INTRODUCTION
Pompe disease is characterized by a deficiency of the acid alpha-glucosidase enzyme, which is responsible for glycogen accumulation in cardiac and skeletal muscles. Its presentation may be early or late, and its diagnosis requires a high index of clinical suspicion and confirmation by molecular tests. 1 Enzyme replacement therapy has proven to improve general motor and lung function, albeit unpredictably.
In adults, enzyme replacement therapy improves or stabilizes skeletal muscle strength, muscle function, respiratory function, and survival; however, the magnitude of response varies among individuals. In 2017, the European Pompe Consortium (EPOC) reached a consensus on when to start and stop enzyme replacement therapy in patients with Pompe disease. Based on the experience of several countries, it was recommended to start when the following conditions were met: 1) diagnosis confirmed through leukocyte enzyme activity or genetic test, 2) symptomatic patient during clinical assessment, and 3) monitoring carried out at least once a year to evaluate the response to treatment.
This effect is even greater when moderate to vigorous aerobic activities and muscle strengthening activities are accompanied by enzyme replacement therapy, showing significant increase in muscle strength and distance traveled in the 6-minute walk test, and a decrease in fatigue and pain. 2-4 This appears to be caused by mechanisms that are not associated with the reduction of glycogen content. 5 Furthermore, specific training of respiratory muscles 6 has shown improvement in the strength of the maximum inspiratory and expiratory pressure in muscles (MIP and MEP).
Short functioning assessment tests may be useful for follow-up in patients with Pompe disease, and the minimal detectable change (MDC) must be taken into account for their application. The following is a clinical case of a patient with late-onset Pompe disease who underwent enzyme replacement therapy with marked improvement in symptomatology, which was evident when performing functional evaluation tests.
CASE PRESENTATION
This is the case of a 34-year-old female mestizo patient, housewife, who consulted due to a clinical picture of proximal weakness experienced since adolescence, which progressively interfered and limited her activities of daily living. The symptoms included hypersomnia, dyspnea and snoring during sleep. On physical examination, she had normal muscle tone and sensitivity, proximal 3/5 and distal 4/5 quadriparesis, thoracic hyperkyphosis, lumbar hyperlordosis and global hyporeflexia, without evidence of tongue muscle involvement or skin lesions. Her family history included a sister with a diagnosis of Pompe disease.
The patient presented with a chronic and progressive pure motor stroke which, given the clinical characteristics of her symptoms (proximal weakness, apparently symmetrical), required ruling out a myopathy first, as some of them frequently involve the respiratory muscles, as is the case of Pompe disease.
Creatine phosphokinase (CPK) was requested as an extension study, yielding an elevated value (790 U/L). Electrodiagnosis of the four limbs was performed, showing fibrillation potentials, especially in the evaluated thoracic paraspinal and proximal muscles, early recruitment, and short duration motor unit potentials. The diagnosis of Pompe disease was confirmed by enzymatic study in leukocytes, finding a marked reduction in the enzymatic activity of alpha-glucosidase (21%). The genetic study demonstrated an abnormal OAG sequence.
Enzyme replacement with alpha-glucosidase was initiated at a rate of 20 mg/kg every two weeks by continuous infusion. One month after the start of treatment, her sleep pattern improved and dyspnoea decreased, but functional performance showed no significant change. After five months of enzyme replacement, respiratory pattern and physical capacity improved (Table 1). There were no adverse drug reactions during the follow-up period.
Table 1 Changes in physical test.
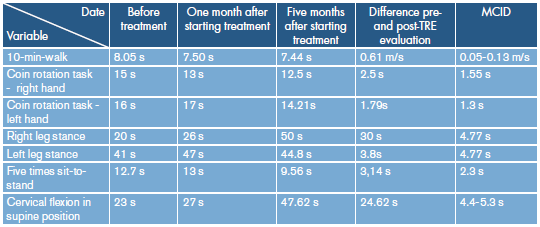
ERT: enzyme replacement therapy; MCID: minimal clinically important difference.
Source: Own elaboration.
Changes in short functional tests were observed from before the start of enzyme replacement therapy until 5 months after beginning the treatment.
DISCUSSION
Pompe disease, also known as glycogen storage disease type 2, is an autosomal recessive disorder considered as a rare inborn error of metabolism, with an estimated frequency of 1 per 40 000 live births. 1 It is characterized by an acid alpha-glucosidase deficiency that causes excessive accumulation of lysosomal glycogen and defects in autophagy, mainly in cardiac and skeletal muscles. Two clinical presentations have been described according to the age of onset: early onset (infantile with or without cardiomyopathy) and late onset (juvenile and adult), which poses greater diagnostic difficulty.
To date, the literature has not described which rapid clinical assessment tests are most appropriate for Pompe disease. The systematic review by Savegnago et al.7 on functional assessment scales in children and adults with Pompe disease included 14 studies, and scales with grade of recommendation (Grade C) were found for the pediatric population (Alberta Infant Motor Scale and Pompe-Pedi). Regarding the adult population, only the Rotterdam Handicap Scale was described -with good reproducibility (ICC=0.94)-, as well as the GMFM (Gross Motor Function Measure) and the FIM (Functional Independence Measure); the last two were used only in one case report. 7 The limitation of these tests lies in the fact that their application is time consuming and must be done by trained personnel.
Functional assessment tests have been widely used in patients with diseases in muscle fibers, so they can be a cost-effective tool to monitor the response to enzyme replacement therapy in patients with Pompe disease and detect changes over time.
In Colombia, several cases of Pompe disease in its juvenile 8 and adult 9 forms have been described; nevertheless, no information has been provided on the improvement of patient's functionality, at least not in an objective manner. In this case, even though the patient showed improvement in symptoms related to respiratory muscle weakness, during the "classical" physical examination no objective functional changes were reported that could be confirmed when the patient performed the functional assessment using simple tests such as the 10 meter walk, coin rotation task, five times sit to stand test, cervical flexion time in supine position and one leg stance (Table 1).
In order to interpret the results and the changes observed in the functional tests, it is important to consider the populations in which they have been validated. To objectively determine whether the change obtained between two consecutive assessments is outside the margin of error, the minimal clinically important difference (MCID) or the minimal detectable change (MDC) are useful if these outcome measures are known. 10,11
For example, the 10 meter test calculates the walking speed in m/s in a short duration, which is useful for monitoring and evaluating the ability to perform safe and independent walking both in the community and at home in adult patients with neuromuscular diseases; it also evaluates the activity domain of the ICF (International Classification of Functionality). During this test, the patient is asked to walk a distance of 10 meters, leaving a space so that the subject can accelerate/decelerate - said space is not included when determining the distance-, and then three trials are averaged to find the speed. This test has excellent test-retest reproducibility (ICC=0.91), and MCID of 0.13 m/s and MDC of 0.05 m/s. 12
The five times sit to stand test measures the strength of the lower limbs and balance when evaluating the IFC activity domain. Three trials are administered and then the average of the three trials or only the value obtained in the first trial is considered. The MDC in patients with vestibular pathology is 2.3s 13; in Parkinson's patients a score >16 is the cut-off point to differentiate subjects with a high risk of falling. 14 Times >12s in older adults were recently described as one of the strongest evidence for predicting risk of future falls. 15
The coin rotation task was designed as a rapid assessment test to measure fine motor dexterity of the fingers. For this assessment, the patient must rotate a coin 20 times using the thumb, index and middle fingers, and the time it takes to develop the activity is measured; the MDC for all ages is 1.55 for the right hand and 1.3 for the left hand. 16
Finally, the single leg stance test screens for balance control and thus the risk of falls. It measures the time that the patient is able to maintain balance using only one leg, without assistance, and with open or closed eyes. It has established normative values for healthy individuals by age group 17; the MDC for the test with closed eyes is 0.48 and with open eyes is 4.77. 18
CONCLUSIONS
Pompe disease is characterized by glycogen accumulation in skeletal muscles, which leads to generalized weakness that progressively limits the functionality of affected patients. Enzyme replacement therapy, along with physical activity interventions, has shown to improve weakness and thus the quality of life of the patients.
This is the case of a rare pathology - late-onset Pompe disease-, which describes the usefulness of functional tests as a quick and effective way of observing the response to treatment in an objective manner. Consequently, these tests are indispensable tools for physiatrists to monitor response to treatment. 19
One of the limitations of this work is that comparisons were not made with other standardized tests to follow up neuromuscular pathologies, although there is broad literature regarding the usefulness of performing such comparisons. The interpretation of these tests must take into account the minimal clinically important difference. Therefore, the coin rotation task, cervical flexion time in supine position, one leg stance, five times sit-to-stand test, and the 10 meter walk test are proposed as tools to objectively measure the response to treatments during interventions.














