Introduction
Cyclooxygenase (COX), the main catalyst enzyme in the prostanoid synthesis, has two main known isoforms, COX-1 and COX-2[1,2]. COX-1, which is responsible for physiological functions, is expressed mainly as a constitutive isoform[3] and, in normal conditions and in distinct regions of the kidney, generates prostaglandins that are responsible for vasodilation and diminished vascular resistance, ensuring adequate blood flow[4]. On the other hand, COX-2, initially considered pathological, is highly expressed in the presence of damaging stimuli[1]. However, this isoform is now also known to appear constitutively in some tissues, like in the kidney, where it is responsible for ensuring the tubuloglomerular feedback, contributing to the establish homeostasis[5].
In this sense, although the COX functions have been well-studied in normal situations, studies have demonstrated that, facing different clinical situations, its isoforms may assume different roles. In cases of hypovolemia and hypertension, this enzyme can develop the physiological role of maintaining the integrity of the kidney, as well as becoming associated with the aggravation of tissue lesion/damage, as observed in the polycystic kidney disease[4,6,10].
Thus, clarifying the true biological functioning of the COX isoforms enables a better comprehension of the kidney physiology and pathology, as well as a better understanding of the consequences of the isoforms' inhibition by medication. This review intended to study the biological role of the COX enzyme in the renal system in different clinical situations.
Cyclooxygenase
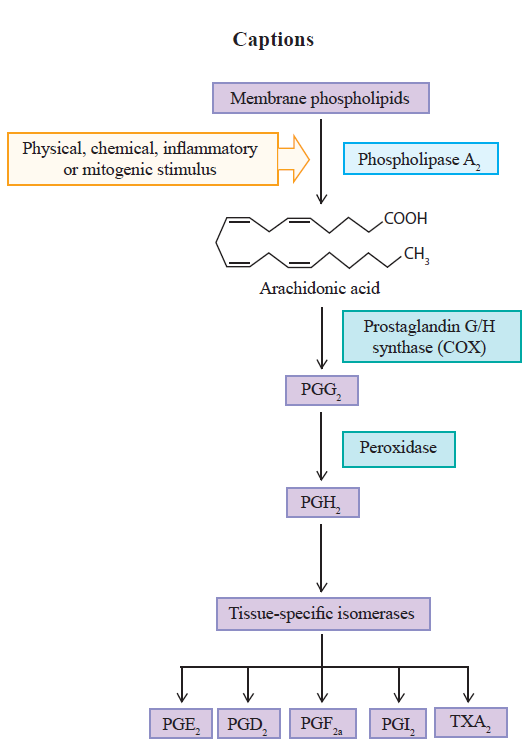
The cyclooxygenase (COX) is the main enzyme that catalyses the prostaglandin synthesis from arachidonic acid. This way, it originates different prostanoids with different functions in the organism[1].
This process is initiated with the archidonic acid, present in the lipid bilayer of the cell membrane, triggering the prostanoid biosynthesis pathway due to the presence of different stimuli[11,16]. The pathway involves a sequence of three main steps. Initially, the arachidonic acid is liberated, by hydrolysis, through the action of the phospholipase A2 enzyme (PLA2). Once liberated, the second step begins, with oxygenation by the prostaglandin G/H synthase, or cyclooxygenase, resulting in the prostaglandin endoperoxide G2 (PGG2). In sequence, the same COX enzymes reduce the PGG2 into prostaglandin H2 (PGH2)[15,17]. The prostaglandin G/H synthase has the cyclooxygenase and peroxidase catalytic sites. The former converts arachidonic acid into PGG2, which is acted upon by the peroxidase enzyme and reduced to the unstable intermediate, PGH2, responsible for the formation of different kinds of prostanoids such as prostacyclin (PGI2), prostaglandin D2 (PGD2), prostaglandin E2 (PGE2), prostaglandin F2a (PGF2a) and thromboxane A2 (TXA2), through its conversion by the tissue-specific isomerases[13,18,20]. These prostanoids provoke different actions in the organism through specific receptors, which depend on the type of cell and tissue involved in the process (Figure 1) [20,22].
There are different isoforms of COX, but the most studied are COX-1 and COX-2[1]. COX-1 was initially named physiological or constitutive for being present in the majority of tissues and being expressed in an almost steadily fashion[13,19,23]. Its main role consists of maintaining a basal rate of the biosynthesis of prostanoids in the body, in order to promote their rapid, short-duration increase. On the other hand, COX-2 is hardly found in most tissues, but its production increases by approximately 20 times in the presence of inflammatory stimuli. For this reason, it has been called the pathological or induced enzyme[13,19,23,24]. However, recent studies have demonstrated that it is also found constitutively in some tissues, being responsible for the formation of prostanoids that maintain homeostasis in different organs and systems of the body[1,24,26].
Cyclooxygenase in renal function Normal Function
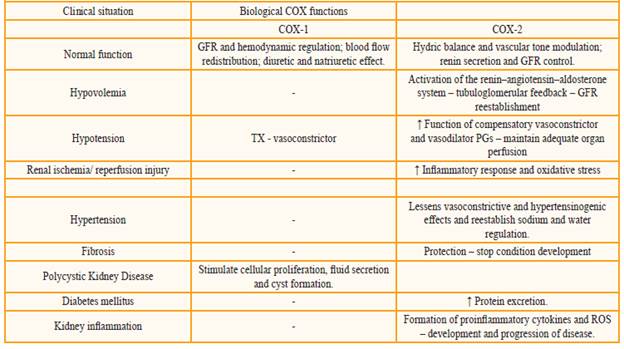
COX-1 is found in the glomeruli, in the medullary and cortical region of the collecting ducts and in the afferent and efferent arterioles[27], ensuring the maintenance of the kidney's physiological functions, such as hemodynamic regulation and glomerular filtration rate (GFR)[13,28,30]. This isoform produces PGE2 and PGD2, which antagonize the vasoconstrictive action of angiotensin II (Ang II), and inhibit the release of norepinephrine, respectively[31].
In this way, these prostanoids promote vasodilation, increasing the perfusion of the organ and causing redistribution in the blood flow from the renal cortex to the nephrons in the intramedullary region[31]. Additionally, the PGE2, alongside the PGF2a, possess the diuretic and natriuretic effects and the PGE2, like the PGI2, antagonize the action of vasopressin[31].
Although COX-2 has been initially considered pathological, is also found constitutively in the kidneys[29,32]. Studies in COX-2 knockout rats demonstrate a grave defect in the renal formation, suggesting this isoform performs an important task in the development of the kidneys[33].
COX-2 is situated in particularly important regions for the renal function, concentrating mainly in the medullary region, and in the cortical region to a lesser extent[32]. Specifically, it can be found in smooth muscle cells of the afferent and efferent arterioles, in the endothelium (straight venules and vasa recta), renal artery, interstitial fibroblasts, and in podocytes in healthy kidneys[34]. It is also expressed in the thick ascending limb of the Henle loop, in addition to being found in the macula densa, which is responsible for mediating the interaction between glomerular filtration and proximal reabsorption, and for controlling the levels of sodium and potassium ions in the lumen of the distal tubule through the renin-angiotensin-aldosterone system[7,8,27,28,35].
Like in COX-1, prostaglandins (PGs) also derive from COX-2, being important physiological modulators of vascular tone and hydric balance in the kidneys[13,35]. The prominent PGs in renal tissue are PGE2 and PGI2, where PGE2 is synthesized by the tubular epithelium and interstitial cells, expressed in the renal tubules regulating the transport of chloride and sodium in the Henle loop, besides auxiliating water transport and blood flow. PGI2 is located in the renal cortex, controlling the glomerular filtration rate and renin secretion[7,29].
Hypovolemia
Hypovolemia is characterized by a decrease in blood volume, being mainly the result of excessive loss of water in the kidney or hemorrhage. As a consequence, arterial pressure drops, with the possibility of hypovolemic shock and death[13,31].
In a state of hypovolemia with consequent depletion of sodium levels, there is an increase of COX-2 in the cortical region, and a decrease of its expression in the medullary region, causing the renin-angiotensin-aldosterone system to be activated. The juxtaglomerular cells secrete the hormone renin, which contributes to the formation of Ang II, responsible for the vasoconstriction of the efferent arteriole together with norepinephrine, guaranteeing the increase of the intraglomerular hydrostatic pressure. The hormone also stimulates the secretion of aldosterone, responsible for promoting the reabsorption of sodium and, ultimately, inducing the liberation of antidiuretic hormone (ADH), which increases water and urea reabsorption. At the same time, angiotensin stimulates the synthesis of renal vasodilator prostaglandins (afferent arteriole), which guarantees the tubuloglomerular feedback, reestablishing the glomerular filtration rate, and ensuring normal blood flow to protect the kidney from acute functional deterioration[5,13,31,35,37].
The administration of selective or non-selective COX-2 non-steroidal anti-inflammatory drugs (NSAIDs) prevents the release of renin and, consequently, the restoration of renal function[38].
Hypotension
Hypotension leads to low effective filtration pressure in the glomeruli, which can become a state of chronic kidney disease. In these situations, the function of compensatory vasoconstrictor and vasodilator prostaglandins (PGE2 e PGI2), provenient from COX-2, is amplified in an attempt to maintain adequate organ perfusion[39,40].
This action favors the resorption of sodium/ water, which maintains a renal homeostasis by the GFR. In agreement, the thromboxane (TX), produced by COX-1, provokes vasoconstriction. Thus, NSAIDs may be use carefully because they can act blocking the COX-1 and COX-2 enzymes and, consequently this mechanism[41]. In the study of Calistro Neto and group (2015), animals that use Parecoxib, a selective inhibitor of COX-2, had renal detrimental effect by the non-activation of renin-angioten-sin-aldosterone system[41].
In disagreement with the results, Tunctan and group (2013) declare that COX-2 induces hypotension, because their vasodilatory capacity. This study concludes that the use of a COX-2 inhibitor is able to restore blood pressure[42].
Renal ischemia/reperfusion injury
The pathology of this injury is marked by the presence of an inflammatory response and oxidative stress[43]. The COX-2 is highly expressed and is able to aggravate the inflammation. So, the use of a NSAID that selectively inhibits COX-2, also called coxib, can be benefic to the renal function by prevention of oxidative injury[27,43,44].
More specifically, the use of parecoxib, a coxib, resulted in renal protection[41]. A similar study that uses indomethacin, a non-selective NSAID, and rofecoxib, a coxib, showed that both drugs were associated with improved renal function and a reduction of proinflammatory cytokine levels[45].
To finish, the use of nimesulida, a COX-2 selective inhibitor drug, also prevented the renal damage[46].
Hypertension
In contrast, in a state of hypertension, COX-2 expression will also be increased. However, unlike in hypotension, this increase will occur in the medullary tissues in an attempt to ensure that the vasoconstrictive and hypertensinogenic effects of Ang II be softened, aiming then to reestablish sodium and water regulation, as well as the maintenance of medullary blood flow[47]. In this sense, the use of any NSAIDs aggravates the hypertension and it must be avoided. The block of PGE2 by drugs lead to sodium retention which exacerbate the deleterious process[38].
Fibrosis
Another alteration of the renal system is fibrosis. It is characterized by the development of glomerular sclerosis and interstitial fibrosis, being one of the consequences of the final stages of several chronic kidney diseases[48]. The COX-2 isoform guarantees protection against fibroses and, by virtue of that, the high quantity of PGE2 generated can hinder the development of the condition[48].
Polycystic kidney disease
As well as renal ischemia/reperfusion injury, the COX enzyme can also present deleterious effects in other situations in the renal system. In the presence of polycystic kidney disease, a autosomal dominant renal disorder frequently hereditary in nature characterized by the continuous growth of cysts, there is an increase in activity of PGE2 and PGI2 (derived from both isoforms of COX), responsible for stimulating cellular proliferation, secretion of fluid and cyst formation[49].
In a humam study, by the blood collection, it was found that patients with autosomal polycystic kidney disease (APKD) have high levels of COX compared to the health group[9]. Agreeing with these authors, by culture of epithelial cells from cysts of patients with APKD, Xu et al. (2012)[50] concluded that the use of a COX-2 inhibitor is capable to reducing the epithelial proliferation, promoting apoptosis of these, and decrease the PGE2 synthesis[50].
Thus, the inhibition of COX-1 or COX-2 by NSAIDs may reduce the progression of the disease[10].
Diabetes mellitus
Diabetes mellitus is a chronic disease that leads to kidney damage. In this context, it has been found that inducing COX-2 in the proximal tubules may lead to an increase in PGE2 production and in protein excretion in diabetic mice[51]. Even so, studies are showing that COX-2 and PGE2 production were increased in patients with diabetes mellitus, leading to the use of a COX-2 inhibition which can attenuate different renal diseases[52].
In agreement with these findings, a study conducted in animal models has determined that, when administering rofecoxib, a selective COX-2 inhibitor, protenuria levels in Zucker rats normalized[53]. In contrast, there are reports in the literature that in Zucker rats (obese) and db/db mice (insulin resistant), renal COX-2 has been positively regulated[54,55].
Diabetic patients have an excessive polyuria and urinary concentration of PGE2. So, the use of a NSAID must be used to control the levels of PGE2 and adequate the urine concentration[27].
In the study of Liu and group (2016), Ibuprofen was administered in rats with renal impairment and type 1 diabetes. The use of ibuprofen was able to decrease urinary protein excretion, blood urea nitrogen, glomerular basement membrane thickening, renal fibrosis, COX-2, and interleukin 1(3 (IL-1P) level, suggesting that this drug can prevent the development of renal damage in the diabetic group[56].
Kidney inflammation
The expression of COX-2 enzyme is considerably increased in response to renal tissue inflammation[57]. When activated in pathological processes, there is the formation of proinflammatory cytokines (interleukin-1 and TNF-a) and reactive oxygen species (ROS), which play a critical role in the renal physiopathology[40,58]. There is also a positive feedback through the signaling pathways that promote the development and progression of the disease[40,58,59] .
Authors state that inflammation is the causal factor that collaborates with renal lesion, especially with PGE2 which, although in physiological conditions contributes to the maintenance of renal homeostasis[40,60,61], in pathological situations might be responsible for unleashing a deleterious process[40].
This difference in the PGE2 action in various clinical situations is due to its attachment to four receptors, EP1, EP2, EP3 and EP4[40,62]. EP1 is responsible for natriuresis and diuresis and regulation of blood pressure. Receptors EP2 and EP4 have been reported to activate production of adenylate cyclase and increase (cAMP) and to increase water reabsorption, on the other hand, EP3 have the opposite effects[27,61,63,64].
A study conducted in rats subjected to nephrectomy has found that, after 4 weeks, the animals presented increased levels of urea and creatinine, reduction of creatinine clearance, and disorders in the metabolism of calcium and phosphorus, highlighting significant kidney failure[40]. However, in rats where PGE2 production was suppressed, there was a notable and stable improvement in all those alterations, suggesting that the PGE2-induced inflammation may be involved in the progression of renal lesion[40].
The table 1 presents a summary of the COX (COX-1 and COX-2) system roles in the different clinical situations mentioned.
Conclusion
Both COX isoforms play important roles in the regulation of renal homeostasis. However, in different clinical situations, their functions might be altered. Thus, the exactly role of COX-1 and COX-2 and their inhibition by the use of NSAIDs still remains controversial. So, it is important that the health care professional be aware of these possible alterations in order to provide the best clinical course of action.











 texto en
texto en