Introduction
In the last decade, modern society has changed its eating habits drastically, a change that we were surely not prepared for. For patients with chronic kidney disease (CKD), the outlook is far more complex if we consider diet as essential to disease control and the prevention of health complications in all their evolutionary stages. Therefore, we must have an updated understanding that enables us to provide these patients with the best information and necessary measures to prevent them from placing enemies in their market baskets. Our objective in this document was first, to carry out an updated review on the implications of phosphorous in the health of kidney disease patients and additionally, through fieldwork, identify its presence in the foods most consumed in our surroundings containing high phosphate levels. With this, our group proposes the creation of more useful and convenient tools to achieve better health results in these patients even from the early stages of the disease.
Phosphorus and Kidney Disease
The discovery of phosphorus dates back to 1669 when the German alchemist Henning Brandt succeeded in isolating it from large quantities of urine as a white dust that glowed in the dark. The word phosphorus, according to its etymology means "bright light", from the Greek phosphoros formed by phos meaning light and phorus meaning carrier, "carrier of light"[1]. It is the second most abundant mineral in the body and represents approximately 1% of body weight. As for its distribution, 85% is in the skeleton, 14% at the cellular level and only 1% is available at the extracellular space[2]. The kidneys play a major role in maintaining the proper excretion of phosphorus in the urine to ensure that their serum levels are adequate for the performance of various functions. The fine balance between ingestion and renal elimination leads to early changes in the concentrations of this ion indicating a compromise of renal function, which allows us, promptly, to intervene patients to stop the foreboding course towards well-recognized complications such as secondary hyperparathyroidism (HPT). Scientists have long been concerned about finding the best strategies to prevent the increase of serum levels. Within these strategies, diet has been one of the most relevant fields of study. Here, we will review the physiology of phosphorus metabolism to discuss this subject better.
Because phosphorus is one of the most abundant minerals in the human body, it is related to important functions such as cell structure (DNA synthesis, membrane lipids, and intracellular signaling), cellular metabolism (ATP generation), maintenance of acid-base homeostasis and bone mineralization[3]. Reference values in adult plasma are in the range of 2.8 to 4.5 mg/dl2. The daily intake of phosphorus orally in the form of phosphate (PO4) varies between 700 and 2000 mg, according to the proportion of food ingested that is rich in this mineral. Once it is available for metabolism, it can be used at the cellular level or be deposited into the bone matrix or soft tissues. Its excretion can be renal at a value between 600 and 1500 mg/day or a lower value close to 500 mg[4] via the digestive tube. The western diet is rich in PO4; some high-content foods include red meat, fish, dairy and food additives. When these foods are ingested, they are absorbed in the small intestine. The active form of vitamin D (1,25 dihydroxy vitamin D) favors this action, which stimulates the activity of NaPillb at the brush border of the luminal membrane. It is important to note that this process is not easily saturable and that the greater the consumption of phosphates, the greater the absorption[5]. The kidneys also play an essential role in the homeostasis of phosphorus because of its ability to reabsorb phosphate from urine; an activity that is strongly influenced by phosphate intake in the diet. An adequate balance in these two systems can be achieved thanks to a specialized set of processes, in which vitamin D, calcium, parathyroid hormone (PTH) and phosphatonins, among others, intervene.
At the renal level, the control of phosphate reabsorption is regulated by these hormonal and metabolic factors[4] (Table 1), modifying the activity of the cotransporters at the proximal tubule level in a matter of minutes, hours or days depending on the type of co-transporter[6,7].
Table 1 Factors that alter renal regulation of phosphates
| Incremento de la absorción renal del fosfato | Disminución de la absorción renal |
|---|---|
| Low phosphate diet | PTH |
| 1,25 dihydroxy vitamin D | Phosphatins (Example: FGF23) |
| Diet rich in phosphates | |
| Metabolic acidosis | |
| Potassium deficiency | |
| Thyroid hormone | Glucocorticoids |
| Dopamine | |
| Hypertension | |
| Estrogens |
FGF23: Fibroblast growth factor 23
Taken and adapted from reference 4 (Clin J Am Soc Nephrol 2015; 10: 1257-1272).
Among the noteworthy hormonal factors in kidney disease, Fibroblast growth factor 23 (FGF23) and PTH carry out an important function, which starts from the impairment of kidney function when phosphorus levels increase in the body. Decreased glomerular filtration rate (GFR) increases serum phosphorus levels, which stimulates the production of FGF23 by osteocytes, which functions because of the presence of the Klotho cofactor produced in the kidney. Klotho activates the FGF1 receiver and decreases the activity of the carrier proteins NaPiIIa and NaPiIIc in convoluted proximal tubule cells. Simultaneously, it decreases the activity of 1-hydroxylase and increases that of the 24-hydroxylase, reducing the circulating levels of 1,25 OH vitamin D, leading eventually to a drop in the intestinal absorption of phosphorus and calcium, restoring norma-phosphatemia. However, the decrease in serum calcium levels stimulates the secretion of PTH, which stimulates phosphaturia by also inhibiting the activity of proteins NaPiIIa and NaPiIIc. PTH; unlike FGF23, it increases the activity of renal 1-hydroxylase, promoting the synthesis of active vitamin D and stimulating osteoclastic activity with greater bone resorption and restoration of serum calcium[4,8,9].
Like this, an effective reduction mechanism of serum phosphate levels is achieved through the two channels mentioned. Dietary intake is counterbalanced by an increase in excretion by nephron unit, achieving a neutral balance of phosphates4. However, the compensation mechanisms described are temporary, and the prolonged exposure to oral phosphate overload associated with a reduction in the nephron mass leads to excess tubular exertion and the generation of tubulointerstitial injuries. Hyperphosphatemia reflects a disparity in phosphate metabolism by renal failure imbalance between intestinal absorption and urinary excretion, which occurs when the GFR nears 30 ml/min; this shows the importance of a proper diet in avoiding the activation of all of the compensating mechanisms exposed[10].
After this brief review of the processes that take place and the agents involved in phosphate homeostasis, we can understand how its dietary consumption is a direct regulator of its renal reabsorption, as it is a protagonist in the course of CKD because of the development of bone and mineral disorders, cardiovascular incidents and the impairment of survival through years of evolution. This problem is well represented in high mortality rates that surpass 20% despite the introduction of dialytic therapy in the second half of the XX century[11]. Since then, the natural history of kidney disease has been rewritten introducing new challenges and new opportunities for intervention.
To respond to the most frequent questions of those who, in their daily work, face the challenge of providing their patients with the most favorable balance, we will discuss and present the evidence on CKD and phosphorus consumption. Moreover, we will suggest tools that are easily-applicable, effective and have a favorable impact on controlling the disease.
How to interpret serum phosphorous levels in patients with CKD.
The common measurement procedure to test serum levels is the valuation of concentrations of inorganic phosphorus in plasma, which is measured by most laboratories in mg/dl. The results of this test represent only a minuscule part of the total concentration of phosphorous in the body; this compartment shows the dietary intake of phosphorus that is added daily, which is then interchanged with tissues and organic phosphorus. Based on this, we can understand first, why measuring intracellular phosphorus (peripheral blood cells) is not practical in the regular clinical setting and second, the limitations of the common measurement procedure to predict intake. In the patient with CKD, the relationship between daily phosphorus intake and serum levels is well established; this is not the case with non-renal patients. A urinary phosphorus test is an additional tool to provide information on intake and intestinal absorption[12]. Hyperphosphatemia must be diagnosed when serum levels are above 4.5 mg/dl.
Can the intake of phosphorus lead to acute kidney disease?
Multiple publications have established the risk of developing acute phosphate nephropathy after exposure to oral solutions of sodium phosphate, sometimes, leading to irreversible kidney damage. These studies have revealed the following risk factors associated with this lesion: female, elderly, arterial hypertension, and established renal disease[13]. The histological characteristics of this lesion are calcium phosphate crystal deposits in cells and tubular epithelial lumens, interstitial inflammation, and fibrosis and apoptosis of tubular cells[14]. The risk in non-renal patients seems noticeably inferior. An assessment of the renal function of a healthy population preparing for colonoscopies showed no association with renal injuries and underscored the importance of hydration during this preparatory phase[15].
Phosphorus and the progression of CKD
In the initial stages of kidney disease, patients can maintain a balance, managing a normal level of phosphorus. As the GFR continues to descend, the impossibility of an adequate phosphate excretion becomes the pathophysiological substrate for mineral and bone disorders. However, the impact of hyperphosphatemia in the comorbidity of the renal patient has other important implications, thus, a large part of the efforts, to date, focus on recognizing its association with the progression of kidney disease, as well as other associated factors and the influence of the phosphorus load, independent from serum levels. A Spanish study published in 2011, described how renal function, to a greater degree, is influenced by sex (female), serum albumin, serum bicarbonate and the use of diuretics and they are significant determinants of serum phosphorus. This same study demonstrated the relationship between serum phosphorus and the speed of deterioration of renal function is more intense the higher the baseline residual renal function, which suggests the independent role of phosphorus or the related factors with phosphate concentrations[16]. Previously, though in a lesser number of publications, it has been described that phosphorus is a strong independent predictor of decreased renal functions in patients with advanced stages of CKD[17]. With the support provided by these findings, this relationship has been established in patients from stage 3. An observational study, which included 225 renal disease patients with a GFR median of 43 +/- 19 ml/min, demonstrated that basal serum phosphate predicts the progression of renal dysfunction during a follow-up of 2.5 years, after adjustments for the known factors of renal progression such as age, proteinuria and systolic blood pressure (SBP)[18]. In this way, the available evidence shows us that the association between phosphorus levels and the speed of renal progression is explained by the gravity of kidney disease.
Is hyperphosphatemia an independent cardiovascular risk factor in patients with CKD?
So far, it has been demonstrated that the classic cardiovascular risk factors such as obesity, hyperlipidemia, diabetes mellitus, hypertension, and smoking cannot satisfactorily explain the major cardiovascular risks in patients with CKD from the early stages of the disease and that high levels of serum phosphorus play a central role in the induction of vascular calcification. A classic study that is a necessary reference tool on this subject was published in the 70s; it describes autopsies performed on patients in advanced stages of kidney disease that had or had not undergone dialysis therapy.
It found that 79% of the patients on dialysis presented calcification in the mean of the vessels compared to 44% of the group who were not undergoing dialysis and whose heart vessels, lungs, stomach, and kidneys were more compromised[19]. Observational studies on individuals with or without clinically manifest kidney disease also show that the highest levels of serum phosphates are associated with cardiovascular disease and vascular calcification[20,23]. Another study concludes that the highest concentrations of phosphates in serum, though still within the normal range, were associated with a greater prevalence of vascular and valvular calcification in individuals with moderate CKD[24]; this provides relevant elements to continue the discussion on the influence of phosphorus loads and the progression of CKD, independently from the values of serum phosphates. Other findings on the effects of phosphorus relate it to increased left ventricular mass in patients with CKD, especially in female populations[25,26].
Can phosphorus control reduce the progression of CKD or improve the prognosis?
Patients with CKD face high mortality rates, therefore, the urgency in the scientific community to find modifiable risk factors that impact the natural history of the disease and quality of life. Phosphorus levels > 5.0 mg/dl have been associated with an increased risk of relative mortality in a population of patients on hemodialysis[11]. These findings are replicable in studies evaluating this aspect in patients with CKD not undergoing dialysis. A publishing appearing in 2005, which involved a study on a population of 6730 veterans during 1999 to 2002, found an association between high serum phosphate levels and the risk of mortality and acute myocardial infarction (AMI) in patients with CKD, irrespectively of renal function and other known confounders with a hazard ratio (HR) of death of 1.9 individuals with phosphorus levels > 5 mg/dl[27]. Of the results obtained in this study, special attention is drawn to the result that the risk increased from a serum level considered within the upper limit of normal (>4.5 mg/dl).
From this, we can clearly see the way strategies should be drawn to reduce serum phosphorus in patients with CKD. However, it is not appropriate to limit therapeutic efforts to initiate phosphorus chelators in patients that develop hyperphosphatemia. A new paradigm has emerged on phosphorus restriction that urges us to take measure detachedly from the levels reported in the blood. The ideal scenario to begin intervention is when the levels of FGF23 begin to rise (from the 2nd stage of CKD); this is the sign of excess phosphate intake in relation to the number of nephrons[28]. Early measures are necessary at this point to prevent tubular damage and hinder the progression of CKD[29]. The lowest levels of FGF23 have been attained in patients with early and advanced kidney disease subjected to low protein diets[30]. The relevance of monitoring using FGF23 serum levels rather than serum phosphorus arises as the former can remain within normal ranges and could explain why studies with large cohorts of early stage CKD patients have not been able to demonstrate an independent association of phosphorus serum and the risk of death and the progression of renal disease[31].
Phosphorus and diet
The relationship between daily dietary phosphorus intake and its contribution to outcomes of patients with CKD is significant. The assessment of the dietary intake of phosphorus is a challenge for clinicians because of the limitations in quantifying its presence in foods and the increasing global use of additives that are sources of inorganic phosphorus.
Phosphates in the diet are present in two forms: organic and inorganic phosphorus.
Organic phosphorus: is found in protein-rich foods both of animal and vegetable origin. Organic phosphorus is hydrolyzed in the intestinal tract and is later absorbed as inorganic phosphorus. Only 30 to 60% of inorganic phosphorus is absorbed, and its main sources are the meats of mammals, birds, fish, eggs and dairy products. Phosphorus from animal protein is found mainly in the intracellular compartment of foods, but it is more digestible than phosphorus from vegetable protein.
Phosphorus of plant origin is abundant in seeds, nuts, and legumes[32]. Unlike the protein of animal protein, vegetable protein is integrated with phytic acid or phytate, an organic acid whose main function is as an energy source for the plant. Phytic acid establishes strong ionic bonds with essential minerals in nutrition, forming insoluble chelates that cannot be absorbed by the body.
Humans and non-ruminant animals have a limited ability to hydrolyze phytic acid as they lack the phytase enzyme to separate the phosphorus from the phytate. Therefore, the availability of phosphorus from vegetable protein is low; usually, under 50%.
In patients with CKD, if they are administered the same amount of phosphorus from animal and vegetable foods, the vegetable protein will not be enough to cover their protein requirements, but it will help to better manage phosphorus even if it may result in an inadequate protein intake.
Inorganic phosphorus: is used mainly as an additive in foods. They have various applications and functions such as pH regulators, antioxidants, protein stabilizers, flavor enhancers, colorants, salts in cheeses, enhancers of dough and chemical yeasts, emulsifiers, humectants, leavens, sequestrants, and thickeners.
Foods such as drinks, frozen foods, breakfast cereals, processed and spreading cheeses, instant products, precooked foods, hot dogs, and cookies, among others, are all common sources of inorganic phosphates[33].
Inorganic phosphorus is not bonded to proteins; instead, it appears as salts (polyphosphates, phosphoric acid) that are quickly dissociated and easily absorbed in the intestinal tract. Between 90 and 100% of inorganic phosphorus is absorbed in the intestines as opposed to 40-60% of organic phosphorus in foods[34-37].
However, food producers are not required to list phosphorus content on food labels; this presents a problem for nutritionists when calculating the phosphorus intake of their patients. Moreover, with the global increase in availability of processed foods triggered by convenience, in turn, the consumption of inorganic phosphorus.
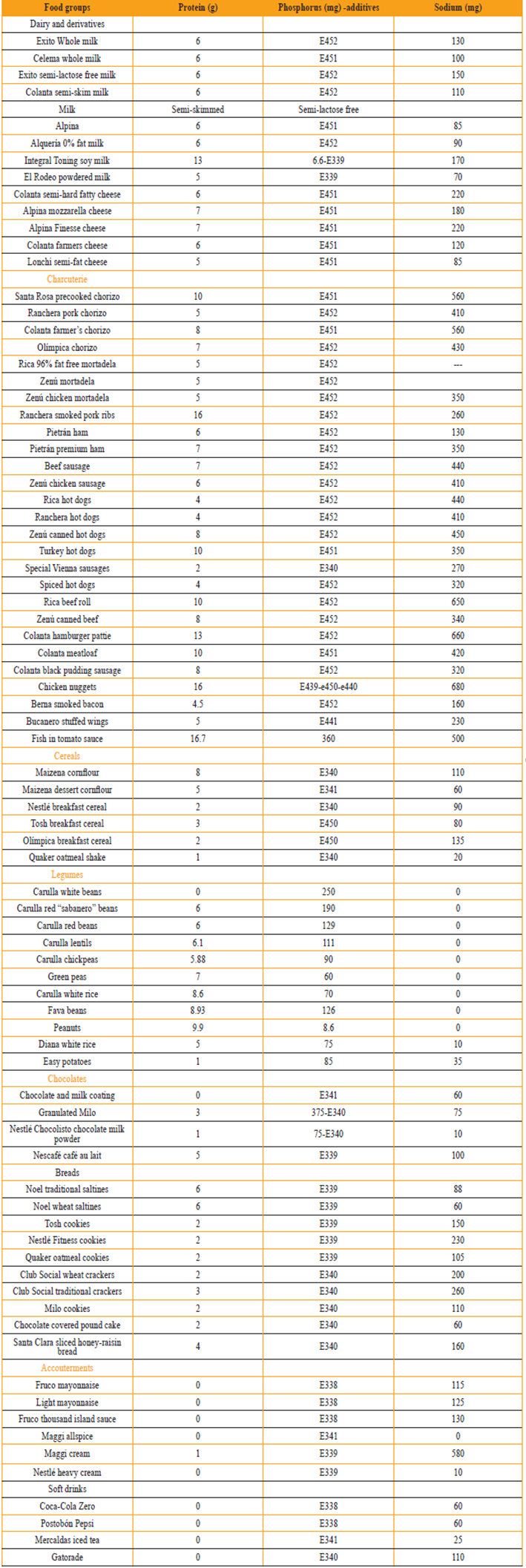
With this in mind, our workgroup regarded the need to calculate the total phosphorus content of foods to enable the structuring of more precise diets for patients since the early stages of the disease. For this purpose, field research was done to document the phosphorus content of foods previously recognized for their high phosphate contents in stores and supermarkets in the city of Manizales. Remarkably, the amount of added phosphorus (in milligrams or grams) did not appear on most of the products selected. This finding led us to search current legislation and found that manufacturers are not obligated to provide such important information. Furthermore, consumers easily dismiss the phosphoric additives that are included because they appear under multiple names. In Table 2 we list some of these with their respective codes under European Union standards.
Table 2 Phosphoric additives. The most important phosphates and derivatives
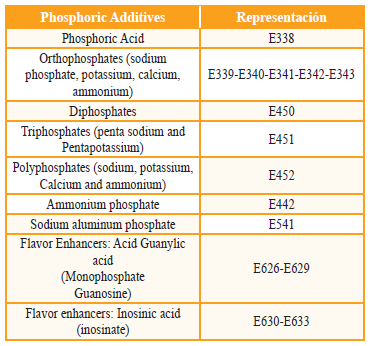
Source: REGULATION (EU) No 1129/2011 OF THE COMMISSION of 11 November 2011 Amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the Council to establish a list of food additives of the Union[38].
For this Community, in all the languages that conform it, the letter E identifies an additive, being understood as a food additive to that substance that, does not itself constitute a food or nutritional value, it is added intentionally to food and drink in minimal quantities to modify their organoleptic characteristics or to facilitate or improve their process of elaboration or conservation[38].
In our study, we used this coding to name the additives found in the products, chosen for their frequent consumption in our diet and for being recognized for high phosphorus content (Table 3). In the last table, it can be seen that the additive is indicated in all products without indicating their weight and only for grains was a numerical figure found (Figure 1).
The Food & Drug Administration of the United States (FDA) defines additive as "any substance the intended use of which results or may reasonably be expected to result (directly or indirectly) in its becoming a component or otherwise affecting the characteristics of any food." This definition includes any substance used in the production, processing, treatment, packaging, transport or storage of foods; it is divided into two groups. The first is direct food additives, which are those that are added to food for a specific purpose; for example, xanthan gum is used in salad dressings, chocolate milk, pastry fillings, puddings and other foods to add texture. Most direct additives are identified on the ingredients label. The second is indirect food additives that become part of the food in trace amounts because of packaging, storage or other types of handling. Food packaging manufacturers must show the FDA that all of the materials that come into contact with the foods are safe before they are allowed to use them for this purpose.
Furthermore, the FDA defines a color additive as any colorant, pigment or substance added or applied to food, drugs or cosmetics, or to the human body, that can (alone or through reactions with other substances) impart color. The FDA is responsible for regulating all color additives to ensure that foods that contain these additives are safe for consumption and that they contain only approved ingredients and are accurately labeled. (http://www.fda.gov/Food/IngredientsPackagingLabeling/FoodAdditivesIngredients/ucm094211.htm)
The FDA, however, does not require listing of phosphorus content, in its various forms, on food labels; it is included in the ingredients lists with names such as monocalcium phosphate and ammonium phosphate.
In the Colombian legislation, Resolution 333 of 2011 (February 10, 2011) of the Ministry of Social Protection, "which establishes the technical regulation on labeling requirements or nutritional labeling that food packaged for human consumption must meet", references phosphorus in Chapter III, "Disclosure of Nutrients"; Article 8, "Disclosure and Presentation of Nutrients "; section 8.2, "Optional Disclosure of Nutrients". In this section, subsection 8.2.4 states that "vitamins and minerals other than vitamin A, vitamin C, iron and calcium naturally present in food can be disclosed only when reference values have been set in chapter IV of this regulation and are present in quantities equal to or greater than 2% of the reference value per serving of food ". In Chapter VI, "Health Claims"; article 23, " Health Claims Related to the Reduction of Risks of Disease"; paragraph 23.1, "Calcium and Osteoporosis", states that "food must meet or exceed the requirements for the term or descriptor "high" in calcium, as defined in this resolution; be present in an assimilable form, and the phosphorus content cannot be higher than the calcium content." They are no other references to other aspects related to phosphorus in the resolution.
Among the possible reasons for the failure to report phosphorus contents in these foods is the rigorous chemical analysis that would be required to assess these foods, which can be considered impractical and costly.
A recent study sought to correlate the intake of phosphorus from organic and inorganic sources with serum levels, both in healthy and renal population. The study results showed a correlation between compromised renal function and low body weight with a significant positive effect on levels of phosphorus. The foods that had the most impact were dairy, cereals, and grains with added inorganic phosphates[40].
Studies like the previous evidence a greater impact of foods with phosphate additives, which contribute to all the implications and hinder the control of the renal patient. There is no doubt that education strategies in this group of patients will benefit therapeutic interventions. This notion was evidenced in a study involving patients in advanced stages of renal disease; their intake of phosphorus rich dietary additives was controlled, and significant improvements were achieved in the level of phosphorus serum (up to 0.6 mg/dl)[41].
Another group of studies questions the implications of low phosphate diets and, thus, in proteins that could affect the patients' state nutritional, generating a significant impact on their overall state of health[42,43]. One explanation for these findings can be found in the mechanisms explained in this review that recognizes the limitation of phosphate serum levels especially in early stages of the disease; hence, the urgency to find, in new markers, a better correlation for a more real control of these patients.
Management strategies to control phosphorus
The assessment of daily phosphorus intake is not under the full control of patients. There are vast limitations; beginning with the difficulty in identifying the content of this element in the most commonly consumed foods that are considered first-line components of the average Colombian diet. Therefore, management strategies should be focused on offering our patients simple recommendations to be constantly enforced by the multidisciplinary group in charge to modify dietary habits that can be sustained in the long-term and be adapted to the culture and financial means of our patients.
With this in mind, our work group suggests the implementation of an educational tool, adapted for Colombia, derived from the initiative of an Italian group of researchers and published in 2015. They propose a "phosphorus pyramid" to instruct patients, easily, on the foods to avoid because of their high phosphorus content, as well as those they should consume given their state of health44 (Figure 2).

Figure 2 Pyramid of phosphorus content in food. Taken and adapted from reference 44 (BMC Nephrol 2015 Jan 20; 16: 9).
The pyramid has seven levels in which elements are organized according to their phosphorus content, their phosphorus/protein ratio, and the bioavailability of phosphorus according to the food group. The organization of foods on the different levels allows patients to easily select foods without having to memorize the phosphate content of each one. Each level has a different color (from green to red) according to the recommended intake frequency, which varies between "no restriction" and "avoid as much as possible." The base or first level of the pyramid (green) shows foods with very low phosphorus content (sugar, olive oil, protein free foods) or very low bioavailable phosphorus (fruits and vegetables). It also includes egg whites, which have a very favorable phosphorus/protein proportion and are a source of protein with a high biological value and no cholesterol.
Intake of this food is unrestricted. However, during a consultation, the health professional must consider the following special considerations and warnings:
Patients with diabetes, who should avoid sugar and not exceed fruit consumption.
Patients who are overweight or obese should reduce sugar, olive oil, vegetable fats, and butter.
Patients on dialysis should limit fruits and vegetables to avoid over intake of potassium.
Lastly, protein-free products should be recommended for patients with CKD on dialysis that require protein restrictions, but high energy consumption.
The second level mainly includes plant foods that are rich in phosphorus, but mainly as phytate, therefore, with lower intestinal absorption. The recommended daily dose is 2 to 3 portions per day. The third level includes food of animal origin and milk and dairy products, with a low phosphate/protein ratio. The recommended daily dose is no more than 1 serving per day. The fourth level shows foods with phosphorus content higher than the proportion of protein. The recommended daily dose is one serving per week. The fifth level contains foods with high content of phosphorus. The recommended dose no more 2 to 3 serving per month. Finally, at the top of the pyramid, the sixth and seventh level includes food with additives with a high content of phosphorus that should be avoided as much as possible. The image of the pot on the left side suggests boiling as the best method of cooking to reduce phosphorus content. This process leads to the demineralization of food, thus reducing the content of some minerals such as phosphorus, sodium, potassium and, calcium, both in vegetables and animal products. The degree of mineral loss is proportional to the amount of boiling water used, the size of the pieces, cooking time and whether the vegetable foods are peeled. After boiling, the reported reduction of phosphorus has been 51% for vegetables, 48% for legumes, and 38% for meat. Note that boiling reduces the content of phosphorus and an insignificant loss of nitrogen, leading to a more favorable phosphorus/protein ratio[45,46].
CONCLUSIONS
The challenge we face is sizeable and subject to the influence of new scientific evidence. We have a clear understanding of the harmful effects of phosphorus on the health of patients with kidney disease, which has caused some authors to call it the "hidden enemy" because of its undetected presence in foods of daily consumption and the obvious limitation in the fulfillment of the control strategies proposed by professionals in charge. An inventive and improved method to instruct patients on the dietary restrictions of phosphates is therefore required[47], even those in early stages of the disease, as it is the only alternative we have to impact the prognosis of these patients before hyperphosphatemia occurs; as an alternative, we also need pharmacological therapy[48,50].











 text in
text in