Introduction
Chronic kidney disease (CKD) is a global public health problem with high prevalence. The identification of the various risk factors related to it is essential to prevent the development and growth of the phenomenon. CKD is, in general terms, the result of various heterogeneous disorders that alter the renal structure and function. The variation of the expression of the disease is directly related to previous pathologies, structural lesions, degree of severity and time of progression.
The association between the exposure to the environment and the development and progression of this disease has been demonstrated through epidemiological studies.
Some forms of CKD have been linked to environmental nephrotoxins, such as exposure to heavy metals, mycotoxins produced by fungi in stored foods, air contaminants and pesticides.1
In the past 50 years, the study of the toxic effects of heavy metals in humans has gained particular importance, since large quantities of these products, discarded as part of the industrial activity, are not biodegradable and persist in the environment for long periods of time.
The exposure to cadmium, lead, and to a lesser extent, to mercury constitute a particular concern, since many epidemiological studies have shown a strong association between the exposure to these heavy metals, the markers of kidney damage and the progression of CKD. The exposure to arsenic continues to be a major public health problem, since millions of people are exposed to water sources with levels above the limit.2
Methodology
In order to evaluate the role of these contaminants in kidney disease, it was carried out an analysis based on studies on the pathophysiology, mechanisms of kidney damage and works in humans and animals. All of them, associated with the exposure to these heavy metals.
Searches in PubMed (http://www.ncbi.nlm.nih.gov/pubmed) were conducted in order to find all published studies evaluating the relationship between kidney disease and exposure to cadmium, arsenic, lead and mercury.
Pathophysiology and mechanism of kidney damage
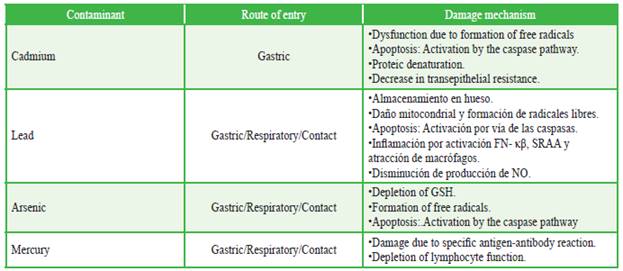
The pathophysiological mechanisms of kidney damage caused by the different environmental contaminants are complex. Several aspects related to their metabolism and mechanisms of body damage are not fully known. To understand their nephrotoxic effects, these mechanisms will be analyzed in a nutshell for cadmium (Cd), lead (Pb), arsenic (As) and mercury (Hg) (Table 1).
Table 1 Mechanism of damage due to heavy metal poisoning in animals. .
| Contaminant | Route of entry | Damage mechanism |
| • Apoptosis: activation by the Caspase pathway. | ||
| Cadmium | Gastric route | • Inhibition of ACE. . |
| • Decreased production of NO. | ||
| Sulfur dioxide | Respiratory route | • Multi-organ DNA damage. |
| Arsenate | Respiratory route | • Alteration of the transport of sodium, phosphate, glucose, andPHA. |
ACE:Angiotensin converting enzyme, NO: Nitric oxide, DNA: Deoxyribonucleic acid, PHA: Paraaminohypurate.
The cadmium that is ingested is bound to the metallothionein (MT) and phytochelatin proteins, which are catalyzed by the hydrochloric acid (HCl) in the stomach, releasing the Cd for its absorption in the jejunum-ileum by the DMT1 (divalent metal transporter 1) and ZIP-8 (Zinc transporter protein 8) transporters.3,4 In the bloodstream it binds to albumin and is transported to the liver, where it binds to glutathione (GSH) and metallothionein-1 (MT1).The Cd-MT1 complex is secreted in the bile; however, it returns to the bloodstream through the enterohepatic circulation.
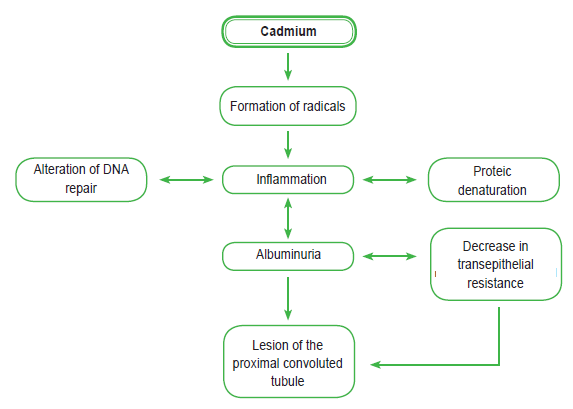
Within the cells of the proximal convoluted tubule (PCT), the Cd-MT1 complex is stored and degraded in the lysosomes. Subsequently, the free Cd is transported to the cytoplasm by the lysosomal DMT-1.5 The activation of the protein kinase C increases the expression of the DMT-1 transporter and, therefore, the tubular toxicity of Cd.6 The free CD is accumulated in the mitochondria, inhibiting the respiratory chain in the complex III. This results in mitochondrial dysfunction and formation of free radicals, which activates the caspase pathway with subsequent apoptosis. In addition, it binds to the sulfhydryl groups in the proteins creating their denaturation (Figure 1).
It has been demonstrated that Cd interferes with the enzymatic activities of the calcium/calmodulin complex, which inhibits the action of Na-KATPase and stimulates the activity of MAPKs (mitogen-activated protein kinases). In the paracellular junctions, it affects the distribution of the proteins and reduces transepithelial resistance.7,8
Another mechanism of nephrotoxicity is that mediated by the formation of antibodies against MT. The exposure to Cd increases the production of MT in the liver and the kidney, which constitutes a defense mechanism to limit its toxicity. However, once the capacity of MTs to store Cd is exceeded, the latter, in its free form, induces the formation of anti-MTs, which are also toxic to the PCT cells.9
Lead is absorbed, mainly, by ingestion and inhalation and, to a lesser extent, through the skin. The intestinal absorption is mediated by the DMT-1 transporter and increases with the deficient intake of iron (Fe) and zinc (Zn). Once in the blood, 99% of the Pb binds to the proteins of the red cells and is distributed to the soft tissues and the bone. This is the main reservoir of Pb in the body, and in periods of bone remodeling, such as adolescence and pregnancy, the passage of Pb into the blood is increased.10
The Pb, bound to low molecular weight proteins (<1% of the total), is freely filtered through the glomerulus and is reabsorbed by the PCT cells by the process of endocytosis. Intracellularly, the Pb causes mitochondrial damage, formation of free radicals, intracellular depletion of GSH and apoptosis.9 The Pb affects enzymatic reactions in which calcium intervenes, even the calcium-sensing receptor can be activated by Pb, suggesting other mechanisms of nephrotoxicity of this metal.10,11 in the body, the urinary excretion is the main route of loss of Pb.
Pb induces the activation of the nuclear transcription factor-κβ, the activation of the intrarenal renin-angiotensin system and the attraction of macrophages. This generates an inflammatory process in the renal interstitium, which could be involved in the development of tubulointerstitial damage and arterial hypertension.12
In endothelial cells, it has been demonstrated that the increase in the formation of free radicals by Pb decreases the production of nitric oxide and the expression of the enzyme guanylate cyclase. This allows to explain the pathogenesis of arterial hypertension induced by this metal.11-16 In addition, it stimulates the activity of NADPH oxidase increasing the production of hydrogen superoxide and peroxide, which affects the oxidative stress and the intracellular oxidation reduction potential.
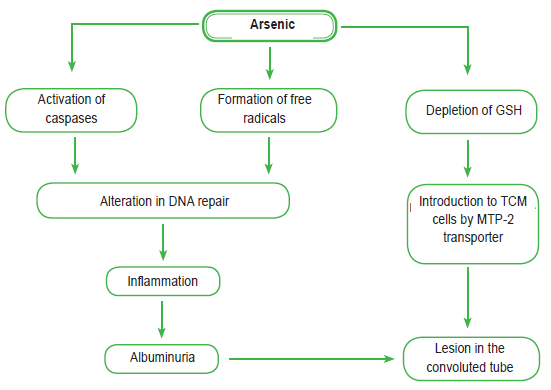
Arsenic is absorbed by ingestion, inhalation and, in smaller amounts, by the skin. Once absorbed, it is distributed throughout all the tissues of the body. The intake of selenium and vitamin B decreases the intestinal absorption of As. It is methylated in the liver in a process mediated by GSH, which decreases its toxicity and facilitates its biliary and urinary excretion.17
The As enters inside the cell through the aquaglyceroporins AQ3 and AQ9. In the liver, the latter is important for the biliary excretion of As.18
The MRP-1 and 2 (ATP binding cassette-multidrug resistance protein) are another group of As transport proteins in the liver, where they transport the As conjugated with GSH to the bile. The MRP-2 transporter is also located in the cells of the proximal tubule, which favors the introduction of As into these cells.19 The toxicity of As in PCT cells is due to the depletion of GSH and the increase in the oxidative activity of free radicals (Figure 2).
Mercury causes toxicity to the body through the skin, inhalation or ingestion. It has a great affinity for the renal tissue. Nephrotoxicity is commonly manifested as nephrotic syndrome and tubular lesion.20,21 The nephrotic syndrome associated with Hg toxicity is reversible after its complete elimination.
Hg induces membranous nephropathy in the long term, due to its small concentrations in cosmetics and occupational contact. The mechanism of renal damage that conditions glomerulopathy due to Hg is not well clarified. It has been suggested that the nephrotic syndrome subsequent to Hg exposure is a non-specific antigen-antibody immune reaction to heavy metals. Mercuric chloride and monomethylmercury inhibit the function of human lymphocytes, including proliferation, expression of cells activated by surface markers and production of cytokines.22-26
Studies in animals
Several studies conducted in animals demonstrate the association between the exposure to the different environmental contaminants and their nephrotoxic effects.
With respect to cadmium, in an experimental model of rat mesangial cells, cellular apoptosis was induced by means of the extrinsic or the mitochondria-mediated (intrinsic) pathways and it was found that camptothecin initiates the intrinsic pathway, with the activation of caspase 9 and cleavage of procaspase 3.There, the tumor necrosis factor gives rise to the activity of caspase 8 and the cleavage of procaspase 3 as a point of convergence of the two pathways. Such study concludes that the concentration of 10 μM of Cd can prevent the fragmentation of DNA (deoxyribonucleic acid), demonstrating that Cd is antiapoptotic in the rat mesangial cells, acting by a mechanism that can, in general, inhibit the caspases.27 In another study, it was found that Cd inhibits angiotensin-converting enzyme (ACE) at low, medium and high doses without a dose-response effect in rats with paradoxically induced hypertension. It was postulated that the vascular effects of Cd prevailed over its central effects in hypertensive rats.28
The role of the kidney injury molecule 1 (Kim-1), a transmembrane glycoprotein excreted in the urine after injury caused by exposure to a nephrotoxic agent, particularly cadmium (Cd), has also been investigated. In one study, the Kim-1 was used as a biomarker to determine the nephrotoxicity of Cd in male rats. In the study, Cd was administered daily for twelve weeks, determining weekly Kim-1, proteins, creatinine, metallothioneins and Clara cell proteins (CC-16) in urine. Significant levels of Kim-1 were detected in the urine at six weeks, which continued to increase throughout the treatment period. The appearance of Kim-1 occurred four to five weeks before the appearance of proteinuria and one to three weeks before the appearance of metallothionein and CC-16.The results suggest to use Kim-1 as a biomarker in the early stages of the Cd-induced lesion in the proximal tubules.29
When studying sulfur dioxide (SO2) as an environmental contaminant, it has been demonstrated that, in rats, the exposure to it causes damage to the DNA of multiple organs and that SO2 derivatives are agents that damage systemic DNA. It is suggested that the exposure to SO2 has a potential risk of DNA damage in multiple organs of mammals and could be related to carcinogenesis or mutation of the genomic DNA.30
It has been noted that arsenate acts at the level of the transport of sodium, phosphate, glucose, and para-aminohippurate (PHA), as well as in the oxidative metabolism in the proximal convoluted tubule of kidneys of rabbits and reduces the net transport of sodium and phosphate. The glucose transport is slightly inhibited, while the secretion of PHA is not affected31
Carbon monoxide (CO) has been studied in kidneys of rats, where it was observed that this gas increased when ferrous 5-aminolevulinic (5-ALA/ Fe2) was administered and, in blood, the maximum concentrations of creatinine and urea nitrogen decreased. In rats treated with 5-ALA/Fe2, the tubular damage and the number of apoptotic cells significantly decreased. These findings suggest that 5-ALA/Fe2 protects the kidney from reperfusion injury in renal ischemia, by reducing macrophage infiltration and decreasing renal cellular apoptosis through the generation of CO.32
Studies in humans
In several epidemiological studies has been postulated the association between the exposure to arsenic (As) and arterial hypertension and other cardiovascular diseases.33 In experimental studies they were exposed to high doses of this compound. In an epidemiological study based on passive information in Utah, Lewis et al.34 found an increase in the mortality rate in kidney disease in men, but not in women, due to exposure to high concentrations of As. Meliker et al.35 found that As in potable water exceeding 200-300 mg/L is associated with high mortality rates in diabetes mellitus, cerebrovascular disease and kidney disease in men and women.
Hsueh et al.36 studied in Taiwan 125 individuals with glomerular filtration rate (GFR) < 60 ml/min and 229 individuals with normal renal function. They found a weak association between the urinary levels of As and the decrease in renal function (r2 = 0.04, p < 0.001). In this study, the high renal excretion of As was linked to a lower level of lycopene in plasma, showing that kidney damage was measured by increased oxidative stress.
As is a promoter of inflammation, oxidative stress and endothelial dysfunction by different mechanisms, including the activation of transcription factors such as protein-1 and nuclear factor κβ.37-40 The biological mechanisms responsible for As-induced diabetes mellitus remain to a large extent unknown. Recent evidence suggests that trivalent arsenicals can suppress the insulin stimulation for glucose uptake by interfering with the mobilization of glucose transporters in adipose cells.41 They can also interfere with the transcription factors involved in the expression of the insulin-related gene.42
To evaluate its nephrotoxicity, several studies have been carried out in populations exposed to Cd, with different degrees of sensitivity and clinical importance. Among these, elevated urinary albumin in the absence of increased excretion of low molecular weight proteins, is an early indicator of glomerular damage and is observed at concentrations of 3.6 to 4.2 μg Cd/g of creatinine in urine.43-44 Low molecular weight proteinuria in people exposed to > 10 μg Cd/g creatinine in urine is irreversible and exacerbates the age-related decline in GFR.45-47 The elevation of the enzymes N-acetyl-ß-D-glucosaminidase (NAG) and alanine aminopeptidase (AAP), has been associated with renal tubular damage with occupational exposures to Cd between 3.7 and 6.3 mg Cd/g creatinine in urine 44,45,48
Liang et al.49 demonstrated that the exposure to the low environmental level of lead (Pb) can accelerate the progressive renal failure in patients without diabetes, but who suffer from chronic kidney disease. The authors observed 202 patients with chronic renal failure during 24 months. Subsequently, 64 subjects with high levels of Pb in blood were divided, randomly, into two groups. During three months, the patients of the 'chelation' group received therapy with calcium disodium ethylenediaminetetraacetic acid (EDTA).Meanwhile, those in the 'control' group received placebo. Based on their results, they suggest that repeated chelation therapy can improve kidney function and delay the progression of renal failure.
Sommar et al.50 conducted a study where they correlated Pb, Hg and Cd with CKD. They analyzed samples from 118 patients to determine the concentrations of Cd, Pb and Hg. Performing a prospective analysis, they found that erythrocytic levels of Pb are associated with CKD, Cd levels increase the development of CKD, while mercury levels are not associated with CKD. However, they mention the need for more studies to assess causality. Gender-specific analyzes suggest possible differences in the susceptibility or reliability of the exposure to the biomarker.
Ni et al.51 conducted tests with cultured human coronary endothelial cells and vascular smooth muscle cells, in which they investigatedreactive oxygen species (ROS) produced by these cells when they are exposed to lead. They found that the exposure to Pb promotes generation of hydrogen superoxide and peroxide in human coronary endothelial cells and in vascular smooth muscle cells. This phenomenon may contribute to the pathogenesis of hypertension and cardiovascular diseases associated with Pb. The authors point out, therefore, the potential benefit of reducing the burden of Pb in the exposed populations (Table 2).
Treatment
There is a lack, to a large extent, of therapeutic measures that lessen the systemic effects of the different contaminants mentioned in this review. In general, there are no established treatments to reduce the levels of these components in the blood or to prevent their toxicity. However, many therapies and functional foods have been tested on animals in order to prevent the nephrotoxicity, for example, of arsenic. The best known is resveratrol, which significantly attenuates the accumulation of arsenic in the renal tissues, decreases the markers of oxidative stress and shows less morphological lesions and tubular necrosis.52
Likewise, other compounds such as naringenin, a natural citrus flavanone and the green tea extract have shown promising effects that protect the kidney against nephrotoxicity.53
For this reason, greater vigilance is needed to reduce the environmental concentrations of these compounds.
Conclusions
In conclusion, the pathophysiological mechanisms of the mentioned contaminants allow, through their toxicity, to know the different effects on the kidney. An increasing number of evidences, obtained both from humans and animals, suggests that these contaminants are a major risk factor for morbidity and that they cause, in most cases, renal tubular alterations, specifically in the proximal convoluted tubule, and, sometimes, acute renal failure, which may progress to chronic kidney disease. This is one of the clinical characteristics associated with chronic exposure. Lead, mainly, can increase the risk of reaching terminal stages, although more studies are needed to evaluate this causality.
Due to the high presence of these contaminants in the environment, as well as the lack of therapeutic measures to reduce their effects, it is necessary to carry out more longitudinal studies to evaluate the relationships of the exposures to such metals with kidney damage. In this way, it would be possible to know the chronic effects and greater environmental surveillance could be done to reduce the concentrations of these compounds. For this reason, it is urgently required to reduce the amounts of heavy metals in industry, mining and the agricultural and livestock sector, particularly in cases where they provide less usefulness. Thus, finally, we consider pertinent the union of efforts between the public and private sector, together with research centers, to advance works that explore and investigate other less toxic substances or metals that can replace or minimize the impact of the heavy metals described in this work.
Ethical responsibilities
Protection of people and animals
The authors declare that no experiments were performed on human beings or animals for this research.
Data confidentiality
The authors declare that they have followed the protocols of their workplace on the publication of patient data.
Right to privacy and informed consent
The authors state that patient data do not appear in this article.
Contribution of the authors
Juan Daniel Díaz García: contributed with the original idea and the process of preparation of the review regarding the search for references, selection of information, structure of the review (abstract, introduction, development, conclusion, bibliography, tables and figures) and changes during its publication process
Emmanuel Arceo: contributed with the search and development of the review.











 text in
text in