coloratum var. makarikariense can tolerate periods of drought followed by flooding (Tischler and Ocumpaugh 2004). A live germplasm collection belonging to INTA was assembled from sites where accessions had been established for more than 10 years and were considered adapted to different environmental and management conditions. Previous studies by our research group showed that P. coloratum is highly heterozygous (Armando et al. 2015) and that accessions in the collection represent panmictic populations (Armando et al. 2017). Aiming to develop a base population as the starting point for a breeding program, we screened the collection of Panicum coloratum var. makarikariense at INTA EEA Rafaela, focusing on the early stages of plant development, i.e. germination and seedling growth.
Since there were differences in origin and possibly exposure to particular selection pressures among accessions in the collection at INTA EEA Rafaela, we hypothesized that there is variability in life history traits and other characters such as salinity tolerance within and among them. In this study we assessed variability in response to salt concentration at germination and seedling stages and molecular variability by ISSR markers in the collection of Panicum coloratum var. makarikariense at INTA Rafaela in an endeavor to identify suitable accessions for use in a breeding program to improve salinity tolerance in the species.
Materials and Methods
Plant material
Four of the 7 accessions of the collection of P. coloratum var. makarikariense at INTA Rafaela Experiment Station (31°11'41" S, 61°29'55" W) were chosen for their good forage and seed production. A complete description of accessions in the collection is reported in Armando et al. (2013) and accessions we used are referred to as DF, ER, UCB and TS (TS corresponds to identified plants of cv. Bambatsi). In January-March 2014 seeds were collected from individuals constituting families of half-siblings from the 4 accessions giving a total of 18 mother plants (5 mother plants from DF and TS and 4 from ER and UCB).
Two different experiments were conducted to evaluate variability in response to increasing NaCl concentration at germination both within and among accessions of P. coloratum var. makarikariense. Incubation was performed in a growth chamber under 12-h photoperiod and at constant temperature (30 ± 4 °C). Seeds were monitored daily for 21 days and germinated seeds were recorded. A seed was considered germinated when the radicle was visible. For each experiment, conditions were set as explained below.
Salt tolerance at the germination stage
Experiment 1. From each of the 18 mother plants samples of 20 seeds were surface-sterilized with sodium hypo-chlorite solution (0.5%) and incubated in 5 cm diameter Petri dishes in 10 mL solutions of increasing NaCl concentrations (0, 100, 200, 300 and 400 mM NaCl) in a complete randomized block design with 3 replicates, i.e. 270 samples.
Experiment 2. For each of the 18 mother plants, samples of 20 seeds were placed in Petri dishes in 10 mL solutions of NaCl concentrations (0, 75 and 150 mM NaCl) in a complete randomized block design with 5 replicates and a total of 270 samples. As before, germination was monitored daily for 21 d, when seeds that had not germinated under the solutions containing salt were rinsed and placed in Petri dishes with distilled water for 10 d to evaluate their capacity to germinate under these conditions.
Analyzed variables
Germination percentage (GP) for each family was calculated as the ratio between the total number of germinated seeds and total number of seeds incubated. GP (% of control) (ratio of germination percentage in salinity treatment and mean germination percentage in control conditions) was calculated as the ratio between GP at each saline concentration and the GP at 0 mM NaCl. Mean germination time (MGT) was calculated as follows:
where: n is the number of seeds germinated on day d; d is the number of days elapsed from the beginning of the test; and N is the total number of germinated seeds in the analyzed period (21 d) (Ellis and Roberts 1980; Raccuia et al. 2004).
The index of germination was estimated using Timson modified index of germination velocity (IG) as follows:
where: G is the seed germination percentage at one-day intervals; and D is the entire germination period analyzed (21 d) (Khan and Ungar 1984).
The recovery of germination percentage was calculated as follows:
R = [(a-b)/(c-b)]x100
where: a is the total number of seeds that germinated in a NaCl solution plus the number that germinated in distilled water after 10 d; b is the number of seeds that germinated in a NaCl solution; and c is the total number of seeds tested (Khan and Gulzar 2003; Wang et al. 2008).
Salt tolerance at the seedling stage
Seeds were grown in pots in a greenhouse during March to June at day/night temperatures of 34 °C/12 °C and light maintained at 623 (imol/m2/sec. Seedlings with 3-4 leaves were placed in plastic trays filled with aerated full-strength Hoagland nutrient solution (Hoagland and Arnon 1950) and maintained under these conditions for 7 days. In salt treatments, salt concentration was then gradually increased (by adding NaCl in increments of 50 mM per day) until final concentration of 200 mM was reached. Nutrient solution without NaCl was used as the control treatment. There were 10 plants per family and treatment in a complete randomized design with 2 replicates. After 12 d of growth at the maximum NaCl concentration, 3 plants per treatment were withdrawn and Na+ and K+ concentrations in fully expanded leaves were determined by using high-performance liquid chromatography (HPLC). Na+ and K+ concentrations and the relationship Na+:K+ were expressed as the concentration of each ion in saline conditions over the concentration in control conditions, and referred to as Na+ (cont), K+ (cont) and Na+:K+ (cont). After 35-day periods, seedling numbers surviving (SN) were determined, 4 plants per family per treatment were removed and plant height (H) and leaf number (LN) were determined. Plants were then separated into aerial parts and roots, dried in an oven at 65 °C for 72 h until constant weight was reached and aerial dry weight (ADW) and root dry weight (RDW) were determined. For each morphological character measured, the reduction of growth (Red) produced by salt stress was estimated as follows (Griffa et al. 2010):
where: MXc is the median for control plants; Xis is the value of each salt-treated plant; and n is the number of replications.
Other variables were also calculated: relative water content of the aerial fraction [(AFW-ADW)/ADW]; and ratio between aerial dry weight and root dry weight (ADW:RDW).
Molecular characterization
The 18 mother plants of P. coloratum var. makarikariense were studied using ISSR markers. DNA extraction was carried out using a modified CTAB (cetyltrimethyl ammonium bromide) method. About 100 mg of homogenized leaf tissue was combined with 600 μ L of CTAB buffer [100 mM Tris-HCl; 20 mM EDTA; 1.4 M NaCl; 1% p/v PVP (polyvinylpyrrolidone); 2% p/v CTAB; 0.2% v/v P-mercaptoethanol] and incubated at 60 °C for 60 min. After adding 600 μ L of chloroform-isoamyl alcohol (24:1), the solution was centrifuged at 13,000 rpm for 10 min. This was followed by precipitation with 500 (iL of ethanol, incubation at 20 °C overnight and centrifuging at 13,000 rpm for 2 min. The pellet was washed with 70% ethanol and dissolved in 200 (L of 1X TE (Tris-EDTA) buffer.
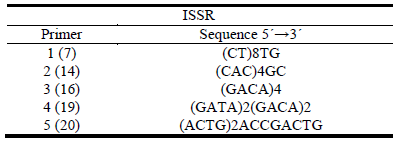
Five ISSR primers were used (Table 1). Amplification reactions were performed in 25 (L volume containing: 30 ng of DNA template; 2.5 mM MgCh; 0.2 mM of each dNTPs (generalized abbreviation for deoxytriphospate nucleotides); 1.2 μ M of primer; and 1.25 U of Taq (Thermus aquaticus) DNA polymerase in 1X buffer. PCR (polymerase chain) reactions were carried out in Thermo cycler BioRad. Initial denaturation was at 94 °C for 3 min, followed by 39 cycles of 94 °C for 30 sec, 40 sec at annealing temperature, 40 sec at 72 °C and a final 5 min extension at 72 °C. Amplification products were resolved on 1.5% agarose gels, run at 70 V in 1X TBE (Tris-borate-EDTA) buffer for 160 min. They were visualized by staining with ethidium bromide and photographed under ultraviolet light.
Salt tolerance at the germination stage. Residuals of GP, GP (% of control) and recovery were not normal. For this reason, total proportion of seeds that germinated was analyzed using generalized linear mixed models (PROC GLIMMIX using events/trials syntax) treating germination as binary (0 = did not germinate, 1 = germinated) (Cordeiro et al. 2014). Residual of variables MGT and IG were normal.
In the first assay, treatments, blocks, accessions and interactions between accessions and treatments were considered as fixed effects.
In the second assay we analyzed the distribution of variability in salinity tolerance in the collection. Block and treatment were considered as fixed effects. Accession and families nested within accession were considered as random effects. To evaluate recovery, accession effect was set as fixed.
Variable response to salt tolerance at the seedling stage and molecular characterization. Principal components analysis (PCA) based on the standardized Euclidean distance was performed with morphological and physiological variables as an exploratory method. Coordinate analysis (PCoA) based on the standardized Jaccard distance (Jaccard 1908) was performed with molecular markers. The following diversity parameters were estimated: genetic diversity; percentage of polymorphic loci (% P); number of alleles; mean effective alleles/ locus; Nei' s expected heterozygosity; number of bands; and percentage of polymorphic bands. For m locus, genetic diversity (D) was estimated as:
where: pij was the frequency of allele i in the locus j. For a locus, genetic diversity was estimated as: D = 1 - 2(i = 1.l)pi2. Then, these parameters were calculated for susceptible and tolerant families according to PCA. Correlation analysis correspondence between phenotypic and molecular matrices containing Euclidean and Jaccard, respectively, was investigated through a Mantel test (Mantel 1967). Statistical significance was determined using 1,000 random permutations. In this test, a correspondence measure (rxy) was calculated between the elements of 2 matrices, X and Y.
All analyses were performed in SAS version 9.2 (SAS 2010) and Infostat/Infogen (Balzarini and Di Rienzo 2011; Di Rienzo et al. 2011).
Results
Salinity tolerance at the germination stage
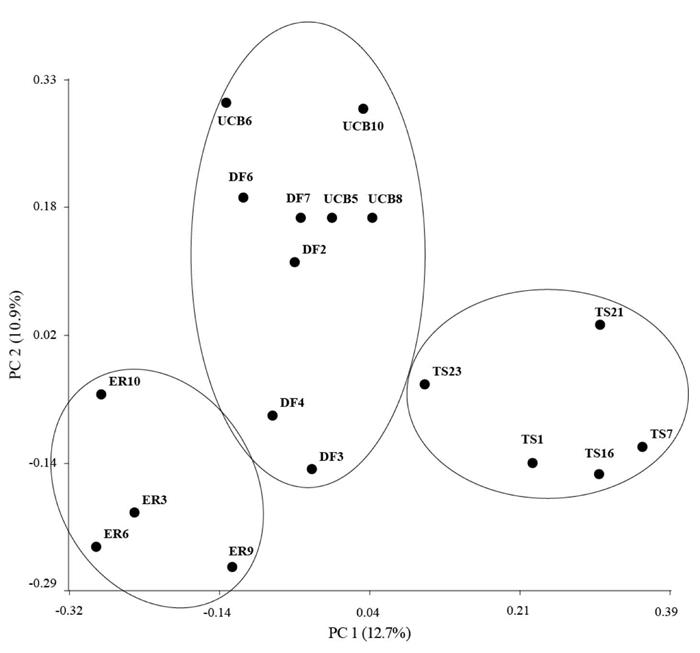
In order to evaluate the variable response to salinity among accessions at germination, 2 assays were carried out with different NaCl concentrations. In the first experiment, the interaction treatment x accession was significant (P = 0.014). While salinity generally reduced the number of germinated seeds, the magnitude of reduction differed among accessions (P<0.001) (Figure 1A). Germination was drastically reduced at concentrations above 200 mM NaCl and no seeds germinated at 400 mM NaCl (data not shown). No significant differences were detected in GP (% of control) with increasing salinity, although means were greater for accession ER at 100 and 200 mM NaCl (Figure 1B).
The second experiment was designed to determine whether variability in response to salinity was better explained by differences between accessions or differences between families within accessions at salt concentrations below 200 mM (Table 2). For all analyzed variables, variance due to accessions was null. Both GP and index of germination velocity (IG) decreased with increasing salt concentrations in the germination media (75 and 150 mM NaCl) with respect to control (0 mM NaCl, P<0.0001). The time necessary for a seed to germinate under saline conditions was longer than the time required for controls (P<0.0001). No significant differences between accessions were detected for GP, IG and MGT (Figure 2).
Table 2 Components of variance of germination percentage (GP), GP (% of control), mean germination time (MGT) and index of germination velocity (IG) of seeds of Panicum coloratum var. makarikariense in saline germination medium.

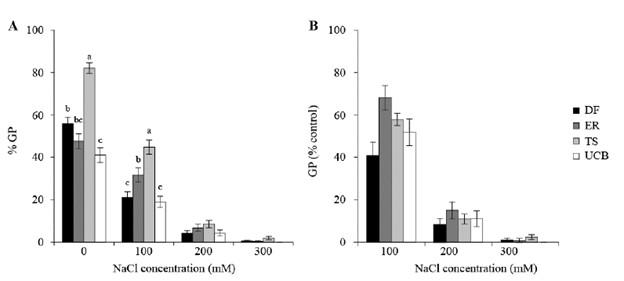
Figure 1 A. Germination % (GP) in accessions of Panicum coloratum var. makarikariense (DF, ER, TS and UCB) in 0, 100, 200 and 300 mM NaCl at 21 d. B. GP (% of control) at 100, 200 and 300 mM NaCl at 21 d. The data represent means ± s.e. Different letters indicate significant differences between accessions within salinity levels (P<0.05).
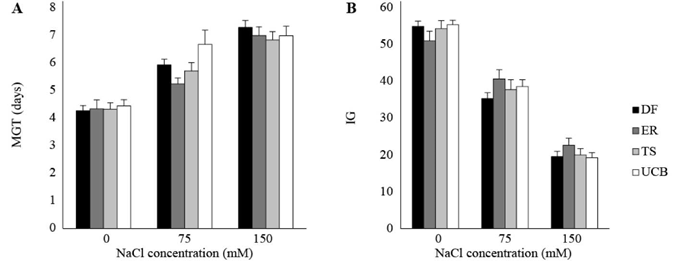
Figure 2 A. Mean germination time (MGT). B. Index of germination velocity (IG) in accessions of Panicum coloratum var. makarikariense (DF, ER, TS and UCB) in 0, 75 and 150 mM NaCl at 21 d. The data represent means ± s.e. No differences between accessions within salinity levels (P>0.05) were detected.
There were significant (P<0.0001) differences between accessions in ability to germinate in distilled water after being exposed to saline conditions (Figure 3). While some remaining seeds of all accessions germinated, those of accessions TS and ER showed higher germination percentage than those of accessions DF and UCB after exposure to both 75 and 150 mM NaCl. The final germination percentage (FGP %), i.e. the total number of germinated seeds from an accession, including those germinating in saline conditions plus those germinating subsequently in distilled water, varied significantly between accessions. While final germination percentages in seeds in the control treatment, i.e. those exposed only to distilled water, were similar for all accessions, final germination percentages in seeds exposed to saline conditions and then to distilled water were significantly greater for ER and TS. Overall, while exposure to saline conditions reduced the total number of seeds which germinated (FGP %) for all accessions, the extent of the reduction was significant for only UCB and DF at 75 mM NaCl and for all 4 accessions at 150 mM NaCl.
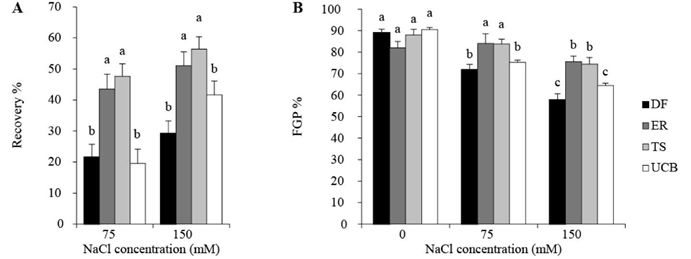
Figure 3 A. Recovery in germination of seed of accessions of Panicum coloratum var. makarikariense (DF, ER, TS and UCB) after exposure to 75 and 150 mM NaCl for 21 days then incubation in distilled water for 10 d (seeds germinating in 0 mM NaCl were not considered for recovery). B. Final germination percentage (FGP %) of seeds of accessions of P. coloratum var. makarikariense including seeds germinating after exposure to saline solutions for 21 days plus seeds germinating after subsequent exposure to distilled water for 10 days. The data represent means ± s.e. In each figure, different letters on columns indicate differences between accessions (P<0.05).
Salt tolerance at the characterization seedling stage and molecular characterization
In order to evaluate the variation in salt tolerance at the seedling stage, an experiment comparing responses of seedlings of all accessions to control (0 mM NaCl) and saline conditions (200 mM NaCl) was carried out under hydroponic conditions. Salinity affected growth of plants, reducing parameters such as height (H), leaf number (LN) and seedling number (SN) (P<0.0001) and resulting in lower biomass of both aerial and root components (ADW and RDW) as depicted in Figure 4. Relative water content of the aerial fraction (AFW-ADW)/ADW was the only variable that did not show differences between families with increasing salinity (P = 0.35) (data not shown). Significant differences in plant growth due to exposure to salinity were detected between families (P<0.001) but not between accessions. In general, reductions in aerial and root growth (RedADW and RedRDW) were significantly lower (P<0.0001) in ER3, ER6, ER10, DF7, TS16 and TS23 than in other families (Figure 4).
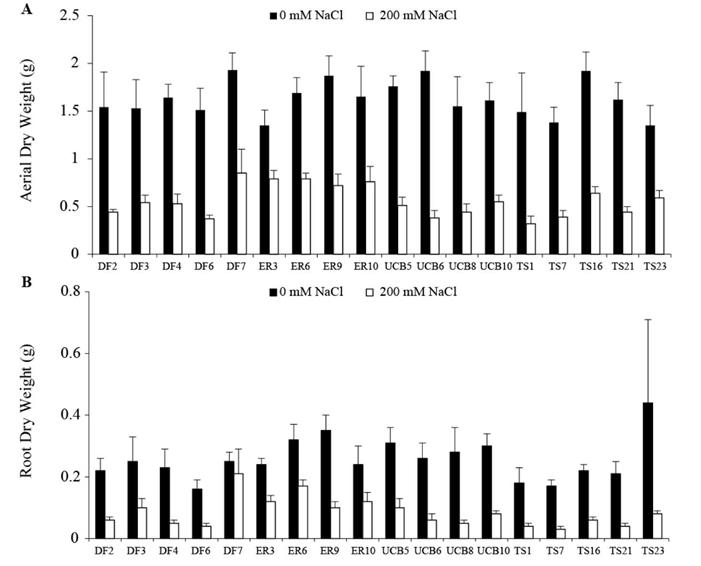
Figure 4 A. Aerial dry weight (ADW). B. Root dry weight (RDW) of plants of 18 families of 4 accessions of Panicum coloratum var. makarikariense in control (no NaCl in the medium) and 200 mM NaCl after 35 d. The data represent means ± s.e.
Tissue concentrations of ions Na+ and K+ of plants growing in control and saline conditions are depicted in Table 3. Ion concentrations in saline conditions relative to those in controls showed significant differences among families in variables such as Na+ (cont) (P<0.0001), K+ (cont) (P = 0.032) (data not shown) and K+:Na+ (cont) (P<0.0001) (Figure 5).
Table 3 Tissue concentrations of ions in leaves of Panicum coloratum var. makarikariense growing in saline (200 mM NaCl) and control (0 mM NaCl) hydroponic conditions in the greenhouse. Values are the means of determinations in 3 plants from the same half-sib family.
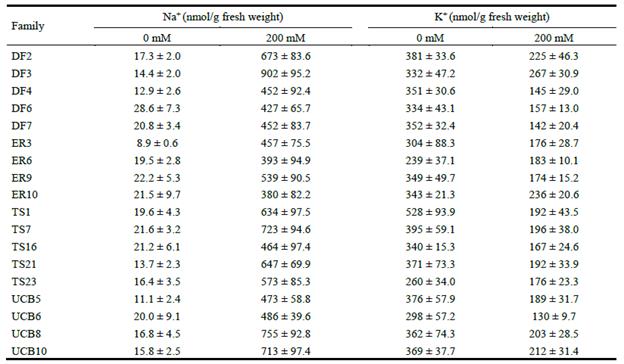
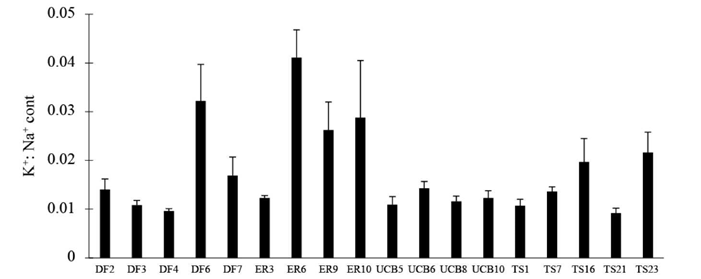
Figure 5 K+:Na+ ratio in tissue of plants grown for 21 days in saline 200 mM NaCl solutions relative to control plants for 18 families of Panicum coloratum var. makarikariense. The data represent means ± s.e.
Evidence of morphological differentiation among families in response to salinity is depicted in the PCA biplot (Figure 6). The first 2 components of the PCA explained 62.1% of the total variation. ADW showed high correlation with PC1, while SN, LN, RedADW and RedRDW were also well represented in PC1. Na+ (cont) and K+ (cont) were correlated with PC2 and K+:Na+ (cont); RedSN was relevant as well. A group of families with low reduction in growth, high values for morphological characters, especially in roots, and low accumulation of sodium in leaves with salinity, comprising ER3, ER6, ER9, ER10, DF7, TS16 and TS23, was clearly differentiated and distinguishable from the rest (Figure 6). Then, by means of the PCA we divided the families into 2 groups, i.e. susceptible and tolerant.
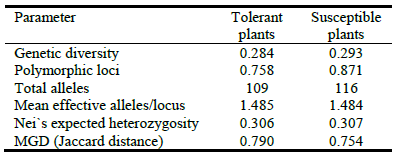
In order to characterize the molecular variability within the evaluated material, DNA from mother plants was extracted and analyzed with ISSR molecular markers. All primers used (5) were polymorphic and produced a total of 124 alleles. Genetic diversity (Jaccard) was 0.317, effective number of alleles per locus was 1.525 and Nei's expected heterozygosity and MGD were 0.326 and 0.775, respectively. When considering plants as belonging to groups (susceptible and tolerant) as differentiated in the PCA (Figure 6), genetic parameters were slightly different as shown in Table 4.
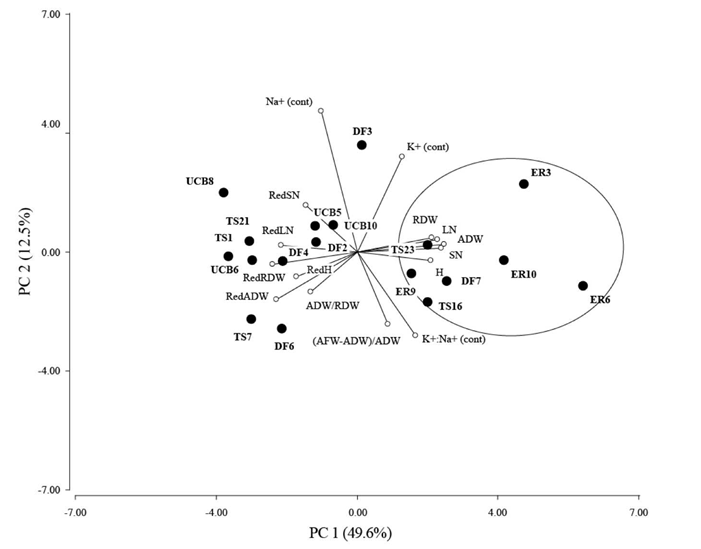
Figure 6 Variables and individual plot of principal components analysis (PCA) based on the Euclidean distance matrix calculated from 15 variables: aerial dry weight (ADW), root dry weight (RDW), number of seedlings (SN), leaf number (LN), height (H), reduction in growth of all variables (RedADW, RedRDW, RedH, RedSN, RedLN), (AFW-ADW)/ADW, (ADW:RDW) and Na+ (cont), K+ (cont) and K+:Na+ (cont), which are the ion concentrations in leaf tissue in saline conditions relative to the concentration in control conditions as explained in methods. Half-sib families (7) identified as tolerant according to morphological variables and ion concentration in green tissue are surrounded by the circle as explained in the text.
Table 4 Genetic parameters in plants categorized as tolerant and susceptible to salinity according to principal components analysis.
PCoA biplot (Figure 7), based on the ISSR distance matrix, provided evidence of molecular differentiation among accessions but failed to distinguish between susceptible and tolerant plants. The first two coordinates of PCoA explained only 23.6% of the total variation.
Finally, the analysis between phenotypic (tested on the progeny) and molecular distances (tested on the maternal plants) by means of a Mantel test showed a significant positive but low correlation (r = 0.24; P = 0.038) between morphological and molecular distances.
Discussion
As expected, germination was inhibited by an increase in salinity. These results are in agreement with previously reported reductions in germination caused by salinity stress in Panicum turgidum (El-Keblawy 2004) and P. miliaceum (Sabir and Ashraf 2008; Liu et al. 2015). Low to no germination at salt concentrations above 200 mM NaCl was also described by Taleisnik et al. (1999) in Panicum coloratum var. coloratum and other Panicum species (Hester et al. 2001; El-Keblawy 2004). However, Khan and Gulzar (2003) claimed that other grass species, like Spartina alterniflora and Aeluropus lagopoides, could tolerate salt concentrations of up to 500 mM. Patterns of response in other parameters associated with germination such as MGT and IG in related species were also similar to those reported by Liu et al. (2015).
Panicum coloratum as a species has been described as relatively tolerant of salinity (Pittaro et al. 2015), especially var. makarikariense (Tischler and Ocumpaugh 2004). However, in order to develop a breeding program to improve tolerance of salinity, it is crucial to evaluate the existence of genetic variability for this character (Vogel and Burson 2004). In addition, it is highly desirable to understand the variability structure in order to select appropriate accessions for crossing to produce the desirable outcomes.
We found that variability among families within accessions was higher than variation between accessions (Table 1). In fact, differences between accessions were hard to detect (Figure 1). This supports previous results by our research group that Panicum coloratum is a highly heterozygous, allogamous species, while accessions can be quite variable displaying a certain degree of phenotypic overlapping (Armando et al. 2015). A very similar pattern of diversity, in a variable related to response to an abiotic stress factor, is reported in the present study. Despite the fact that the accessions we studied were collected from non-saline areas (Armando et al. 2013) and differences in salinity tolerance between accessions were unexpected, a considerable level of variability in this attribute was encountered. Recovery tests are usually used to determine whether non-germinated seeds following exposure to saline conditions are killed or germination is merely prevented by saline stress (Guan et al. 2009). In our assay, many seeds, which failed to germinate in NaCl solution, maintained the ability to germinate after transfer to distilled water. This result suggests that the inhibitory effects of salinity could be due to osmotic effects and not to sodium toxicity. Following exposure of seeds to saline solution, salt would enter the seed causing an inhibition of germination, although it was not directly toxic, allowing seeds to recover afterwards in pure water (Zhang et al. 2012). This ability of seeds to remain viable after exposure to salinity and to germinate when saline stress is reduced is an important mechanism enabling persistence under a stressful unpredictable environment (Keiffer and Ungar 1995). Although all accessions had the capacity to recover, differences between accessions were detected. In particular, the superior recovering ability of seeds from ER and TS (Figure 3A) makes these accessions potential candidates for selecting genetic material that can germinate when saline conditions are removed, as might occur after a rainy season preceded by a dry period.
As is the case with germination, salt tolerance during early seedling stages is critical for the establishment of plants in salty soils (Al-Khateeb 2006). In our assay, saline conditions suppressed seedling growth in all evaluated families but the magnitude of the suppressions varied markedly. In addition, plants growing in saline solutions accumulated additional sodium in their leaves, while, in general, potassium level was decreased. The reduction in the ratio of K+:Na+ in response to salinity has been reported for other forage species such as Panicum antidotale (Ahmad et al. 2010).
In our experiments, we encountered variability in almost all characters between families at the seedling stage under saline conditions, indicating that plants suffered damage due to high salt concentration with different levels of injury. Morphological and biochemical variables allowed us to differentiate tolerant from susceptible families as shown in Figure 6. We identified tolerant plants as those producing the most biomass under salinity and showing lower levels of damage (reduction in growth). These families also showed lower values for ADW:RDW, indicating preferential allocation of biomass to roots, and also showed comparatively less damage at root level. This characteristic is crucial in saline areas because the main function of roots is to absorb water and nutrients. The lower decrease of RDW relative to ADW is an indicator of an adaptation to continue to absorb water and nutrients, albeit at a level below the optimum, even under salt stress conditions. Additionally, accumulation of toxic ions like Na+ in roots increases as root biomass increases, thus minimizing its negative effects in the shoot (Marschner 1995; Acosta-Motos et al. 2005). In general, the family groups pointed out as salt-tolerant in Figure 6 also showed high values of K+:Na+(cont) (Figure 5) indicating lower amounts of Na+ in leaf tissue than the susceptible ones.
Characterization by molecular markers showed high levels of variability in the germplasm collection, although utilization of more markers would have depicted the variation in the available germplasm more clearly. Actually, the first 2 coordinates in the PCoA (Figure 7) explained only 23.6% of total variation. Despite the low representation of the variability in the first 2 axes, plants congregate in the plot according to their provenance, i.e. the accession that they belonged to. This pattern of distribution of genetic variation (i.e. a higher proportion between accessions and a lower proportion within accessions) is not normally expected for an allogamous plant but it is in agreement with previous results from our research group (Armando et al. 2015).
Molecular analysis did not allow differentiation based on salinity tolerance. The low correspondence between phenotypic and molecular variation (r = 0.26) was not unexpected, since most DNA markers constitute a sample of random genomic sites in which polymorphism has no effect on phenotypic characters (Holderegger et al. 2006). Given that P. coloratum is an allogamous species, considerable levels of genetic variation are required in a base population prior to performing selection to avoid problems due to inbreeding depression (Vogel and Burson 2004). This should not be a problem with this set of individuals since, when ER3, ER6, ER9, ER10, TS16, TS23 and DF7 (tolerant plants according to PCA) were considered in a separate group, they retained relatively high levels of variation, pointing out that this population, although small, could be used in a breeding program to improve tolerance to salinity (Table 2).
In this study, we reported considerable variation in salt tolerance in a germplasm collection of P. coloratum var. makarikariense at both germination and seedling stages, as well as variability in molecular characterization. As expected for an allogamous species, variability between families within accessions was higher than between accessions for characters related to germination. Pheno-typic characterization at the seedling stage allowed differentiation between tolerant and susceptible families by means of morphological traits and concentrations of ions in the leaves. This tolerant material, although coming from a low number of mother plants (n = 7), provides considerable levels of variability to carry out further cycles of selection. These preliminary findings can be tested by incorporating selected accessions in breeding programs to improve salinity tolerance in this already valuable pasture species.