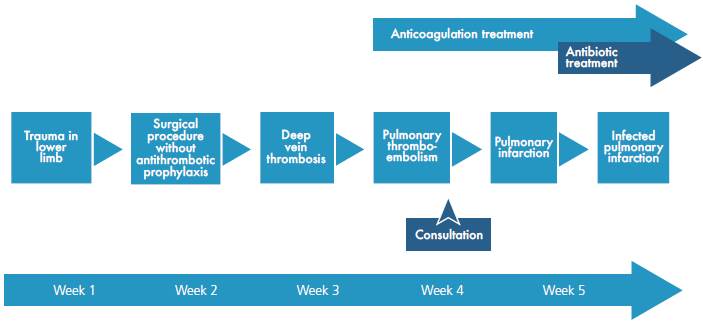
INTRODUCTION
Pulmonary thromboembolism (PTE) is the third leading cause of death related to cardiovascular disease, in which acute right ventricular failure and pulmonary infarction are the main complications 1. Some studies have reported that pulmonary infarction occurs in 29% to 32% of patients with PTE 1-3.
Pulmonary infarction secondary to pulmonary embolism is more common in patients with low cardiopulmonary reserve 4. A study showed that pulmonary infarctions in patients with PTE occurred in 36% of congestive heart failure cases, and in 54% of patients with hypotension and shock 5. Nevertheless, more recent studies have indicated that young patients with good health status prior to PTE can have a higher incidence of pulmonary infarction 6,3. In addition, a high thrombotic burden has been associated with an increased probability of pulmonary infarction, although this is observed in small pulmonary arteries 7.
In general, pulmonary infarction causes pleuritic pain, tachypnea, dyspnoea and, in extremely rare cases, coughing with hemoptysis 8; therefore, radiological differentiation from other entities such as masses, atelectasis and pneumonia is difficult.
Pulmonary infarction is usually observed in subpleural regions and occurs predominantly in the lower lobes, especially the right lobe. The most common radiological finding is a triangular consolidation of the pulmonary parenchyma (50%), although segments with a ground glass pattern (35.9%) and Hampton sign (14.5%) can also be observed, which help to differentiate it from masses and pneumonia 6,9,10.
Multiple complications have been associated with pulmonary infarction, including pneumonia, pneumothorax, bronchopulmonary fistula, alveolar hemorrhage, cavitations, empyema and pulmonary abscess 11. Infection associated with pulmonary infarction is a rare but important complication, as it may result in the formation of abscesses and empyemas that may require surgical drainage.
The obstruction of a pulmonary vessel secondary to a sterile plunger is suggested to be the cause of blood extravasation into the surrounding tissues, of edema in adjacent bronchial walls, and of the increase of endo-bronchial secretion, which forms a favorable environment for colonization and bacterial infection by air or blood 12. Bashir & Benson 13, based on a series of postmortem studies, estimated the incidence of lung infarction infection in 2 to 7% 13. The presence of fever, sweating, tachycardia, purulent sputum, as well as of deterioration of the general condition and leukocytosis after pulmonary infarction, is highly suggestive of infection, which is why the spectrum of etiological agents is considered similar to that described in nosocomial pneumonia 11. In cases of infected pulmonary infarction, anticoagulant and antibiotic therapies should be initiated timely and selected according to local resistance patterns, associated pathogens, patient characteristics and clinical context in general 5.
This paper reports a case compatible with pulmonary thromboembolism complicated by infected pulmonary infarction.
CASE PRESENTATION
49-year-old female, public accountant, from Bogotá, Colombia who consulted due to a two-day long clinical picture involving pleuritic pain in the left hemithorax, irradiated to the dorsal region, associated with sudden dyspnea, edema, and warm sensation on the lower left limb; no fever, coughing or hemoptysis were reported. Two weeks prior to consultation, the patient underwent meniscus reconstruction, and anterior and posterior cruciate ligament and left knee chondroplasty secondary to a closed trauma. No postoperative antithrombotic prophylaxis was performed after the procedure due to unknown reasons. The patient had an important history of class 1 obesity, with no previous pathological, toxic or allergic symptoms nor previous hospitalizations.
Physical examination on admission showed a normal general condition, with blood pressure of 97/63 mmHg, heart rate of 106 bpm, respiratory rate of 20 rpm, oxygen saturation at 96%, oxygen inspired fraction of 0.21, and axillary temperature of 36.5°C. Her height was 160 cm and weight 83 kg, which showed a body mass index (BMI) of 32.4 kg/m2. Thorax auscultation showed P2> A2 without murmurs; fine crackles were also identified in the base of the left lung. Edema in the foot and proximal third of the leg was found in the lower left limb, with local erythema and superficial pain on palpation.
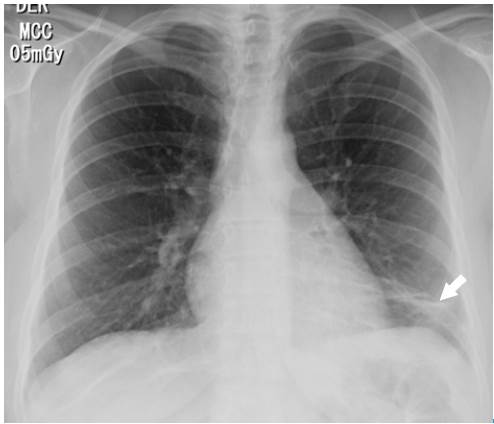
The following tests were performed: blood count, electrolytes, blood urea nitrogen (BUN) and creatinine, which were within normal limits. On the other hand, the electrocardiogram showed sinus tachycardia, while the echo-cardiogram had a structural and functionally normal heart. Finally, the chest X-ray revealed left basal lamellar atelectasis and right atrium growth (Figure 1).
Source: Own elaboration based on the data obtained in the study.
Fig 1 Posteroanterior chest X-ray. Image in left basal band corresponding to atelectasis (arrow).
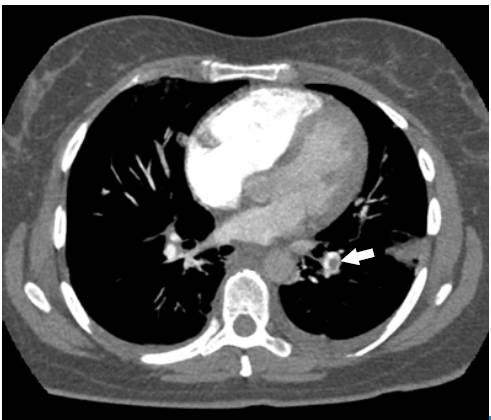
The Wells scale was applied, finding a high probability for pulmonary thromboembolism. Anticoagulation was initiated with low molecular weight heparin (enoxaparin) at a dose of 1 mg/kg every 12 hours, and a computed axial angiography (AngioCAT) of the chest was performed, confirming the diagnostic suspicion by reporting pulmonary artery obstruction in the trunk of the lower left basal segment (Figure 2).
Source: Own elaboration based on the data obtained in the study.
Fig 2 Angiotomography of the chest. Axial plane. Pulmonary embolism, filling defect in the shape of a life preserver sign (arrow).
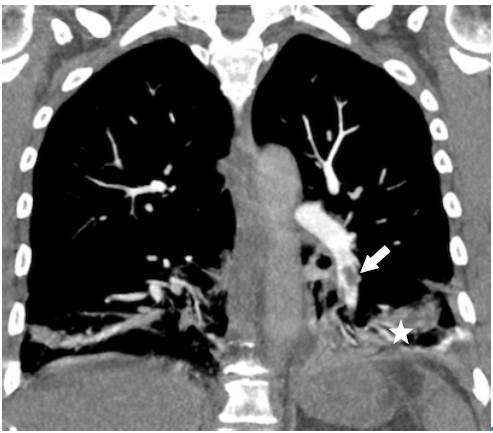
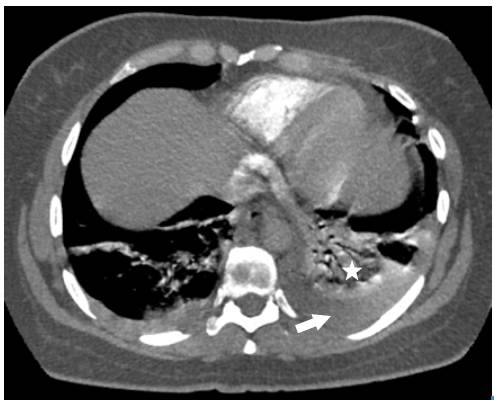
Surprisingly, after initiating anticoagulation and other general treatment measures indicated for pulmonary thromboembolism, and about three days after her hospitalization, the patient required increased oxygen flow through nasal cannula, her chest pain worsened, and presented with dyspnea, fever, leukocytosis, tachycardia, and tachypnea. Control para-clinical exams were requested, revealing high C-reactive protein and a significant increase in leukocytes at neutrophils expense. A new chest tomographic evaluation was performed, in which wedge densities were identified in the middle and lower right lobes and in the lower left lobe, as well as an aerial bronchogram of the right lower lobe and bilateral pleural effusion (Figure 3 and 4). No blood cultures or sputum cultures were performed.
Source: Own elaboration based on the data obtained in the study.
Fig 3 Angiotomography of the chest. Coronal plane. Pulmonary embolism, filling defect configuring the railway track sign (arrow), left basal consolidation of the pleural base and air bronchogram corresponding to pulmonary infarction (star).
With this in mind, infected pulmonary infarction was diagnosed and broad-spectrum antibiotic therapy with piperacillin + tazobactam was initiated at a dose of 4.5 g intravenously every six hours, considering that the infection was nosocomial and that the patient had undergone a recent hospitalization, thus increasing the risk of resistant germs.
The patient improved progressively, and oral anticoagulation with warfarin was initiated at a dose of 5 mg every 24 hours. After completing the seventh day of antibiotic treatment and with INR (International Normalized Ratio) within the therapeutic range, she was discharged with anticoagulation prescription for three months initially, and internal medicine outpatient controls. No adverse reactions to treatments during hospitalization were observed. Figure 4 shows the timeline of the reported case
Source: Own elaboration based on the data obtained in the study.
Fig 4 Chest AngioCAT. Broad base consolidation, hilar apex, obtuse borders that coincide with pulmonary infarction (star), pleural effusion (arrow).
DISCUSSION
This case shows the clinical evolution of a patient with PTE, which progressed to pulmonary infarction and infection of the necrotic lung tissue. This case is relevant from several points of view, since it points the importance of thrombus prophylaxis in patients at risk, and exposes complications that endanger patients' lives and generate diagnostic, therapeutic and research challenges.
Many risk factors are associated with pulmonary thromboembolism; in this case, the patient presented trauma, surgery, immobility and obesity 14. The incidence of PTE can be reduced by performing thrombus prophylaxis in patients undergoing orthopedic knee surgery, particularly in the presence of additional risk factors 15. Therefore, prophylaxis until patients recover mobility is highly recommended 16.
PTE diagnosis was obtained by following the guidelines for clinical practice 17, that is to say, by classifying the probabilities through Wells scale and confirming the diagnosis with the test of choice, in this case, chest AngioCAT.
On the other hand, the evolution of the patient's clinical picture, besides the development of systemic inflammatory response, elevation of acute phase reactants, exacerbation of pleuritic pain and respiratory deterioration, caused the suspicion of infected pulmonary infarction 8. This clinical presentation, along with compatible diagnostic images, was the basis of the diagnosis. In this case, a significant improvement was achieved, allowing hospital discharge and a good short- and long-term prognosis.
Nevertheless, it is important to mention that this report has significant limitations: the lack of microbiological isolates to determine the etiological agent, the lack of clinical practice guidelines for the diagnosis and treatment of infected pulmonary infarction, and the radiological difficulty to differentiate bacterial pneumonia and pulmonary infarction 11. In addition, antibiotic treatment was selected based on the extrapolation of the local guidelines for the treatment of nosocomial pneumonia. Further research on the subject is necessary to better understand, diagnose, treat and prevent this entity, in order to improve care and clinical outcomes of patients.
In conclusion, pulmonary infarction is a common complication of pulmonary thromboembolism, which can become infected on rare occasions. Consequently, a high degree of clinical suspicion and adequate interpretation of the evolution are necessary to establish the diagnosis and to provide timely management.
Patient Perspective: the patient expressed gratitude for receiving medical care in an appropriate and humanized way.
Informed consent: the patient consented the publication of the information used in this case report.











 text in
text in