El tratamiento con glucocorticoides en pacientes con síndrome de dificultad respiratoria aguda. Una revisión sistemática de la literatura y metaanálisis
Glucocorticoid treatment in patients with acute respiratory distress syndrome. A systematic review and meta-analysis
Nelson Darío Giraldo Ramírez1; Augusto Quevedo Vélez2; Lord Larry Posada Uribe3; César Augusto Rodríguez Quijano4
1 Médico Intensivista, Unidad de Cuidados Intensivos adultos, Hospital Pablo Tobón Uribe, Medellín, Colombia.
]]> 2 Profesor de Pediatría, Departamento de Pediatría, Unidad de Cuidados Intensivos Niños, Hospital Universitario San Vicente Fundación y Universidad de Antioquia, Medellín, Colombia. quevedoaugusto@gmail.com3 Profesor de Cirugía de Tórax, Departamento de Cirugía, Facultad de Medicina, Universidad de Antioquia, IPS Universitaria, Medellín, Colombia.
4 Médico Ginecoobstetra, Departamento de Ginecología y Obstetricia, Hospital Manuel Uribe Ángel, Envigado, Antioquia, Colombia.
Recibido: enero 30 de 2012
Aceptado: julio 16 de 2012
RESUMEN
]]> Introducción: el síndrome de dificultad respiratoria aguda (SDRA) es secundario a inflamación originada en una enfermedad pulmonar primaria o una afección extrínseca al pulmón. Es frecuente en cuidado intensivo y conlleva alta mortalidad.Objetivos: determinar la eficacia y seguridad de los glucocorticoides en dosis bajas en personas mayores de 18 años con SDRA, en términos de mortalidad, días libres de ventilación mecánica, incidencia de infecciones nosocomiales, neuromiopatía y sangrado digestivo.
Metodología: se hizo una búsqueda sistemática de ensayos clínicos controlados que compararon glucocorticoides con placebo, en adultos con SDRA en los desenlaces descritos. También, búsqueda secundaria de ensayos clínicos referenciados en los artículos primarios.
Resultados: se encontraron siete ensayos clínicos. Se demostró disminución de la mortalidad hospitalaria al día 28 (OR: 0,56 [0,38-0,81], ganancia de 3,5 días libres de ventilación mecánica, disminución en la incidencia de infecciones nosocomiales y neumonía adquirida en el hospital. No hubo diferencias en la neuromiopatía asociada con esteroides, pero sí una tendencia, no significativa, al aumento del sangrado digestivo.
Conclusión: los esteroides en dosis bajas disminuyen la mortalidad al día 28 de los adultos con SDRA y aumentan los días libres de ventilación mecánica sin aumentar los efectos adversos significativos.
PALABRAS CLAVE
Esteroides, Ensayo clínico controlado, Síndrome de Dificultad Respiratoria del Adulto (SDRA)
SUMMARY
Background: The Acute Respiratory Distress Syndrome (ARDS) is a lung inflammation secondary to primary or extrinsic pulmonary pathology. It is a common disease in the intensive care unit and its mortality rate is high.
Objectives: To determine the efficacy and safety of corticosteroids in patients with ARDS older than 18 years, in terms of mortality, mechanical ventilationfree days, and safety in regard to nosocomial infections, health-care related pneumonia, neuromiopathy, and gastrointestinal bleeding.
]]> Search methods: A systematic search of electronic and manual literature was done, without restriction of language, of controlled clinical trials involving adults with ARDS, randomized to placebo vs. steroids, and that measured the outcomes described.Results: Seven clinical trials were found showing a decrease in hospital mortality (OR 0.56 [0.38-0.81], 3.5 more days free from mechanical ventilation, a decrease in nosocomial infections and in hospitalacquired pneumonia. There were no differences in the presentation of steroid-associated neuromiopathy. There was a non-significant tendency to increase in bleeding from the digestive tract.
Conclusion: Low-dose steroids seem to have a beneficial effect on mortality and ventilator-free days in adult patients with ARDS with no increase in adverse effects.
KEY WORDS
Adult Respiratory Distress Syndrome (ARDS), Controlled Clinical Trial, Steroids
INTRODUCCIÓN
El síndrome de dificultad respiratoria aguda (SDRA) fue descrito por primera vez en 1967 (1) y se caracteriza histológicamente por reacción inflamatoria alveolar difusa, aumento de la permeabilidad vascular, colapso y pérdida del tejido alveolar disponible para el intercambio gaseoso (2). Esta inflamación afecta también la membrana capilar alveolar pulmonar (3). La característica clínica más importante es el síndrome de dificultad respiratoria acompañado por hipoxemia grave. Los individuos afectados tienen aumento de la mezcla venosa y del espacio muerto, disminución de la distensibilidad (compliance) estática y aumento del trabajo respiratorio.
El SDRA se puede originar por lesiones pulmonares directas como neumonía, broncoaspiración, inhalación de humo y contusión pulmonar, o por mecanismos indirectos secundarios a enfermedades extrapulmonares como sepsis, traumatismos y pancreatitis, o a transfusiones de hemoderivados. Los criterios diagnósticos iniciales del SDRA se establecieron en el Consenso Americano-Europeo de 1994. Se deben cumplir todos los siguientes requisitos: enfermedad inflamatoria desencadenante (factor de riesgo), comienzo agudo (menor de 72 horas), radiografía simple de tórax con infiltrados alveolares en los cuatro cuadrantes, relación PAO2 / FIO2 menor de 200 y presiones normales de llenado ventricular (presión en cuña pulmonar menor de 18 mm Hg) o sin sospecha de disfunción auricular izquierda (4). En el Consenso de Berlín (2012) se modificaron los criterios diagnósticos del SDRA y ahora la definición incluye: comienzo dentro de los siete días de haber presentado el factor de riesgo, infiltrados bilaterales en la radiografía de tórax, falla respiratoria no explicada por disfunción cardíaca o sobrecarga hídrica, presión positiva al final de la espiración (PEEP) de cinco o más cm de agua (mBar), compromiso en la oxigenación definido por la relación PAO2 / FIO2 menor de 300. Además, se clasifica el SDRA según su gravedad en leve con PAO2 / FIO2 menor de 300, moderado con PAO2 / FIO2 menor de 200 y grave con PAO2 / FIO2 menor de 100. El SDRA grave debe incluir también PEEP de diez o más cm de agua (mBar), infiltrados bilaterales por lo menos en tres de los cuatro cuadrantes en la placa de tórax, distensibilidad (compliance) estática menor de 40 cm H2O y volumen minuto mayor de 10 L/min (5-7).
]]> El SDRA cursa en cuatro fases: temprana o exudativa, proliferativa, de lesión vascular y fibrótica. La evolución a esta última es un marcador de mortalidad que es cercana al 60% en quienes llegan a ella (8). El uso de esteroides podría teóricamente limitar la evolución del proceso fibrótico y disminuir la mortalidad y los días de ventilación mecánica. Además, la terapia con glucocorticoides está ampliamente disponible y es económica.Los pacientes con SDRA tienen una actividad disminuida del receptor glucocorticoide lo cual permite la sobreexpresión y transcripción del receptor nuclear KB con persistencia de actividad inflamatoria exagerada, a pesar de niveles séricos elevados de cortisol. Esta condición se ha denominado Insuficiencia de corticoides relacionada con el paciente crítico (CIRCI) (9). El uso de esteroides exógenos disminuye la inflamación y aumenta la reparación tisular (10).
En el SDRA la inflamación demostrada por el infiltrado inflamatorio obtenido de los lavados broncoalveolares (LBA) dura más de dos semanas y parece explicar el efecto desfavorable de la interrupción brusca del tratamiento con esteroides antes de este tiempo, y por esto se recomienda desmontarlo paulatinamente en cuatro semanas (11,12).
En los dos trabajos de Meduri (13,14) la recomendación es usar los esteroides por 14 días, seguidos de un período de disminución gradual hasta suspenderlos el día 28.
Los esteroides mejoran la función pulmonar cuando se continúan hasta por 18 días después de la extubación (13,14). El estudio de la SDRA Network con los esteroides no mostró mejoría en la mortalidad de los pacientes tratados. Sin embargo, esto pudo relacionarse con que los esteroides se suspendieron 12 a 36 horas después de la extubación (15).
Existen dos metaanálisis con recomendaciones divergentes en cuanto al beneficio de los esteroides en el tratamiento del SDRA temprano. Su recomendación obedece más a consenso de expertos. Es necesario evaluar las nuevas evidencias disponibles para ayudar a los clínicos en la toma de decisiones (16).
OBJETIVOS
Determinar la eficacia y seguridad de los glucocorticoides en el paciente adulto (mayor de 18 años) en términos de mortalidad al día 28, días libres de ventilación mecánica al día 28, infecciones nosocomiales, neumonía asociada al ventilador, sangrado del tracto digestivo y neuromiopatía.
]]> MÉTODOS DE BÚSQUEDA
Se realizó la búsqueda de acuerdo con los lineamientos de la Guía Iberoamericana Cochrane y se revisó según el PRISMA (17). Se hizo la búsqueda sin restricción de idioma, estado de publicación y forma de presentación, en las bases de datos PubMed, Web of Science, EMBASE, Cochrane-CENTRAL y DARE, LILAC, GoogleScholar y clinicaltrial.gov, y en páginas de internet de registro de ensayos de Japón, Australia y Nueva Zelanda. Además, se efectuó búsqueda de bibliografía de los artículos de revisión de los últimos tres años en las revistas NEJM, Lancet, BMJ, JAMA, Chest, Critical Care Forum, Critical Care Medicine, AJRCC, Intensive Care y AJCCM.
Los términos de Búsqueda MeSh y el tipo de texto aparecen en el apéndice 1.
CRITERIOS DE SELECCIÓN
Características de los estudios
Ensayos clínicos con asignación aleatoria que cumplían las características PICOS (población, intervención, comparación, desenlace y tipo de estudio) que compararan todas las causas de mortalidad, días de ventilación mecánica, infecciones nosocomiales, entre estas neumonía; además, hemorragia del tracto digestivo que necesitó intervención como transfusión o endoscopia, en adultos (mayores de 18 años) con diagnóstico de SDRA según los criterios del consenso Americano- Europeo y que hayan sido asignados aleatoriamente a recibir terapia estándar con esteroides o sin ellos.
Características de los reportes
Ensayos clínicos con asignación aleatoria, sin restricciones de idioma ni en cuanto a estados, forma o tiempo de publicación.
]]> COLECCIÓN DE DATOS Y ANÁLISIS
Extracción de datos
Dos revisores independientes extrajeron la información de los ensayos clínicos, incluyendo autores, año de publicación, localización del estudio, número de pacientes, características basales de los pacientes como diagnóstico de la enfermedad de base y tipo de SDRA (intrínseco o extrínseco).
Medición de riesgos de sesgos
La medición del riesgo de sesgos se hizo con base en la recomendación de Cochrane (18), que califica cada uno de los ítems siguientes, como riesgo alto, bajo o no claro:
El análisis estadístico se realizó tanto con el modelo de efectos fijos como aleatorios y se reportan ambos.
La medición de la heterogeneidad (19) se hizo gráficamente y con el estadístico I2.
Análisis de subgrupos
Se exploró con base en el comportamiento biológico de los subgrupos así: estudios clínicos con mayor y menor riesgo de sesgos, SDRA intrínseco o primario y el extrapulmonar. Se hizo prueba de heterogeneidad y se reporta el I2 así: menor de 40% es leve, entre 40% y 60%, moderado y mayor de 60%, importante (20).
El análisis estadístico se hizo con el software RevMan 5.1 de la colaboración Cochrane, su redacción siguió las pautas de la guía PRISMA (17) y la bibliografía se editó con el software Mendeley.
RESULTADOS
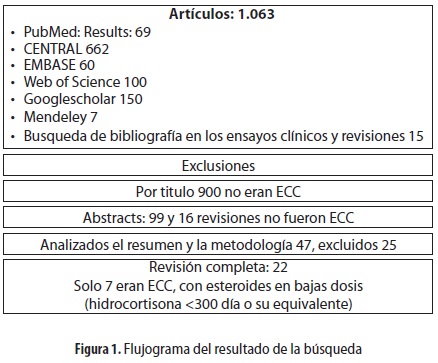
]]> Descripción de los estudiosEn la búsqueda electrónica y manual se hallaron 1.063 estudios, de los cuales siete fueron ensayos clínicos controlados con dosis bajas de esteroides (figura 1) (13,21-26). Además encontramos una actualización del estudio de Steinberg 2006, publicada en 2009, en la que se analizó la neuromiopatía asociada al SDRA (27).
Hubo acuerdo entre los evaluadores en la decisión de incluir o excluir los artículos y en el juicio de los riesgos potenciales de sesgos de los estudios. La medición de sesgos se ilustra en las figuras 2 y 3.
En los siete ensayos clínicos se demostró disminución de la mortalidad hospitalaria con el uso de esteroides. Sin embargo, algunos definieron ''mortalidad hospitalaria'' como la ocurrida a los 28 días (OR: 0,56 [0,38-0,81]), resultado que fue similar con el modelo de efectos fijos (figura 4) y el de efectos aleatorios (figura 5).
Hay una ganancia promedio de 3,5 días libres de ventilación mecánica (IC95%: 1,90-5,11) (figura 6). También hay disminución en la incidencia de infecciones nosocomiales en general (figura 7) y en la de neumonía adquirida en el hospital (neumonía asociada a la atención salud) (figura 8). No hay diferencia en la incidencia de neuromiopatía con el uso de esteroides (figura 9) y sí hay una tendencia a aumentar el sangrado del tracto digestivo (figura 10). Cada uno de los desenlaces se grafica con el modelo de efectos fijos. En la tabla 1 se presentan los resultados globales del metaanálisis: el número de estudios incluidos por desenlace, el número de participantes, las medidas de efecto y los resultados de la prueba de heterogeneidad.
Análisis de subgrupos
Excluyendo el estudio de Yates con mayor riesgo de sesgos no hubo diferencia importante en los resultados (figura 11).
Con los datos publicados no es posible hacer un análisis de sensibilidad por subgrupos de pacientes con SDRA primario o secundario, porque no hubo reportes diferenciales.
]]> Parece que existe riesgo de sesgo de publicación por la asimetría en el gráfico de embudo (Funnel Plot), pero hay pocos estudios y el estadístico de análisis no es confiable (figura 12).
DISCUSIÓN
Este metaanálisis muestra que la terapia con esteroides en dosis bajas en pacientes con SDRA disminuye la mortalidad hospitalaria (OR: 0,56 [0,38- 0,81]), hay una ganancia promedio de 3,5 días libres de ventilación mecánica, disminuyen las infecciones nosocomiales en general y la neumonía adquirida en el hospital (neumonía asociada a la atención en salud). No encontramos diferencias importantes en los efectos adversos.
Los resultados de este metaanálisis son congruentes con los de cinco revisiones sistemáticas previas en las que también se demuestra la disminución global de la mortalidad y de los días de ventilación mecánica (28- 32). Al igual que en estas revisiones, no hay diferencias significativas en la incidencia de eventos adversos. Un metaanálisis no encontró mejoría importante en la mortalidad por el SDRA con los esteroides pero en él se incluyeron estudios con dosis altas y bajas de estos fármacos (29) y se ha encontrado que las dosis altas se asocian con aumento de la mortalidad (33).
También hay que considerar que en los estudios recientes de uso de esteroides en pacientes con neumonía grave y durante la reciente epidemia de influenza H1N1, muchos de cuyos pacientes cumplían con los criterios de SDRA primario (23,25,34-36), el uso de esteroides se asoció con aumento de la mortalidad (37,38).
Una debilidad importante de este estudio fue no poder hacer un análisis de sensibilidad en pacientes con SDRA primario y secundario, pero los estudios originales no discriminaron este tipo de información.
CONCLUSIONES
]]> El uso de esteroides en dosis bajas en pacientes con SDRA se asocia con una disminución de la mortalidad y de los días de ventilación mecánica, pero hay heterogeneidad importante en los trabajos, que puede explicarse por la causa que originó el SDRA, el momento de inicio de los esteroides y la duración de la terapia. En el futuro se deberán hacer ensayos clínicos que comparen el SDRA primario con el secundario.
DECLARACIÓN DE CONFLICTO DE INTERÉS
Los autores declaran no tener conflicto de interés económico sobre el tema ni ningún tipo de patrocinio.
REFERENCIAS BIBLIOGRÁFICAS
1. Ashbaugh DG, Bigelow DB, Petty TL, Levine BE. Acute respiratory distress in adults. Lancet. 1967 Aug 12;2(7511):319–23. [ Links ]
2. Katzenstein AL, Bloor CM, Leibow AA. Diffuse alveolar damage--the role of oxygen, shock, and related factors. A review. Am J Pathol. 1976 Oct;85(1):209–28. [ Links ]
]]>3. Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med. 2000 May 4;342(18):1334–49. [ Links ]
4. Bernard GR, Artigas A, Brigham KL, Carlet J, Falke K, Hudson L, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med. 1994 Mar;149(3 Pt 1):818–24. [ Links ]
5. Raoof S, Goulet K, Esan A, Hess DR, Sessler CN. Severe hypoxemic respiratory failure: part 2--nonventilatory strategies. Chest. 2010 Jun;137(6):1437–48. [ Links ]
6. European Society of Intensive Care Medicie [Internet]. [Citado 2012, mayo] p.2, Disponible en: http://www.esicm.org/flash-conferences/lisbon-2012/3412 [ Links ]
7. Wexler HR, Lok P. A simple formula for adjusting arterial carbon dioxide tension. Can Anaesth Soc J. 1981 Jul;28(4):370–2. [ Links ]
8. Martin C, Papazian L, Payan MJ, Saux P, Gouin F. Pulmonary fibrosis correlates with outcome in adult respiratory distress syndrome. A study in mechanically ventilated patients. Chest. 1995 Jan;107(1):196–200. [ Links ]
9. Meduri GU, Muthiah MP, Carratu P, Eltorky M, Chrousos GP. Nuclear factor-kappaB- and glucocorticoid receptor alpha- mediated mechanisms in the regulation of systemic and pulmonary inflammation during sepsis and acute respiratory distress syndrome. Evidence for inflammation-induced target tissue resistance to glu. Neuroimmunomodulation. 2005 Jan;12(6):321–38. [ Links ]
10. Meduri GU, Tolley EA, Chrousos GP, Stentz F. Prolonged methylprednisolone treatment suppresses systemic inflammation in patients with unresolving acute respiratory distress syndrome: evidence for inadequate endogenous glucocorticoid secretion and inflammation-induced immune cell resistance to glucoc. Am J Respir Crit Care Med. 2002 Apr 1;165(7):983–91. [ Links ]
11. Kellum JA, Kong L, Fink MP, Weissfeld LA, Yealy DM, Pinsky MR, et al. Understanding the inflammatory cytokine response in pneumonia and sepsis: results of the Genetic and Inflammatory Markers of Sepsis (GenIMS) Study. Arch Intern Med. 2007;167(15):1655–63. [ Links ]
12. Barraud D, Gibot S. Triggering receptor expressed on myeloid cell 1. Crit Care Clin. 2011 Apr;27(2):265–79. [ Links ]
13. Meduri GU, Golden E, Freire AX, Taylor E, Zaman M, Carson SJ, et al. Methylprednisolone infusion in early severe ARDS: results of a randomized controlled trial. Chest. 2007 Apr;131(4):954–63. [ Links ]
14. Meduri GU, Headley AS, Golden E, Carson SJ, Umberger RA, Kelso T, et al. Effect of prolonged methylprednisolone therapy in unresolving acute respiratory distress syndrome: a randomized controlled trial. JAMA. 1998 Jul 8;280(2):159–65. [ Links ]
15. Steinberg KP, Hudson LD, Goodman RB, Hough CL, Lanken PN, Hyzy R, et al. Efficacy and safety of corticosteroids for persistent acute respiratory distress syndrome. N Engl J Med . 2006 Apr 20;354(16):1671–84. [ Links ]
16. Marik PE, Meduri GU, Rocco PRM, Annane D. Glucocorticoid treatment in acute lung injury and acute respiratory distress syndrome. Crit Care Clin. 2011 Jul;27(3):589–607. [ Links ]
17. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JPA, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med. 2009 Aug 18;151(4):W65–94. [ Links ]
18. Higgins JPT, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011 Jan;343:d5928. [ Links ]
19. Bennett DA, Emberson JR. Stratification for exploring heterogeneity in systematic reviews. Evid Based Med. 2009 Dec;14(6):162–4. [ Links ]
20. Guyatt GH, Oxman AD, Kunz R, Woodcock J, Brozek J, Helfand M, et al. GRADE guidelines: 7. Rating the quality of evidence--inconsistency. J Clin Epidemiol. 2011 Dec;64(12):1294–302. [ Links ]
21. Annane D, Sébille V, Bellissant E. Effect of low doses of corticosteroids in septic shock patients with or without early acute respiratory distress syndrome. Crit Care Med . 2006 Jan;34(1):22–30. [ Links ]
22. Confalonieri M, Urbino R, Potena A, Piattella M, Parigi P, Puccio G, et al. Hydrocortisone infusion for severe community-acquired pneumonia: a preliminary randomized study. Am J Respir Crit Care Med. 2005 Feb 1;171(3):242–8. [ Links ]
23. Fernández-Serrano S, Dorca J, Garcia-Vidal C, Fernández-Sabé N, Carratalà J, Fernández-Agüera A, et al. Effect of corticosteroids on the clinical course of community-acquired pneumonia: a randomized controlled trial. Critical care (London, England). 2011 Jan;15(2):R96. [ Links ]
24. Meduri GU, Headley AS, Golden E, Carson SJ, Umberger RA, Kelso T, et al. Effect of prolonged methylprednisolone therapy in unresolving acute respiratory distress syndrome: a randomized controlled trial. JAMA. 1998 Jul 8;280(2):159–65. [ Links ]
25. Steinberg KP, Hudson LD, Goodman RB, Hough CL, Lanken PN, Hyzy R, et al. Efficacy and safety of corticosteroids for persistent acute respiratory distress syndrome. N Engl J Med. 2006 Apr 20;354(16):1671–84. [ Links ]
26. Yates CR, Vysokanov A, Mukherjee A, Ludden TM, Tolley E, Meduri GU, et al. Time-variant increase in methylprednisolone clearance in patients with acute respiratory distress syndrome: a population pharmacokinetic study. J Clin Pharmacol. 2001 Apr;41(4):415–24. [ Links ]
27. Hough CL, Steinberg KP, Taylor Thompson B, Rubenfeld GD, Hudson LD. Intensive care unitacquired neuromyopathy and corticosteroids in survivors of persistent ARDS. Intensive Care Med . 2009 Jan;35(1):63–8. [ Links ]
28. Tang BMP, Craig JC, Eslick GD, Seppelt I, McLean AS. Use of corticosteroids in acute lung injury and acute respiratory distress syndrome: a systematic review and meta-analysis. Crit Care Med. 2009 May;37(5):1594–603. [ Links ]
29. Khilnani GC, Hadda V. Corticosteroids and ARDS: A review of treatment and prevention evidence. Lung India. 2011 Apr;28(2):114–9. [ Links ]
30. Lamontagne F, Briel M, Guyatt GH, Cook DJ, Bhatnagar N, Meade M. Corticosteroid therapy for acute lung injury, acute respiratory distress syndrome, and severe pneumonia: a meta-analysis of randomized controlled trials. J Crit Care. 2010 Sep;25(3):420–35. [ Links ]
31. Adhikari N, Burns KEA, Meade MO. Pharmacologic therapies for adults with acute lung injury and acute respiratory distress syndrome. Cochrane Database Syst Rev. 2004 Jan;(4):CD004477. [ Links ]
32. Peter JV, John P, Graham PL, Moran JL, George IA, Bersten A. Corticosteroids in the prevention and treatment of acute respiratory distress syndrome (ARDS) in adults: meta-analysis. BMJ. 2008 May 3;336(7651):1006–9. [ Links ]
33. Adhikari N, Burns K, Meade M. Tratamientos farmacológicos en adultos con lesión pulmonar aguda y síndrome de dificultad respiratoria aguda (revisión Cochrane traducida). La Biblioteca Cochrane Plus. Oxford Update Software Ltd. 2008. [ Links ]
34. Chon GR, Lim C-M, Koh Y, Hong S-B. Analysis of systemic corticosteroid usage and survival in patients requiring mechanical ventilation for severe community-acquired pneumonia. J Infect Chemother. 2011 Aug;17(4):449–55. [ Links ]
35. Meijvis SCA, Hardeman H, Remmelts HHF, Heijligenberg R, Rijkers GT, van Velzen-Blad H, et al. Dexamethasone and length of hospital stay in patients with community-acquired pneumonia: a randomised, double-blind, placebo-controlled trial. Lancet. 2011 Jun 11;377(9782):2023–30. [ Links ]
36. Snijders D, Daniels JMA, de Graaff CS, van der Werf TS, Boersma WG. Efficacy of corticosteroids in community-acquired pneumonia: a randomized double-blinded clinical trial. Am J Respir Crit Care Med. 2010 May 1;181(9):975–82. [ Links ]
37. Brun-Buisson C, Richard J-CM, Mercat A, Thiébaut ACM, Brochard L. Early corticosteroids in severe influenza A/H1N1 pneumonia and acute respiratory distress syndrome. Am J Respir Crit Care Med. 2011 May 1;183(9):1200–6. [ Links ]
38. Martin-Loeches I, Lisboa T, Rhodes A, Moreno RP, Silva E, Sprung C, et al. Use of early corticosteroid therapy on ICU admission in patients affected by severe pandemic (H1N1)v influenza A infection. Intensive Care Med . 2011 Feb;37(2):272–83. [ Links ]
Términos de búsqueda:
''respiratory distress syndrome, adult''[MeSH Terms] OR ''acute respiratory distress syndrome''[Text Word] OR ''Shock Lung''[TW] OR ''Lung, Shock''[TW] OR ''ARDS, Human''[TW] OR ''ARDSs, Human''[TW] OR ''Human ARDS''[TW] OR ''Respiratory Distress Syndrome, Acute''[TW] OR ''Acute Respiratory Distress Syndrome''[TW] OR ''Adult Respiratory Distress Syndrome''[TW]
AND
''steroids''[MeSH Terms] OR ''Steroids''[Text Word] OR ''methylprednisolone''[MeSH Terms] OR ''methylprednisolone''[Text Word]) OR ''glucocorticoids''[MeSH Terms] OR ''glucocorticoids''[All Fields] OR ''glucocorticoids''[Text Word] OR ''hydrocortisone''[MeSH Terms] OR hydrocortisone[Text Word] OR ''dexamethasone''[MeSH Terms] OR ''dexamethasone''[Text Word] OR ''prednisone''[MeSH Terms] OR prednisone[Text Word] OR ''prednisolone''[MeSH Terms] OR ''prednisolone''[Text Word] OR ''betamethasone''[MeSH Terms] OR ''betamethasone''[Text Word] OR ''cortisone''[MeSH Terms] OR ''cortisone''[Text Word]
AND ]]>
'randomized controlled trial'/exp OR 'randomized controlled trial'
'randomized controlled trials'/exp OR 'randomized controlled trials' 'randomization'/exp OR 'randomization' OR ''random AND allocat* OR ''randomly AND allocat* OR 'double blind procedure'/exp OR 'double blind procedure' OR 'doubleblind method'/exp OR 'double-blind method' OR 'single blind procedure'/exp OR 'single blind procedure' OR 'single-blind method'/exp OR 'single-blind method' single AND blind* OR ''double AND blind* OR triple blind* OR 'triple blind procedure'/ exp OR 'triple blind procedure' OR 'clinical trials'/exp OR 'clinical trials' OR 'clinical trial'/exp OR 'clinical trial'
AND
''hospital mortality''[MeSH Terms] OR hospital mortality[Text Word] OR
''mortality''[Subheading] OR ''mortality''[MeSH Terms] OR mortality[Text Word] OR ''respiration, artificial''[MeSH Terms] OR mechanical ventilation[Text Word] OR ''cross infection''[MeSH Terms] OR hospital infection[Text Word] OR
''pneumonia, ventilator-associated''[MeSH Terms] OR ventilator associated pneumonia[Text Word] OR gastrointestinal hemorrhage[All Fields] OR ''gastrointestinal hemorrhage''[MeSH Terms]