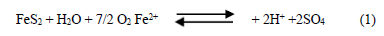
Techniques to correct and prevent acid mine drainage: A review
Técnicas correctoras y preventivas de las aguas ácidas de mina: Una revisión
Santiago Pozo-Antonio a, Iván Puente-Luna b, Susana Lagüela-López c & María Veiga-Ríos d
a Departamento de Ingeniería de los Recursos Naturales y Medio Ambiente, Universidad de Vigo, España. ipozo@uvigo.es
b Departamento de Ingeniería de los Recursos Naturales y Medio Ambiente, Universidad de Vigo, España.. ipuente@uvigo.es
c Departamento de Ingeniería de los Recursos Naturales y Medio Ambiente, Universidad de Vigo, España. susiminas@uvigo.es
d Departamento de Ingeniería de los Recursos Naturales y Medio Ambiente, Universidad de Vigo, España. mveigarios@gmail.com
Received: June 14th, de 2013. Received in revised form: March 10th, 2014. Accepted: March 31th, 2014
Abstract
Acid mine drainage (AMD) from mining wastes is one of the current environmental problems in the field of mining pollution that requires most action measures. This term describes the drainage generated by natural oxidation of sulfide minerals when they are exposed to the combined action of water and atmospheric oxygen. AMD is characterized by acidic effluents with a high content of sulfate and heavy metal ions in solution, which can contaminate both groundwater and surface water. Minerals responsible for AMD generation are iron sulfides (pyrite, FeS2, and to a lesser extent pyrrhotite, Fe1-XS), which are stable and insoluble while not in contact with water and atmospheric oxygen. However, as a result of mining activities, both sulfides are exposed to oxidizing ambient conditions. In order to prevent AMD formation, a great number of extensive research studies have been devoted to the mechanisms of oxidation and its prevention. In this work, we present an explanation and theoretical valuation of the pyrite oxidation process and a compendium on the measures most frequently employed for its prevention and correction.
Keywords: Acid Mine Drainage, mining, oxidation, pyrite, prevention, pollution.
Resumen
En la actualidad uno de los problemas medioambientales con mayor necesidad de actuación es la contaminación por la formación de drenajes ácidos de mina (AMD: "Acid Mine Drainage") procedentes de estériles de mina. Este es el término utilizado para describir el drenaje generado por la oxidación natural de sulfuros minerales que son expuestos a la acción combinada de agua y oxígeno atmosférico. Los minerales responsables de la generación de AMD son los sulfuros de hierro (pirita, FeS2 y en menor medida la pirrotita, Fe1-XS), los cuales son estables e insolubles mientras no se encuentren en contacto con agua y oxígeno atmosférico. Sin embargo, como consecuencia de la actividad minera, estos dos sulfuros son expuestos a condiciones ambientales oxidantes. La necesidad de prevenir la formación de AMD ha desarrollado numerosas investigaciones sobre los mecanismos de oxidación y su prevención. En el presente trabajo además de realizar una explicación y valoración teórica del proceso de oxidación de la pirita también se realiza un compendio de las medidas preventivas y correctoras más empleadas.
Palabras clave: Aguas Ácidas de Mina; minería; oxidación; pirita; prevención; contaminación.
]]> 1. Introduction
Acid mine drainage (AMD) formation has been widely recognized as one of the major environmental problems caused by mining worldwide, as evidenced by numerous studies [1-6].
Minerals responsible for the generation of AMD are iron sulfides (pyrite, FeS2, and pyrrhotite, Fe1-XS), which are stable and insoluble while not in contact with water and atmospheric oxygen [7]. Effective AMD management and correction of its impacts can be achieved only if the processes that influence the release and transport of trace metals are known [8].
Contamination occurs when these acidic waters reach surface or ground water systems used for human or industrial consumption [9] and as a result, many river courses worldwide have metal contamination from sulfide oxidation processes taking place in surface mining.
The need to prevent AMD formation has led to the development of numerous studies on the oxidation mechanisms and their prevention [9]. Notwithstanding the wide variety of existing studies, sulfides oxidation is very complex and its effects vary greatly between different places and conditions, so a proper AMD management requires the understanding of the various processes that control spatio-temporal variations in the sulfide oxidation.
Most oxidation processes that occur in natural environments produce a simultaneous modification of the acid-base properties of the system, generally increasing acidity [6].
As a first approximation, steps that give rise to degradation of water quality by introducing dissolved heavy metals are [1-10]:
The oxidation of metal sulfides is a complex process, that includes various types of reactions (oxidation-reduction, hydrolysis, ionic complex formation, solution, precipitation, etc.) giving rise to some oxidized iron forms, sulfate anions and a strong acidity.
The acid production is greatly favored by the pyrite grain fineness and influenced by temperature, porosity that allows oxygen penetration, moisture, hydrogeological characteristics of the site, etc.
There are two mechanisms of oxidation [11-13]:
1st) Direct reaction of oxygen with the pyrite to produce sulfate ions and acidity, Eq. (1).
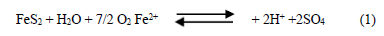
This is an inorganic mechanism, where oxygen acts as the oxidizing agent. This reaction is clearly limited by the oxygen availability, which is usually low due to the difficulties to diffuse presented by this element, and its kinetics is dependent on the speed at which molecular oxygen reduces [14]. Thus, no reaction takes place when the O2 concentration in the medium is low (Eh<300 mV). The diffusion rate depends on the physical relationships between the solid and fluid phases, mainly on the solid porosity and liquid water content. Bornstein et al. observed that the diffusion rate in soil, subsoil and sand was a maximum for water content between 10 and 20%, relative to dry weight, and percentages of air filled pores between 30 and to 70% [15]. Moreover, the H+, Fe2+ and SO42- concentrations also affect the speed of the reaction, which decreases as these concentrations increase. Furthermore, the temperature has a great influence on the oxidation rate as its value doubles every 10°C increase.
2nd) Oxygen replacement by the ferric ion, Eq. (2):
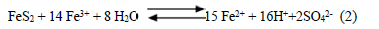
For this reaction to occur, the prior oxidation of the ferrous ion in situ, Eq. (3), is necessary:
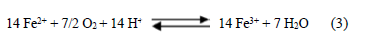
The oxidation of ferrous ion to ferric is an essential step in triggering the formation of AMD, which occurs at a low speed in normal conditions. Some bacteria act as catalysts, accelerating this reaction from 4 to 50 times, consequently increasing water acidity [10]. Thus, this is a pH- and Eh- dependent mechanism that is performed by acidophilic chemolithotrophic bacteria populations of the genus Thiobacillus, although other groups such as Metallogeniun and Sulfolobus can also contribute [17]. Bacteria population of the genus Metallgenium (an acid tolerant filamentous bacteria) were found at pH values between 3.5 and 4.5, and these bacteria carry the pH to values below 4. That is when the bacteria of the genus Thiobacillus ferrooxidans perform their catalytic works. Sphaerotilus natans and Gallionella ferruginea bacteria species were isolated at pH between 6 and 9 [18]. Although in this case the bacterial oxidation is unclear, the inorganic oxidation contribution is high. Even though the oxidation of the ferrous ion is a thermodynamically favorable process, the kinetics is very slow at pH values of about 4 or greater. However, the Thiobacillus ferrooxidans oxidizing bacteria can significantly increase the rate of oxidation by providing a mechanism involving a different transition state and lower activation energy to achieve the oxidation.
Due to the complexity of the process, the predominant reaction per pH is not clearly known, which are the reactants and products and how they evolve, therein lies the difficulty to perform kinetic studies and the variability in the data found [19].
Oxidation of a pyrite fragment depends both on the kinetics of the chemical reaction and on the diffusion of the oxidant (either Fe3+ or O2) and the products.
As a result of oxidation, a number of precipitates with different crystallinity are obtained; these are combinations of iron with oxygen or sulfate. Sulfates can evolve by dehydration and oxidation towards ferrihydrite or goethite, which are the most stable compounds in most natural environments. Jarosite is only stable in strongly acidic media (pH less than 4) and very high reduction potential (Eh), with high sulfate concentrations [20].
Throughout the entire pyrite oxidation process, the acid production is continuous, albeit its amount varies depending on the iron oxidation degree, the oxidation mechanism and the reaction products [21,22].
2. Preventive and corrective techniques of acid mine drainage
]]> Preventive techniques are those whose objective is to prevent the production of acid effluents from the mine. On the other hand, corrective techniques are designed to treat the acidic waters produced by mining, so that they stop being a threat to the environment [10].It is easy to understand that when planning the mine abandonment, priority should be given to suitable preventive techniques. The use of corrective techniques is only justified for the most serious and unforeseen cases of acid water pollution. Therefore, the mine waste treatment must be included in the mining project in order to minimize the exposure of these materials to inclement weather and to reduce the mine waste volume to be processed and stored [19].
2.1. Preventive techniques for acid mine drainage
In mining areas with high concentrations of pollutants, AMD treatment costs can have a significant impact on the economic performance, even to compromise the project viability, so that, AMD formation should be prevented.
Preventive techniques act on any of the three elements that are essential in the formation of acidic waters: oxygen, water and Thiobacillus ferrooxidans bacteria.
Next subsections show the currently employed techniques and some still in development.
2.1.1. Surface water diversion
This technique tries to reduce the water supply to those potentially acid-generating materials, by using safeguarding channels, pipes, waterproofed channels, slope changes around the material area, etc. The application of this technique should be preceded by accurate hydrological and hydrogeological studies, and it is precisely the lack of these which sometimes causes the unsuccessful application of this method [23,24].
The systematic application of this technique can be erroneous, because an effort (which can be costly in time and money) to divert the surface water to reduce large masses of water supply to areas where subsequent acid production is not extremely high, can lead to the reduction of the quantities of water entering areas that a priori are most dangerous as they can have an extremely high production of acid. Even so, this is a cheap technique, easy to apply and that works well in conjunction with other techniques.
2.1.2. Soil compaction ]]> Compaction of the mining wastes decreases their permeability. This compaction achieves on the one hand the reduction of water flow, which remains longer in the tailings, hindering the oxidation of the materials and therefore the rate of acid production, and on the other, the decrease of oxygen diffusion, which slows down pyrite oxidation and makes the tailings less habitable for ferro-oxidant bacteria.
A decrease of the porosity means a decrease of both the hydraulic conductivity and oxygen diffusion. Thus, the oxygen diffusion decreases by 3 to 4 orders of magnitude when the degree of saturation, i.e. water content, reaches 85% [25].
Although this is not itself a restoration method, it works effectively with any other put in practice.
2.1.3. Dry covers
The application of dry covers can be done with the aim at different objectives: (1) mining waste stabilization to prevent erosion by wind and water, (2) aesthetic appearance improvement and (3) both prevention and inhibition of pollutant release. The latter can be achieved with methods that prevent waste penetration by oxygen and water (Fig. 1). Without oxygen, chemical and microbial oxidation of sulfide minerals decreases. A minimized water flow causes a reduction in the drainage water production so that the metals are spread very slowly, resulting in a "dilution with time" [26].
There are a multitude of cover designs that cover the waste with soil or rock and use cementitious materials to form a crust. The effectiveness of the method increases when choosing a high and dry location for the dump.
The cover should be stable and provide long-term protection. This generally requires at least two layers of different materials, as it is rare that the required properties are found together in the same material. First, a bottom sealing layer with low-permeability is required to decrease the oxygen diffusion and to hold large amounts of water, acting as a barrier to oxygen. Suitable materials for this layer are clays, especially bentonite. However, these clay barriers are particularly prone to breaking during dry conditions letting in oxygen and water. Organic materials may be a good substitute to clay provided that the layer is
thick enough. Covers rich in organic materials have been used to act as a radon barrier [28]. Some artificial materials, as plastic membranes, have higher impenetrability than natural materials. However, care must be taken to prevent membranes from physical damage. There are also doubts about how long it will last before the chemical and microbial degradation appears [29]. Above the low permeability layer, a drainage layer can be placed to protect it from destruction by erosion, drying, freezing, influence of man, animals and plants and microbial destruction [26]. This drainage layer can also act to prevent the upward capillary migration of metals and salts. Additionally to this physical cover, there are chemical covers where materials that react with small-sized minerals are added to form an impermeable crust. Furthermore, additional measures can be taken to increase the reduction in the flow of oxygen as the addition of oxygen consuming microorganisms or the compaction of the mining wastes [30].
The typical sequence of operations starts with the flattening and leveling of the site, creating a backup channel to prevent the runoff effects on the dump. Later, the surface is compacted and the impermeable material extended and compacted if necessary. Above this, a layer of permeable material with at least 50 cm width is placed, and on top of it the soil is extended to sow. The plants selection must be done taking into account the potential threat that deep-rooted vegetation poses especially in the long term.
]]> Table 1 shows the materials that nowadays are used and their advantages and disadvantages [23].2.1.4. Covers with sludge
Sludge as a cover insulating material has been studied in Sweden since 1989 [31]. In this study, the mud used to cover the residue from a treatment plant in a nearby factory, was stabilized by the addition of fly ash produced by the combustion of coal and wood from factories. As fly ash contains calcium oxide, it can therefore increase the pH. Furthermore, a protective layer composed of tree bark waste overlaps on the layers of sludge.
Other studies have highlighted the importance of sludge use on roof tops or directly on the tailings to improve the establishment of vegetation. Studies have been conducted in different lithology dumps and even in areas devastated by toxic gases [32]. The use of sludge is often combined with lime supplements to increase the pH [33-34]. In most cases, productivity and fertility of the soil disturbed by mining have been substantially improved by sludge applications and the yields are generally higher than when using inorganic fertilizers. The use of sewage sludge to recover a coal mine spoil in Colorado resulted in significant increases in biomass of the soil surface layer [35]. This treatment gave better results than any other treatment with inorganic fertilizers (nitrogen, phosphorus). Recovery of places using chemical fertilizers and lime can produce the initial establishment of vegetation, but later the vegetation cover deteriorates due to poor physical conditions [4].
Covering with sludge improves some of the soil properties which help the establishment of life on the ground. The most important factor is the content of organic matter that improves the physical conditions of landfills, as it is an energy source for soil microorganisms [36]. The sludge contains nutrients and neutralizing components that increase the landfill pH and improve its fertility. Sometimes, a considerable extra input of nitrogen and phosphorus is required, but rarely potassium, while the need for additional calcium, magnesium and sulfur changes with the sludge composition.
In brief, mine waste covers can decrease oxygen and water penetration, and thus reduce the oxidation of sulfide minerals and/or the transport of hazardous compounds. Inorganic covers can be combined with sludges to improve sealing properties and soil physical conditions and to promote vegetation cover, which is essential to prevent erosion and to improve the aesthetic appearance of the area [31].
2.1.5. Sealing with clay
Often, mining transfer acidity is produced through groundwater discharge slopes. Then it is convenient to coat them with clay as the mine hole is filled, thereby reducing the water input through the mining wastes.
This is a very common phenomenon in abandoned mine pits where an inland lake can be formed, if this circumstance were not taken into account. This lake becomes an acidic water reservoir, therefore, it is also desirable to seal the abandoned mine workings.
]]> 2.1.6. Handling tailingsSometimes there are some materials with a particularly high potential to generate acidity within the tailings, so if they can be stored in a waterproof trap, the amount of drained acidic water will be considerably reduced. This is precisely the basis of this technique that, while expensive mainly due to the large number of analyses to be performed prior to the preparation of the waste, usually gives very good results [23].
To properly isolate the materials with higher acid generation rates, they should be located well below the surface but always above the water table in the area. They must also have a sufficiently compacted waterproof coverage (usually clay or plastic) that must be umbrella shaped to prevent water from coming in contact with the mining wastes.
2.1.7. Application of chemicals
Addition of chemicals that interact with the tailings reduces the release of acid, iron and other heavy metals into the reservoir since their solubility decreases as the pH increases (Fig. 2). There are some accessible and inexpensive alkaline chemicals that can be added to the mining wastes to neutralize the acid pH in the drainage. However, this method does not always prevent the re-acidification after a few years [26].
Placing a lime layer near the surface allows achieving an alkaline environment which inhibits bacteria proliferation and that neutralizes partly the generated acid. Because of this and its low cost, it has become a common practice in almost all restorations of tailings potentially generating acidic waters.
Several studies and case studies have concluded that this is an effective method to prevent acidic water in non-established systems, i.e., those that have not yet generated acidic water, while in established systems the improvement is temporary and insignificant, so another restitution method must be found. This phenomenon is due to the low neutralizing power of the lime as it is interrupted, in the case of very acidic water, once a layer of iron hydroxide that inhibits neutralization is deposited on its surface [1]. Frequently, requirements are both very large amounts of lime that can cause an imbalance in Ca/Mg ratio, and phosphate availability, with the consequent impairment of plant growth in the area. Liming is often used in combination with other methods, like another covering method.
Due to the key role played by iron in leaching, some chemical treatments seek to interfere with the chemistry of
]]> iron in the dump. The removal of iron from solution by precipitation is achieved by applying neutral ores or siliceous materials such as fly ash, blast furnace slag and Portland cement or by the complexation of iron either in active or reactive form, which inhibits oxidation. (Fe3(PO4)2 and FePO4) which diminish the amount of ferric iron that can react with pyrite. Subsequent laboratoryFurthermore, phosphate and phosphate clay inhibit pyrite oxidation by creating a cover around sulfuric mineral grains. The actuation mechanism goes through the formation of relatively insoluble iron phosphates studies showed that the addition of crushed apatite (Ca5(OH, F)(PO4)3) reduced the production of acidic water by 96% at doses below 3 ppt [44]. A field experiment subsequently performed by the same authors indicated that the addition of a 3 ppt amount of phosphate slowed the acid production from materials such as coal and slate for around a year from when they were added, but this inhibitory effect seemed to decrease in the second year [38].
Meanwhile, organic material is a suitable pH buffer that can eliminate iron chelation. A specific technique is called alkaline injection. In any mine dump, AMD flows through specific areas. If they can be neutralized by injecting some bases such as soda, lime or sodium carbonate where the waters circulate, then more restoration work on the surface of the waste can be avoided, while reducing costs [1]. Among others, the injection advantages would be the pH increase, which leads some metals to precipitate over the pyrite inside the mine tailings, stopping the pyrite oxidation, and the alkalization of the medium, which inhibits oxidizing bacteria and thus contributes to the improvement of water quality. Moreover, as a temporary technique it can be very suitable for abandoned tailings.
2.2 Corrective techniques for acid mine drainage
These are techniques that can be used to purify the acidic waters produced by a mine. They are conceptually opposed to the preventive techniques whose main objective is to avoid the formation of those acid waters. Corrective techniques are applied without distinction to underground or surface mines.
Many developed countries have recently enacted the adoption of some type of environmental legislation, which has led mining to install treatment plants for their effluents. This creates serious problems for the operating companies, but they often become critical when planning mine abandonment. In countries with the most restrictive laws, mining operators face the dilemma of implementing effective preventive measures or being forced to pay the effluent's treatment for years, after the mine is abandoned.
The most prevalent method for treating AMD contamination is the addition of alkaline reagents such as lime, limestone, sodium carbonate or sodium hydroxide. This treatment aims at neutralizing acidic water and the precipitation of heavy metals. However, maintenance requirements are high and large sludge amounts are produced that are mainly composed of calcium sulfate and some metal hydroxides. In an oxidizing environment, limestone is coated with reaction products until it becomes ineffective. This problem is solved by using passive anoxic limestone drains (ALDs) [39], which consist of shallow sealed trenches excavated in the spoil and filled with limestone.
The most used techniques will be described in the next sections.
2.2.1. Chemical neutralization plants
In these plants, the water recovery takes place in three phases: neutralization, oxidation and precipitation, as follows [1]:
2.2.2. "In-line system" plants
They are based on a simple and low maintenance method that simultaneously neutralizes and aerates the water, using the energy of water impulsed by two parallel injection pumps and a static mixer [40].
Injection pumps suck in air while collecting the mine acid waters, causing water aeration. After leaving the pump, the water goes to a static mixer consisting of a series of propylene resin tubes where liquid is kept in a turbulent regime by using a propeller that promote mixing and reactivity.
In-line system plants have been successfully tested to treat effluents with an input content of 200 mgl-1 Fe+2, which were reduced to an output concentration of around 3 mgl-1. At an industry level, in-line system plants are cheaper than conventional ones, and as they have no moving parts, their maintenance is simpler and more economical [41].
2.2.3. Treatment plants by ion exchange
This technology is based on the property of certain substances to make reversible ion exchanges within saturated solutions. These substances may be different in nature, synthetic resins with high molecular weight being the most commonly used.
This method is applicable to the acid mine drainage treatment with metal ions according to the reaction expressed in Eq. 8 [23]:
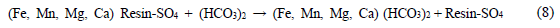
Subsequently, the resin is regenerated using alkaline solutions and then it is reused.
]]> Among the main advantages of this method are, according to Fernández-Rubio and Lorca, the sludge susceptibility to be dried, the low hardness and amount of the dissolved solids in the final effluents and the low lime consumption [23]. The greatest disadvantages are the handling difficulty and toxicity of the sludge obtained.Nowadays there are several systems to perform this technique such as: the continuous upward washing system in multi-chamber adsorption columns, the continuous descending washing system in mobile packed bed adsorption columns, upward washing in open tank adsorption columns, etc. [42].
Further studies have been conducted where separation techniques were designed for copper and zinc in the Iberian pyritic belt. These studies showed the possibility to apply resins with carboxyl groups for the selective separation of both metals [43].
2.2.4. Neutralization plants by reverse osmosis
This technique employs semipermeable membranes that allow the passage of water while retaining dissolved or suspended particles. The typical membranes, spongy and porous, have a 100 microns thickness, are made of modified cellulose acetate with one side covered with a dense layer [23].
One of the most important consequences of the experiments performed so far is the ease with which the membranes become obstructed, however, this is easy to fix by using sodium thiosulfate.
Although recoveries of up to 80% are obtained, this is an expensive method.
2.2.5. Natural neutralization with river waters. "Synergistic" solutions
Synergistic solutions are very attractive and low cost methods to easily neutralize acidic waters. The entire development that will be exposed below is based on a description of the particular case of the "Dober Mine" using the "Iron" river waters in Michigan (USA), summarized and generalized by Fernández-Rubio and Lorca [23].
The method uses the neutralizing capacity of a river, so that if the river flow is much larger than the strictly necessary to neutralize the mining acid drainage of the area, a portion of the flow can be diverted into a branch constituted by a set of ponds arranged in series to occur the precipitation of the metals and the subsequent neutralization of the water in a controlled manner.
]]> The process then consists in driving the acidic water coming from the mine to a circuit of staggered settling ponds, through which water circulates by gravity. In these ponds, if the flow is slow enough, the iron sulfate will precipitate and a slow increase of the water pH will gradually take place. The system is designed so that water is clean and properly neutral in the pond before its discharge to the river (located downstream of the inlet), so that the riverbed will remain untainted in its entire course.This is not a universally applicable technique, being this perhaps its most important limitation, as there are many drawbacks that prevent its application in many cases. The necessary conditions for the application of this method are:
In any case, the conclusions to be drawn are not for a particular case but they depend on the specific problem that occurs. Therefore, the natural neutralizing action of river waters can help and even resolve the problem of the acidic water if the control of some key parameters is achieved and the initial conditions are adequate.
3. Conclusions
Acknowledgments
The authors want to give thanks to the Spanish Ministries of Economy and Competitiveness, Education, Culture and Sport and University of Vigo for the financial support given; Human Resources programs (BES-2010-034106 and FPU AP2009-1144) and project BIA2009-08012.
References
[1] Kalin, M., Fyson, A. and Wheeler, W.N. The chemistry of conventional and alternative systems for the neutralization of acid mine drainage. The Science of the Total Environment, 366 (2-3), pp.395-408, 2006. [ Links ]
[2] Klapper, H. and Geller, W., Water quality management of mining lakes- a new field of applied hydrobiology. Acta Hydroch. Hydrob, 29, pp. 363-374, 2002. [ Links ]
[3] Doupé, R.G. and Lymbery, A.J., Environmental risks associated with beneficial end uses of mine lakes in southwestern Australia. Mine Water and the Environment, 24 (3), pp. 134-138, 2005. [ Links ]
[4] McCullough, C.D. and Lund, M.A., Opportunities for sustainable mining pit lakes in Australia. Mine Water Environ, 25, pp. 220-226, 2006. [ Links ]
[5] McDonald, D.M., Webb, J.A. and Taylor, J., Chemical stability of acid rock drainage treatment sludge and implications for sludge management. Environ. Sci. Technol. 40 (6), pp. 1984-1990, 2006. [ Links ]
[6] Saarinen, T., Mohämmadighävam, S., Marttila, H. and Klove, B., Impact of peatland forestry on runnof water quality in areas with sulphide- bering sediments: How to prevent acid surges. Forest Ecology and Management, 293, pp. 17-28, 2013. [ Links ]
[7] Cruz, R. and Monroy, M., Evaluation of the reactivity of iron sulfides and mining wastes. A methodology based on the application of cyclic voltammetry. Quim. Nova, 29 (3), pp.510-519, 2006. [ Links ]
[8] Monterroso, C. and Macías, F., Drainage waters affected by pyrite oxidation in a coal mine in Galicia (NW Spain): Composition and mineral stability. Science of the Total Environment, 216 (1), pp.121-132, 1998. [ Links ]
[9] Younger, P.L., Coulton, R.H. and Froggatt, E.C., The contribution of science to risk-based decision-making: lessons from the development of full-scale treatment measures for acid mine waters at Wheal Jane, UK. Science of the Total Environment, 338, pp.137-154, 2005. [ Links ]
[10] Johnson, D.B. and Hallberg, K.B., Acid mine drainage remediation options: a review. Science of the Total Environment, 338 (1-2), pp. 3-14, 2005. [ Links ]
[11] Nordstrom, D.K., Mine waters: Acidic to circumneutral. Elements, 7 (6), pp. 393-398, 2011. [ Links ]
[12] Stumm, W. and Morgan, J.J., Aquatic chemistry: An introduction emphasizing chemical equilibria in natural waters. NY, Wiley-Interscience; 1981. [ Links ]
[13] Urrutia, M., Graña, J., García-Rodeja, E. and Macías, F., Pyrite oxidation processes in surface systems: Acidifying potential and its interest in minesoils reclamation). Caderno do Laboratorio Xeolóxico de Laxe, 1987. [ Links ]
[14] Sáez-Navarrete, C., Rodríguez-Córdova, L., Baraza, X., Gelmi, C. and Herrera, L., Hydrogen kinetics limitation of an autotrophic sulphate reduction reactor, DYNA, 172, pp. 126-132, 2012. [ Links ]
[15] Bornstein, J., Hedstrom, W.E and Scott, F.R., Oxygen diffusion rate relationships under three soil conditions. Life Sciences and agriculture experiment station technical bulletin 98. 1980 [ Links ]
[16] Wiegand, U., Schreck, P., Schreiter, P., Lerche, I. and Glaesser, W., Restoration of open pit lignite mining in the former GDR: Lessons to be learnt from Zwenkau. Energy and Environment, 14 (4), pp.437-449, 2003. [ Links ]
]]>[17] Nancucheo, I. and Johnson, D.B., Selective removal of transition metals from acidic mine waters by novel consortia of acidophilic sulfidogenic bacteria. Microbial Biotechnology, 5 (1), pp.34-44, 2012. [ Links ]
[18] Umrania, V.V., Role of acidothermophilic autrotrophs in bioleaching of mineral sulphides ores. Indian Journal of Biotechnology, 2 (3), pp. 451-464, 2003. [ Links ]
[19] Kontopoulos, A., Acid Mine Drainage Control. In: Castro, S.H., Vergara, F., Sánchez, M.A. (Eds.), Effluent treatment in the mining industry. University of Concepción, 1998, pp. 57-118. [ Links ]
[20] Silva, L.F.O., Fdez-Ortiz de Vallejuelo, S., Martínez-Arkarazo, I., Castro, K., (...), Madariaga, J.M. Study of environmental pollution and mineralogical characterization of sediment rivers from Brazilian coal mining acid drainage. Science of the Total Environment, 447, pp.169-178, 2013. [ Links ]
[21] Millero, F.J. and Pierrot, D., A chemical equilibrium model for natural waters. Aquatic Geochemistry, 4 (1), pp.153-199, 1998. [ Links ]
]]>[22] Christov, C. and Moller, N., A chemical equilibrium model of solution behavior and solubility in the H-Na-K-Ca-OH-Cl-HSO4-SO4-H2O system to high concentration and temperature. Geochimica et Cosmochimica Acta, 68 (18), pp.3717-3739, 2004. [ Links ]
[23] Fernández-Rubio, R. and Lorca, D.F., Mine water drainage. Mine Water and the Environment, 12, pp.107-130, 1993. [ Links ]
[24] Grande, J.A., Impact of AMD processes on the public water supply: Hydrochemical variations and application of a classification model to a river in the Iberian Pyritic Belt, SW Spain. Hydrology Research, 42 (6), pp.472-478, 2011. [ Links ]
[25] Molson, J.W., Fala, O., Aubertin, M. and Bussière, B., Numerical simulations of pyrite oxidation and acid mine drainage in unsaturated waste rock piles. Journal of Contaminant Hydrology, 78 (4), pp.343-371, 2005. [ Links ]
[26] Swedish Environmental Protection Agency, SNV PM 4202, Solna, Sweden, 1993. [ Links ]
]]>[27] Ledin, M. and Pedersen K., The environmental impact of mine wastes - Roles of microorganisms and their significance in treatment of mine wastes. Earth-Science Reviews, 41, pp. 67-108, 1996. [ Links ]
[28] Landa, E.R., Microbial biogeochemistry of uranium mill tailings, Advances in Applied Microbiology, 57, pp.113-130, 2005. [ Links ]
[29] Palmer, C. and Young, P.J., Protecting water resources from the effects of landfill sites: Foxhall landfill site. Water and Environmental, 5 (6), pp.682-696, 1991. [ Links ]
[30] Pulford, I.D., A review of methods to control acid generation of pyritic coal mine waste, In Davies MCR, Land Reclamation: an end to dereliction? Elsevier, London, 1991. pp.269-278. [ Links ]
[31] Broman, P.G, Haglund, P. and Mattson, E., Use of sludge for sealing purposes in dry covers- development and field experiences, In Proc. 2nd Int. Conf. Abatement of Acidic Drainage, Ottawa, Canada, 1991. pp. 515-527. [ Links ]
]]>[32] Neculita, C.M., Zagury, G.J. and Bussière, B., Passive treatment of acid mine drainage in bioreactors using sulfate-reducing bacteria: Critical review and research needs. Journal of Environmental Quality, 36, pp. 1-16, 2007. [ Links ]
[33] Beauchemin, S., Fiset, J.F., Poirier, G. and Ablett, J.M., Arsenic in an alkaline AMD treatment sludge: Characterization and stability under prolonged anoxic conditions, Applied Geochemistry, 25 (10), pp.1487-1499, 2012. [ Links ]
[34] Kuyucak, N., Effective passive water treatment systems in extremely cold Canadian climatic conditions, SME Annual Meeting and Exhibit, 2010, pp. 50-54. [ Links ]
[35] Paschke, S.S., Banta, E.R., Dupree, J.A. and Capesius, J.P., Introduction, conceptual model, hydrogeologic framework, and predevelopment groundwater availability of the Denver Basin aquifer system, Colorado, US Geological Survey Professional Paper 2011, pp. 1-93. [ Links ]
[36] Peppas, A., Komnitsas, K. and Halikia, I., Use of organic covers for acid mine drainage control. Minerals Engineering, 13 (5), pp. 563-574, 2000. [ Links ]
]]>[37] Coulton, R., Bullen, C. and Hallett, C., The design and optimisation of active mine water treatment plants. Land Contamination and Reclamation, 11 (2), pp. 273-280, 2003. [ Links ]
[38] Kleinmann, R., At-source control of acid mine drainage. International Journal of Mine Water, 9 (1-4), pp.85-96, 1990. [ Links ]
[39] Brodie, G.A., Staged, aerobic constructed wetlands to treat acid drainage: Case history of Fabius Impoundment 1 and overview of the Tennessee Valley Authority's program. In Moshiri G.A., Constructed Wetlands for Water Quality Improvement, Boca Raton, FL: Lewis Publishers, 1993, pp.157-165. [ Links ]
[40] Bott, T.L., Jackson, J.K., Mctammany, M.E., Newbold, J.D., Rier, S.T., Sweeney, B.W. and Battle, J.M., Abandoned coal mine drainage and its remediation: Impacts on stream ecosystem structure and function. Ecological Applications, 22 (8), pp. 2144-2163, 2012. [ Links ]
[41] Ackman T. and Erickson, P., In-Line Aeration and Neutralization (ILS)-Summary of Eight Field Tests. In AIME/SME/TMS 115th Annual Meeting. New Orleans, LA., 1986. 34 P. [ Links ]
]]>[42] Himsley, A. and Bennett, JA., New continuous packed-bed ion exchange system applied to treatment of mine water. Ion Exch Technol 1984, pp.144-152. [ Links ]
[43] Valiente, M., Diez, S., Masana, A., Frías, C. and Muhammed, M., Separation of copper and zinc from waste acidic mine effluents of Río Tinto area. Mine Water and the Environment, 10 (1), pp.17-27, 1991. [ Links ]
[44] Egiebor, N.O., Oni, B., Acid rock drainage formation and treatment: A review, Asia-Pacific Journal of Chemical Engineering, 2 (1), pp. 47-62, 2007. [ Links ]
Santiago Pozo-Antonio received a Bs. Eng in Mining in 2009, an MSc degree in Environmental Technology in 2010, and a PhD degree in Environmental Technology in 2013, all of them from the University of Vigo, España. From 2010 to 2013, he worked in the Department of Natural Resources Engineering and Environment in the University of Vigo in several projects related with Mining and Conservation and Restoration of Built Heritage. From 2013-2014 he received an internship in the Getty Conservation Institute in Los Angeles (USA). His research interests include: cultural built heritage, mining techniques, landscape management and landscape assessment.
Iván Puente-Luna graduated in 2009 in Mining Engineering from the University of Vigo, and received his MSc. in Environmental Technology in 2010. Soon afterwards, he joined the Applied Geotechnologies research group, where he is currently enrolled in a PhD program about the applications of terrestrial and mobile LiDAR technology to infrastructure inspection and management. He was a visiting researcher in Carnegie Mellon University (USA, 2012) and Delft University of Technology (The Netherlands, 2013).
]]> Susana Lagüela-López. PhD in 3D thermography in 2014 from the University of Vigo, during which she had research stays at the University of California Berkeley and the ITC-CNR in Italy. She is author of more than 15 papers, and received the Prize for Young Researchers in New Technologies from the Council of Pontevedra, Spain (2011), and the Prize "Ermanno Grinzato" to Researchers under 30 from the International Conference in Thermography, AITA2013. Her research interests are the combination of thermography with geometry obtained with geomatic techniques for applications such as evaluation of energy efficiency in buildings, water management, land uses.María Veiga-Ríos received a Bs. Eng in Mining in 2009, an MSc degree in Environmental Technology in 2010, and a PhD degree in Environmental Technology in 2011, all of them from the University of Vigo. Her research interests include: rock science, mining techniques, landscape management and landscape assessment.
]]>