Cinética de la eliminación de fention en leche luego de una aplicación tópica en dos razas de ganado lechero
Cinética da excreção do Fenthion em leite aplicado de forma tópica em gado Holandês e HolandêsXBON
Nicolás F Ramírez Vásquez1,, MV, MS *; Jhon D Ruíz Buitrago1,4, MV, MS; Luis F Restrepo3 Est, Esp, Carlos Lopez1, Quim, MS.
1Grupo Interdisciplinario en Análisis de Residuos (GIAR) . Facultad de Ciencias Exactas y Naturales, Universidad de Antioquia . AA 1226 Medellín, Colombia.
2 Grupo de Investigación en Ciencias Veterinarias, Centauro, Escuela de Medicina Veterinaria, Facultad de Ciencias Agrarias, Universidad de Antioquia . A .A . 1226 Medellín, Colombia.
3 Docente Facultad de Ciencias Agrarias, Universidad de Antioquia . A .A . 1226 Medellín, Colombia.
4Grupo de Investigación en Ciencias de los Animales . Facultad de Medicina Veterinaria y Zootecnia, Universidad CES, Medellín, Colombia.
(Recibido: 21 julio 2008; aceptado: 26 febrero 2009.)
]]>Summary
The mammary excretion of fenthion was determined in cross-bred (Bon x Holstein) and pure Holstein cows following a topical (pour-on) application of a commercial formulation. Two groups of five cows each received a single application at the recommended dosage of 10 mg/kg BW on the withers. Milk samples were collected twice a day, from the morning and afternoon milkings during a 5-day period and analyzed for fenthion and total fat contents. A bi-compartmental analysis was used to describe the pharmacokinetic parameters. Overall fenthion milk concentrations increased progressively to reach maximum peaks (Cmax) of 0.4 ppm and 0.2 ppm at about 33 hr post-treatment in cross-bred and pure Holstein cows, respectively. However, since total residues in milk were strongly correlated with total fat content, for any particular day the concentrations attained in the morning milking were lower than those for the afternoon collection. The elimination half-lives (T1/2) for the two cattle breeds were 52.5 and 59.3 hr for cross-bred and Holstein respectively as indicated by milk concentrations. Fenthion concentrations declined to the maximum residue limit of 0.05 ppm set by the OMS FAO at 8.0, and 6.61 days post application, in cross-bred and pure bred Holstein cows respectively. These results suggest that withdrawal time for milk from lactating dairy cows treated with topical fenthion, could be of 8 days approximately.
Key words: dairy cows, fenthion residues, insecticides, withdrawal time .
Resumen
La excreción mamaria de fention se determinó en vacas productoras de leche cruzadas (Bon x Holstein) y Holstein puras, tras la aplicación tópica de una formulación comercial del compuesto. El producto se aplicó a dos grupos de cinco vacas por raza, a la dosis de 10 mg/Kg de peso vivo. Se colectaron muestras de leche dos veces diarias, en el ordeño de la mañana y de la tarde, por un período de cinco días y se midieron los niveles grasa y fention en leche. Se usó un análisis bicompartimental para describir los parámetros farmacocinéticos. Las concentraciones de fention en leche aumentaron progresivamente hasta alcanzar el pico máximo (Cmax) de 0.4 y 0.2 ppm a las 33 horas postratamiento en las vacas cruzadas y Holstein, respectivamente. Sin embargo, a pesar de que los residuos totales del fention se correlacionaron con el contenido de grasa, las concentraciones observadas en el ordeño de la mañana fueron menores que aquellas observadas en el ordeño de la tarde para las muestras de ese mismo día. Los tiempos medios de eliminación t1/2 fueron de 52,5 y 59.3 horas para el cruce y las vacas holstein puras, respectivamente. Las vacas cruzadas y las vacas Holstein puras alcanzaron el nivel máximo de fention permitido por la OMS-FAO (0.05 ppm) a los 8.0 y a los 6.61 días después de la aplicación, respectivamente. Estos resultados sugieren que el tiempo de retiro de la leche de vacas en lactancia tratadas con fention vía dérmica podría ser 8 días aproximadamente.
Palabras clave: fention, residuos en leche, tiempo de retiro, vacas lecheras.
Resumo
]]> A excreção mamaria de Fention foi determinada em vacas produtoras de leite dos grupos genéticos: Holandês e Holandês X BON, depois da aplicação dérmica de uma formulação comercial composta. O produto foi aplicado em cinco vacas de cada grupo genético, a uma doses de 10 mg/Kg do peso vivo. Coletaram-se amostras de leite nas ordenhas da amanha e da tarde, durante cinco dias para medir os teores de gordura e de fenthion em leite. Foi realizada uma análises bicompartimental para descrever os parâmetros farmacocinéticos. As concentrações de fenthion em leite aumentaram progressivamente até alcançar o pico máximo (Cmax) de 0.4 y 0.2 ppm as 33 horas postratamento nas vacas cruzadas y Holandês, respectivamente. Embora, os resíduos totais do Fention correlacionaram-se com os teores de gordura, em um dia em particular, as concentrações observadas na ordenha da amanha foram menores que as observadas na ordenha da tarde. Os tempos médios de eliminação t1/2 foram de 52,5 e 59.3 horas para as vacas cruzadas e Holandesas, respectivamente. As vacas cruzadas e Holandesas alcançaram o nível máximo de fenthion permitido pela OMS-FAO (0.05 ppm) aos 8.0 e aos 6.61 dias após da aplicação, respectivamente. Estes resultados sugerem que o tempo de retiro do leite em vacas tratadas com fenthion via dérmica, poderia ser de 8 dias aproximadamente.Palavras chave: fenthion, resíduos no leite, tempo de retiro, vacas leiteiras.
Introduction
Milk is one of the most important products of animal origin intended for human consumption. Current markets demand not only high quality milk but also an innocuous product that fulfills consumer expectations (4). Besides a proper nutrient profile, milk should be free of bacteria and forbidden antibiotics such as chloranfenicol and nitrofurans. It should also comply with the thresholds for pesticides residues officially approved for bovines (1).
Organophosphates are one of the main groups of insecticides used to control flies. Their mode of action is by inhibiting acetylcholinesterase, affecting the neuromuscular transmission of susceptible parasites (13, 17). One of such insecticide is fenthion (O,Odimethyl O-4-methylthio-m-tolyl phosphorothioate), which is an oily substance exhibiting a brownyellowish color (2). Fenthion (FEN) is a very lipophilic insecticide that once gained widespread use in cattle for the control of flies and lice. However, its application in lactating dairy cattle was never approved because milk was shown to be a substantial route of excretion in treated animals (11, 15). In spite of its widespread use in livestock and agriculture commodities over the past four decades, that was contraindicated in lactating cows probaby explains the lack of information on the kinetics of excretion in milk. At present, fenthion continues to be regularly used in Colombian cattle at any stage of production, including lactation, which predisposes milk and its by-products to contain residues. According to the Colombian Agricultural and Livestock Institute (“Instituto Colombiano Agropecuario”, ICA) 12.150 liters of fenthion were sold in Colombia during 1999. Later on, 13.187 and 13.396 liters were sold during 2000 and 2001, respectively (5). In the Northern and Eastern regions of Antioquia, it is currently the most widely used product to control flies in dairy cattle (12). Interestingly, the distribution and sales of all products containing fenthion has been prohibited in the USA since June of 2004, and the external application to animals is no longer registered in Europe. Because it has been extensively used since the early 70s, data for permissible residues in animal and agricultural commodities are available from several regulatory agencies in different countries. In this respect, an evaluation on fenthion by a Joint Meeting of Pesticide Residues from the World Health Organization (18) established a Maximum Residue Limit (MRLs) of 0.05 ppm for fenthion in milk. The aim of this investigation was to study the withdrawal times that would meet the OMSFAO recommendation for milk after a pour-on administration of fenthion at the recommended dosage of 10 mg/kg in two breeds of dairy cattle.
Materials and methods
Ethics committee
This work was conducted in accordance with the law for animal protection of the Republic of Colombia.
]]> AnimalsTwo groups of 10 pure bred Holsteins and 10 cross-bred Holstein cows (Bon x Holstein) at different stages of lactation were randomly selected and kept under field conditions throughout the experiment. Holstein cows were grazed on a lush pasture primarily composed of Kikuyo grass (Pennisetum calndestinum) at a University farm (San Pedro district) located at 2360 m of elevation and with a mean annual temperature of 16 °C. They were between first and seventh parity, averaging 6.6 years of age (range 2 -11 years) and 608 kg bodyweight (511 -700 kg). Mean milk yield was 29.2 L/day (range 19.6 -41.3 L). Cross-bred BON x Holstein cows were kept at a different University farm (Vegas de la Clara) located at 1080 m of elevation with a mean annual temperature of 25 °C. The pasture at this location consisted of Signal grass (Brachiaria decumbens), African grass (Cynodon plectostachyus), Bahia grass (Paspalum notatum), and Jaragua grass (Hyparrhenia rufa). Cows were between the second and sixth parity, averaged 6.7 years of age (range 4 -9), weighted 538 kg (range 450 -600 kg) and had an average milk yield of about 15.1 L/day (range 11.9 -19.4). Animals were supplemented with 1 kg of feed per each 5 kg of milk produced. Animals were parasite-free and their health monitored prior to and throughout the study. They were consistently negative to the California Mastitis Test.
Treatment and sampling
Ten animals from each breed were randomly assigned to receive fenthion (F, n=5) or no treatment (C, n=5). A commercial formulation containing 20% fenthion (Tiguvon®, spot-on, Bayer) was topically applied on the withers to the Fenthion group at a rate of 5 ml per 100 kg liveweight; this dosage corresponded to the manufacturer recommendation of 10 mg/kg. Cows were milked twice a day at 0, 10, 24, 34, 48, 58, 72, 82, 105 and 129 h posttreatment. Total milk yields were recorded, and samples of 40 mL were collected from every cow, and stored frozen at 4 °C until analysis.
Analytical method
Samples of milk were analyzed for fenthion by gas chromatography using a nitrogen and phosphorus detector according to Di Muccio et al (3). A description of the extraction and standardization procedure in our laboratory is further detailed elsewhere (15). Fenitrothion (O,Odimethyl O-4-nitro-m-tolyl phosphorothionate) was used as the internal standard. Recovery values from fortified samples of whole milk were between 92.8 and 116.0%. Intra assay coefficients were between 1.28 and 6.62%. The detection limit of the assay was 0.0075 ppm.
Fat assessment
Separate samples of 40 ml homogenized milk were also collected from 9 treated cows (5 Holsteins and 4 Bon x Holstein) for collection times 0, 10, 24, 34 48, 58 and 72 h. Samples were kept in plastic tubes and refrigerated for no more than 24 h until analysis. Fat content was assessed by mid-infrared absorption spectroscopy using a Milkoskan® 4000 equipment (Foss Electric, Hillorod, Denmark).
Pharmacokinetic and statistical analysis
The concentration versus time curves obtained for milk was fitted to a bi-compartmental method to calculate the pharmacokinetic parameters (PK Solution computer software, Summit Research Services, Montrose, CO). Based on the terminal elimination rate constant, a single component exponential model was then used to calculate the approximate withdrawal time according to the formula ∆t = Ln (Ct/Ci) / λz where:
]]> ∆t: time spent between Ct and CiLn: natural logarithm
Ct: 0.05 ppm (maximum residue limit set by the
World Health Organization (OMS-FAO, 1995)
Ci: initial concentration
λz: Kel (0.693/T1/2)
T1/2: half-life of excretion in milk
Statistical analysis
One-way and two-way ANOVA were conducted using the GLM procedure of SAS (SAS Inst. Inc., Cary, NC). The variables analyzed were fenthion level vs sample time, fat level vs sample time.
]]> Results
Quantification of fenthion residues in milk
None of the control cows in both farms had fenthion residues in milk at any sampling times. Before applying fenthion to any cows (time zero) fenthion was undetected in milk, indicating that residues were a direct result of the treatment. Milk from Bon x Holstein cows had different fenthion levels at the different sampling times, with higher levels during the afternoon milking (0.339 ppm average, at 10, 34, 58, 82 and 129 hours) compared to the morning milking (0.148 ppm average, at 24, 48, 72 and 105 hours) (p<0.05). Similarly, Holstein cows showed higher levels during the afternoon milking (0.176 ppm average, at 10, 34, 58, 82 and 129 hours) compared to the morning milking (0.115 ppm average, at 24, 48, 72 y 105 hours) (p< 0.05).
According to excretion kinetics a constant decrease in milk fenthion levels would be expected. Nevertheless, such a pattern was not always observed in this study, where some afternoon levels were numerically higher than the morning results for the same or even previous days, although no statistical difference was observed.
Milk fenthion and milk fat relationship
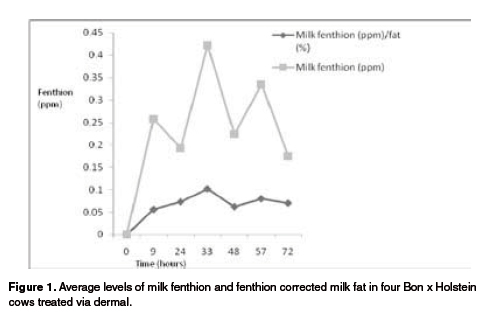
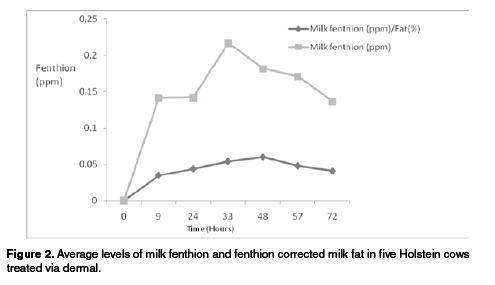
Fenthion and fat levels varied with a similar pattern along the milkings from 0 to 72 hours for the Bon x Holstein cows (see Figure 1). On the other hand, the Holstein cows did not show such a close pattern (see Figure 2). Milk fat was higher during the afternoon versus the morning milkings (p<0.05) for the Bon x Holstein cows (4.14 vs. 2.71%, respectively), and also for the Holstein cows (3.86 vs. 3.14%, respectively) (see Figure 2). This can be explained by the dilution effect created by a lower milk yield observed during the afternoon milking, which in turn was due to a shorter period of time between morning to afternoon versus afternoon to morning milking.
Bon x Holstein cows showed a positive correlation between fenthion and milk fat levels (p<0.05). This study confirmed the dilution effect of milk yield on fenthion concentration, which in turn depends on the fat level. Cows showed an inverse correlation between fenthion and milk yield: morning milkings had higher yields with lower fenthion concentrations, and the opposite was observed for the afternoon milkings. This correlation was not as clear for the Holstein cows. Although there was a numerical negative correlation between fenthion and yield for both breeds, it was not significant (p>0.05).
]]>
Pharmacokinetic analysis
Bon x Holstein cows. A non lineal relationship was observed in a semi-logarithm graph of fenthion levels versus time. Two slopes were obtained, one between 0 to 33 hours, and the other between 48 to 129 hours, representing the absorption or distribution phase and the excretion phase, respectively (see Figure 3). This was represented by a bi-compartmental model, in which blood was the central compartment, and the udder was the other compartment.
Holstein cows. Similar to the Bon x Holstein cows, a non lineal relationship was observed for fenthion levels versus time. Two slopes were obtained (0 to 33 hours, and 48 to 129 hours), representing absorption and excretion phases, respectively (see Figure 4).
]]>
The fenthion excretion phase in milk was calculated between 48 and 129 hours after dermal application for both groups of cows.
Pharmacokinetic parameters based on a regression analysis for the fenthion excretion phase are presented in Table 1.
Δt = Ln (Ct/Ci) / λz
In our case:
Δt: Ln (0.05/0.4076) ppm / (0.693/52.53) ppm/hour Δt: 160.68 hours
This time (Δt) plus the time taken for the compound to reach its maximum concentration is equivalent to the approximate withdrawal time:
]]> 160.68 hours + 33 hours = 193.68 hours, equivalent to 8.07 days.Table 1 also presents the parameters used to establish the approximate withdrawal time for Holstein cows:
Δt: Ln (0.05 ppm/0.2168 ppm) /0.693/59.35 hours
Δt: 125.7 hours At 125.7 hours, we have to add the time taken for the compound to reach the maximum concentration;
125.7 hours + 33 hours = 158.7 hours, equivalent to 6.61 days.
Discussion
A non lineal relationship was observed for fenthion levels versus time for both Holstein and Bon x Holstein cows. Two slopes can be observed in the graphs (see Figures 3 and 4), one between 0 and 33 hours and the other between 48 and 129 hours, representing the absorption or distribution phase and the excretion phase, respectively. According to Shargel and Yu (16), in order to apply pharmacokinetic analysis to multicompartment models (e.g. the open model with two compartments), it must be assumed that the flow rates of the substance, in or out of the individual compartments, are first order processes. According to this, a bi-compartmental model has a plasma concentration change in time that is best described by the addition of two processes which have a first order rate. The equation that describes a first order pharmacokinetic process is:
where:
A: Concentration of the substance. A0: Initial concentration of the substance.
]]> k: First order rate constant, expressed in hr-1.t: time.
: inverse of ln –kt.
According to our results, the fenthion absorptiondistribution phase (represented by the maximum excretion level in milk) was obtained 33 hours after fenthion application to both Holstein and Bon x Holstein cows. These results do not agree with a report in which a 3% fenthion solution was applied in the same manner as we did. Those researchers applied two different doses (9.4 mg/kg and 28 mg/kg), reporting maximum excretion levels in milk (0.81 and 2.82 ppm, respectively) at 9.6 hours in both cases (cited in reference 18). Even earlier peak levels were reported by O´Keeffe et al (14), who applied 125 ml of a 2% fenthion solution to the dorsal line of grazing and confined cows. They found maximum excretion levels of 0.11 ppm and 0.075 ppm at 6 and 12 hours, respectively. The different results could be explained by the greater volume applied when a lower fenthion solution is used. The lower the fenthion concentration used, the higher the volume applied, which implies a greater area of skin covered, which in turn favors a faster absorption, leading to maximum excretion levels in a shorter period of time.
Fenthion excretion phase was calculated between 48 and 129 hours after skin application for both groups of cows in this study. Fenthion excretion half-life was 52.53 hours and 59.35 hours for Bon x Holstein and Holstein cows, respectively. The similarity between both results reflects their reliability, considering that they were obtained from two different breeds located in two different ecological areas. None of the studies revised reported fenthion excretion half-life.
Milk fenthion levels fluctuated in a similar pattern as milk fat levels did throughout milking times. This occurred in both breeds, but it was more evident for the Bon x Holstein cows (p<0.05). These results could imply that milk fat is an important factor in fenthion excretion. This can be explained by the fact that fenthion is highly fat-soluble, having an octanolwater partition coefficient of (Log Kw) 4.8 (7). None of the studies reviewed refer to the effect of milk fat level on fenthion excretion (8, 9, 18).
Fenthion fluctuation with milk fat level could be explained by the dilution effect of milk yield, which was higher during the morning compared with the afternoon milking. This is evident when yield is plotted against fenthion level. This numerical effect was more evident in Bon x Holstein cows, although no significant difference was found (p>0.05). Our results are in agreement with O´Keeffe et al (14), who did not find an association between fenthion level and milk yield.
In many countries fenthion use is not recommended during lactation, but it appears to be very effective to control flies in stables. Literature findings on the maximum residue limits (MRLs) and withdrawal times are discussed below. In the present study, the level of fenthion in milk accepted by OMS-FAO (0.05 ppm) was reached in 8 and 6.61 days (7 days) for Bon x Holstein and Holstein cows, respectively. The study by Knowles and Arthur (11), cited by OMS (18) found that less than 0.001 ppm fenthion residues in milk was reached after 14 days, but did not mention when MRL reached 0.05 ppm. They applied 13 mg/kg fenthion on the skin of two 360 kg lactating cows. On the other hand, O´Keeffe et al . (1983) reported <0.05 ppm fenthion residues 22 hours after applying 125 mL of a 2% fenthion solution on the dorsal line of grazing cows, and <0.05 ppm fenthion residues after 30 hours on confined cows fed on silage (14). Two studies cited by OMS (18) applied a 3% fenthion solution (9.4 mg/kg), obtaining <0.05 ppm three days after the treatment. Another study also cited by OMS (18) applied a 2% fenthion solution (10 mg/kg), reporting that milk was fenthion-free 7 days later, but it did not explain when 0.05 ppm MRL was reached. One more study, cited by the World Health Organization (18), applied a 2% fenthion solution (125 ml/cow), reporting less than 0.005 ppm milk fenthion 48 hours later. It should be noticed that this concentration is much lower than the MRL established by OMS-FAO, indicating that the withdrawal time found should be much less than 48 hours in that study. All the cited reports contrast with the present study, in which a 20% fenthion solution was used, and the withdrawal time, based only on the original molecule, was 7 and 8 days for Holstein and Bon x Holstein cows, respectively.
A highly concentrated fenthion solution (98.25% purity) was used in a more recent study (6). Researchers uniformly applied this radio-labeled fenthion solution on the skin, over the dorsal line, of a Jersey cow. 14C-fenthion residues in milk at 6, 12 and 18 hours after the treatment were ≤ 0.05 ppm (6). These levels are much lower than the 0.266 ppm and 0.142 ppm observed in our study nine hours after treatment for the Bon x Holstein and Holstein cows, respectively.
As stated before, according to FAO-OMS (10), MRL is 0.05 ppm fenthion plus fenthion metabolites in whole milk. Our results show a minimum withdrawal time of 8 and 7 days for Bon x Holstein cows at 1080 m and for Holstein cows at 2360 m of elevation, respectively. Considering such long withdrawal times, it could be argued that fenthion utilization is not cost-effective in our dairy farms due to the amount of milk that should be discarded. To avoid such loss of income, farmers using fenthion during lactation would probably deliver all the milk to the markets, posing a serious threat to public health.
]]> It is out of the scope of the present study to explain the reasons for having a similar time to reach the MRL in both breeds under different ecological areas, and to understand why our results were higher compared to other studies. Those reasons could be related to the different nutritional and environmental conditions for this study, compared to other countries. According to this, it could be argued that regulations for toxic residues in milk should be based on research results obtained under similar conditions such as genetics, environment, nutrition, and even economic and cultural traits.In conclusion, the minimum withdrawal time for milk intended for human consumption after skin application of a 20% fenthion solution at 10 mg/kg in Holstein and Bon x Holstein cows under the conditions of the present study should be 8 days. This complies with the MRL (0.05 ppm) recommended by OMS-FAO (10).
Acknowledgements
Authors thanks to Comité para el Desarrollo de la Investigación (CODI) from Universidad de Antioquia at Medellín, Colombia, for financial support, and to Dr. Jorge H. Agudelo for critical comments and reviewing of the manuscript.
References
1. Aboix i Arzo, M. Martin-Jiménez T. Aspectos Terapéuticos y de salud pública de los residuos farmacológicos. In: Botana, L. Landoni, F. Martín-Jiménez, T (eds). Farmacología y terapéutica veterinaria. Madrid: McGraw- Hill-Interamericana; 2002. pp 681- 689. [ Links ]
2. Budavari S, O´Neil M, Smith A, Heckelman P, Kinneary J. The Merck Index. 20th ed. Merck Research laboratories. 1996. [ Links ]
3. Di Muccio A, Pelosi P, Camoni I, Attard Barbini D, Dommarco R, et al. Selective, solid-matrix dispersion extraction of organophosphate pesticide residues from milk. J Chromatography 1996; 754: 497-506. [ Links ]
4. Harding F. World milk production. Milk quality. 1st ed. Gaithersburg: Aspen Publisehrs; 1995. [ Links ]
5. Instituto Colombiano Agropecuario ICA. Comercialización de plaguicidas en 1999. 2001. [ Links ]
6. International Programme on Chemincal Safety. Chemical Safety Information from Intergovernmental Organizations (INCHEM). Fenthion. (1995b). [Último acceso 26 de mayo 2008.] URL: http://www.inchem.org/documents/jmpr/jmpmono/v95pr07.html. [ Links ]
7. International Programme on Chemincal Safety. Chemical Safety Information from Intergovernmental Organizations (INCHEM). Fenthion. JMPR, (2006). [Último acceso 26 de mayo 2008.] URL: http://www.inchem.org/documents/icsc/icsc/eics0655.htm. [ Links ]
8. International Programme on Chemincal Safety. Chemical Safety Information from Intergovernmental Organizations (INCHEM). Pesticides residues in food. (1980). [Último acceso 26 de mayo 2008.] URL:http://www.inchem.org/documents/jmpr/jmpmono/v080pr19.htm [ Links ]
9. International Programme on Chemincal Safety. Chemical Safety Information from Intergovernmental Organizations (INCHEM). Pesticide residues in food (1997). Toxicological and environmental evaluation 1994. [Último acceso 26 de mayo 2008.] URL: http://www.inchem.org/documents/jmpr/jmpmono/v097pr08.htm. [ Links ]
10. Joint Meeting of the FAO Working Party of Experts on Pesticide Residues and the WHO Expert Committee on Pesticide Residues, Wld Hlth Org. techn. Rep. Ser., No. 502; FAO Agricultural Studies, (1972), No. 88. [Último acceso 26 de mayo 2008.] URL:http://www.inchem.org/documents/jmpr/jmpmono/v071pr06.htm [ Links ]
11. Knowles CO, Arthur BW. Metabolism of and residues associated with dermal and intramuscular application of radiolabeled fenthion to dairy cows. J Econ Entomol 1966; 59:1346-1352. [ Links ]
12. Loaiza A, Jaramillo J, Tamayo F. Incidencia de factores sociales, económicos, culturales y técnicos en el uso de agroquímicos por pequeños productores del departamento de Antioquia. ICA - PRONATTA. 2000. [ Links ]
13. Muñoz M. Antiparasitarios Externos. In: Botana L, Landoni F, Martín-Jiménez T (eds). Farmacología y Terapéutica Veterinaria. Madrid: Mac-Graw Hill- Intermericana; 2002. p. 505-516. [ Links ]
14. O´Keeffe M, Eades K, Strickland J. Fenthion residues in milk and milk products following treatment of dairy cows for warble-fly. J Sci Food Agric 1983; 34:192-197. [ Links ]
15. Ramírez N, Betancur M, Ruíz J, López C, Restrepo L et al. Validación de un método analítico para la determinación de fention en leche bovina. Actual Biol 2006; 28:115-124. [ Links ]
16. Shargel L, Yu A. Applied biopharmaceutics and pharmacokinetics. 3a ed. Prenctice Hall International. 1992. [ Links ]
17. Taylor P. Anticholinesterase agents. In: Hardman J, Limbird L. Goodman & Gilman´s (eds). The Pharmacological basis of therapeutics. 10th ed. McGraw-Hill. 2001. p. 175 -191. [ Links ]
18. World Health Organization. Evaluations of Some Pesticide Residues in Food. Geneve. WHO Pesticide Residues Series, N° 1. WHO 1972. [Último acceso 26 de mayo 2008.] URL: http://www.inchem.org/documents/jmpr/jmpmono/v071pr06.htm [ Links ]
]]>