Transesterificación de triglicéridos en el sistema de reacción heterogénea con óxido de calcio como catalizador
Mónica Becerra Ortega, Aristóbulo Centeno Hurtado, Sonia Azucena Giraldo Duarte*
Centro de Investigaciones en Catálisis (CICAT). Escuela de Ingeniería Química. Universidad Industrial de Santander (UIS). Carrera 27 Calle 9. Bucaramanga. Colombia
Abstract
In this work, the behavior of the CaO as a potential catalyst for the transesterification of triglyceride towards biodiesel production was studied. The effect of the alcohol type, the ratio of alcohol/triacetin, the amount of catalyst, and the chain length of triglyceride on the catalytic behavior of CaO was analyzed. Total conversion was obtained at room temperature with a 6:1 molar ratio of methanol to triacetin over 1% of CaO, after 1 h. It was demonstrated that the whole reaction occurs in heterogeneous phase. During five reaction cycles the CaO maintained a high catalytic activity, showing its good stability. Additionally, it was established that the length of the triglyceride used influenced the transesterification reaction yield due to the steric hindrances and diffusional limitations in the fluid phase.
Keywords: Biodiesel, triacetin, basic catalysis, triolein.
Resumen
En este trabajo se estudió el comportamiento del CaO como potencial catalizador en la transesterificación de trigliceridos para la producción de biodiesel. Se analizó el efecto del tipo de alcohol, la relación molar alcohol/triacetina, la cantidad de catalizador y el tamaño de la cadena del triglicérido sobre su comportamiento catalítico. Los resultados mostraron que a temperatura ambiente, con una relación molar metanol/triacetina de 6 y 1% de CaO se logró conversión total de la triacetina en 1 h de reacción. Se demostró que la reacción ocurre en fase heterogénea. El CaO mantuvo una elevada actividad catalítica durante cinco ciclos de reacción, lo que demuestra su estabilidad en el sistema de reacción. Además, se pudo establecer que la longitud del triglicérido empleado influye en el rendimiento de la reacción de tranesterificación por causa de impedimentos estéricos y limitaciones difusionales en las fases fluidas.
]]> Palabras clave: Biodiesel, triacetina, catálisis básica, trioleina.Introduction
The biodiesel synthesis by transesterification at industrial level is developed mainly through basic homogeneous catalysis, a process that requires neutralization and purification stages that increase production costs and generate significant contaminant effluents. A variety of basic solids that includes zeolites, hydrotalcites, supported alkali metals and alkali earth oxides have been tested for transesterification at laboratory scale, in order to solve these problems and simplify the production process [1-4]. The CaO is prominent among these solids because of its high catalytic activity at mild reaction conditions, stability, and low cost. Zhu et al [5] obtained more than 93% conversion of Jatropha oil using CaO and methanol at 70°C. Reddy et al [6] produced biodiesel from soybean oil at room temperature using nanocrystalline CaO; however, this process required 24 h for complete conversion. Gryglewicz [2] reports low solubility of CaO in the reaction mixture the transesterification of rapeseed oil over methanol. Kouzu et al [7] investigated the transesterification of soybean oil with CaO and reported that the catalyst was not deactivated even in the fourth reaction cycle.
In this work, the effect of alcohol type, the molar ratio alcohol/triacetin, and the amount of CaO as catalyst on the conversion of triacetin were studied. The triacetin was selected as a model molecule because it only differs from common triglycerides in size, and its chemical and structural simplicity facilitates the identification and quantification of reaction products. Emphasis was placed on analyzing the stability of CaO by evaluating the presence of calcium in the reaction mixture by atomic absorption (AA), aiming to study the dissolution degree of CaO, and performing several reaction cycles with the same catalyst to analyze their lifetime. Furthermore, the influence of the chain length of triglyceride on the CaO catalytic activity in the triolein transesterification with methanol to methyl oleate was studied.
Experimental
Materials
Triacetin & (99.0%), dodecane (>99.0%) and triolein (65%) were purchased from Sigma-Aldrich. Butanol (99.8%) was obtained from Carlo Erba. Ethanol (>99.5%), methanol (99.9%), isopropanol (99.8%) and calcium oxide (>97.0%) were obtained from Merck. The CaO in small pieces (~3-20 mm) was pulverized and used as catalyst without further treatment. Standards of methyl esters that were used for CG calibration were obtained from Supelco.
Characterization of CaOThe textural properties of CaO were obtained from nitrogen adsorption-desorption isotherms (NOVA 1200 Quantachrome); the CaO was previously degassed at 100°C under vacuum for 12 h. Specific surface area was determined by the BET method and pore size by the BJH method. The basic strength (H_) and the basicity of CaO were determined by the Hammett titration method. 0.1 g of CaO was mixed with 2 mL of an indicator solution (0.1 mg of indicator in 1 mL of methanol). This suspension was titrated with 0.01 M benzoic acid in methanol. The following indicators were used: bromothymol blue (H_= 7.2), phenolphthalein (H_= 9.3), 2.4-dinitroaniline (H_= 15.0), 4-nitroaniline (H_= 18.4) and 4-chloroaniline (H_= 26.5).Although this method conventionally used non polar solvents [8], in this work methanol was used to measure the basic properties of the catalyst under reaction conditions.
Catalytic activity tests
]]> The CaO behavior for the transesterification of triacetin was evaluated in a batch reaction system composed of a three-neck flask with condensation, sampling, temperature control, and magnetic stirring systems. The reactor charge was composed of alcohol, triacetin, dodecane used as internal standard for chromatographic analysis and catalyst. The preliminary reaction conditions were: reaction volume = 60 mL, molar ratio alcohol/triacetin Ral/tr= 10, ambient temperature Ta= 28°C and 3% of CaO based on the weight of the reaction mixture. The sample analysis was performed in a HP 6890 gas chromatograph equipped with a capillary column HP-INNOWax of 30 m x 0.32 mm x 0.25 µm. The CaO activity was expressed through the conversion of triacetin. The diacetin, monoacetin, and glycerin percent selectivity (Si) based on the accepted reaction scheme [9] was calculated using the equation 1, where Ci is the massconcentration of each component. 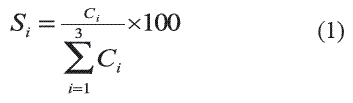
The CaO stability was evaluated by analyzing the presence of Ca in the reaction mixture by atomic absorption and performing several reaction cycles with the same catalyst. After using the CaO in the transesterification for 1 h, it was recovered by filtration, washed by Soxhlet extraction with ethanol during 2 h, dried at 60°C, and reused. In each cycle, an amount of reagents proportional to the weight of the recovered catalyst was used to maintain the initial reaction conditions.
Transesterification reactions of triolein were carried out to evaluate the influence of the chain length of triglyceride. Due to the low solubility of glycerol in esters, a phase separation occurs: a lower glycerol-rich layer and an upper methyl-ester rich layer. The upper phase, containing the methyl oleate, was analyzed by gas chromatography. The injector and detector temperatures were set to 255 and 260°C, respectively. After an isothermal period of 4 min at 90°C, the GC oven was heated at 20°C/min to 220°C and held for 4 min, and finally heated at 10°C/min to 230°C and held during 9 min. The content of methyl oleate was determined and expressed as weight percentage. The activity for the transesterification of triolein was expressed by the yield to methyl ester produced. The yields to monoglycerides and diglycerides were not determined, and it is assumed that the ester yield was close to the triglyceride conversion. This may be a good approximation for a conversion higher than 30-40% because the selectivity to mono, and diglycerides is important only at low triglyceride conversion. As the reaction proceeds, the selectivity to mono and diglycerides rapidly decreases, and therefore, the yield to these compounds is very low at high conversion, and much lower in comparison with the methyl ester yield. Hence, it is accepted that ester yield is a reasonable measure to evaluate the catalytic activity [10].
The percent yield (Y) to the methyl esters with respect to triglyceride charged amount is expressed by equation (2).

Where the factor 3 is stoichiometric, i.e. 1 mol of triglyceride is required to produce 3 mol of ester.
Results and discussion
]]> CaO characterizationThe CaO BET specific surface is 7 m2/g and pore volume 0.03 cm3/g. Regarding the distribution of basic CaO strength, indicators bromothymol blue and phenolphthalein rapidly showed the color of its basic form on the catalyst, but with the next indicator, 2,4-dinitroaniline (H_= 15.0), there was no color change to their conjugate base. Therefore, the strength of basic sites is within the range 7.2<H_<9.3, with 2.4 and 9.3<H_<15.0 with 0.2 mmol/g of catalyst basic site concentration for a total active site concentration of 2.6 mmol/g. Although the Hammett method can give qualitative information of the basic properties, it provides controversial measurement because diffusional problems of the indicator molecules within the catalyst pores may occur [11].
Catalytic activity tests
Effect of alcohol type on the conversion of triacetin
Figure 1 shows the effect of alcohol type on triacetin transesterification using four different alcohols: methanol, ethanol, butanol, and isopropanol. Marked differences in the reaction rate by varying the alcohol can be observed in figure 1. It is observed that when the alcohol has longer chain, the activity of CaO decreased. Marchetti and Errazu [12] reported similar results using enzymatic catalysis in the esterification of oleic acid. The transesterification by heterogeneous basic catalysis proceeds via formation of an alkoxide, which attacks the carbonyl group of the triglyceride and creates a tetrahedral intermediate or transition state [13]. Asakuma et al [14] proposed that in this transition state, a polygonal ring between the carbonyl carbon of the triglyceride and the alkoxy group is formed, and the larger the chain length of alcohol, the larger the ring size. On this basis, a larger alkyl chain of alcohol may increase the steric hindrances that affect their reactivity.
Complete conversion is achieved with methanol within 1 h of reaction. This alcohol is the best option at the established conditions, among the alcohols studied. According to Stavarache et al [15], the smaller size of methanol could facilitate the simultaneous attack of the three-triglyceride chains, while a longer chain alcohol must follow the pathway from triglyceride to diglyceride, and from diglyceride to monoglyceride in order to obtain the alkyl esters.
]]>
The final conversion achieved with ethanol and butanol is very low. This shows that the operating conditions employed are not the best for these alcohols. With 2-propanol, there was no appreciable conversion, and probably because it is a secondary alcohol, there is even greater steric hindrances that impede their access to the catalytic sites, which causes the reaction rate to be very low compared to the normal-chain alcohols [15]. On the other hand, it was found that the basic CaO strength is high enough to extract the proton from the alcohol and, thereby, generate nucleophilic attack, and its catalytic performance in the reaction with methanol is related to the wide distribution of basic sites on their surface (7.2<H_<15.0).
Effect of the catalyst amount on the triacetin conversion
Figure 2 shows triacetin conversion with methanol for concentrations of 1 and 3% of CaO in the reaction mixture, in both cases, total conversion is achieved at 1 h of reaction. Without catalyst addition, the transesterification is not carried out within this reaction time. Differences in the reaction rates were detected. The high stirring speed, the fine particle size (powder), the extremely low pore volume of CaO, and the relatively low percentage of catalyst into the reactive mixture used in our experiments lead us to think that the external and internal diffusions are not limiting steps. For that reason, we have considered that the reaction rate was governed by the surface reaction. X. Liu et al [16] have showed that under reaction conditions similar to the ones presented in this study, the intrinsic kinetic controls the reaction rate.
Effect of the alcohol/triacetin molar ratio (Ral/tr) on triacetin conversion
The stoichiometry ofthe transesterification requires 3 mol of alcohol per mol of triglyceride to produce 3 mol of alkyl ester and 1 mol of glycerine. An excess of alcohol is required to drive the reaction towards the formation of products, and theoretically an increase of the alcohol to triglyceride molar ratio can raise biodiesel production. However, the high Ral/tr contributes to increased solubility, making more difficult the separation of glycerin from ester [17]. Moreover, much energy is required to recover the unreacted alcohol. Figure 3 shows the influence of the molar ratio Ral/tr in the range 5 to 15 on the conversion. By increasing the molar ratio of 5 to 6, the reaction rate increases significantly. Nevertheless, the changes over the conversion are minimal with higher molar ratios, and complete conversion is achieved during 1 h in all cases. Therefore, it is possible to conclude that the best methanol/triacetin molar ratio is 6 under the operation conditions.
Catalyst stability
Figure 4 shows triacetin conversion in several reaction cycles with the same catalyst. CaO did not lose its activity in more than 5 h of reaction reaching conversions of the same order in every cycle. Furthermore, in a reaction carried out at Ta= 28°C over 3% of CaO, the atomic absorption measurements indicated the presence of 430 ppm of Ca. This shows that the amount of dissolved CaO is only 2.3%. Gryglewicz [2] also reported a low CaO solubility in the presence of methanol. However, in homogeneous catalysis, a low concentration of a very active catalyst is enough to carry out the reaction [18]. To verify whether the detected Ca in solution is involved in the reaction, at the end of the reaction the catalyst was removed by centrifugation and filtration; triacetin was added to the filtered solution. No conversion was found during 1 h of stirring at reaction conditions, confirming that catalysis was associated only with the solid phase of the material. This is an expected result because dissolved calcium species are not basic.
]]> Selectivity in the transesterification of triacetinFigures 5 and 6 show the selectivity as a function of both triacetin conversion and reaction time, respectively. The results are consistent with the reaction scheme [9], where the diglycerides are the first products formed, and thus the selectivity towards diacetin at low conversion is near 100%. This selectivity rapidly decreases while glycerin and methyl ester are produced. Monoacetin does not reach high selectivity values and has a tendency to disappear with the reaction time, while the glycerine tends to reach 100% selectivity. By varying the CaO amount and the molar ratio Ral/tr, selectivity does not change significantly.
Catalytic activity of calcium oxide in the transesterification of triolein
For reactions carried out under the best conditions found in the transesterification of triacetin with methanol and CaO (Ta= 28 ° C, Ral/tr= 6 and 1% of CaO), the yield to methyl oleate did not exceed 1% in 5 h unlike the behavior of triacetin reaction where complete conversion was reached within 1 h of reaction. There is a clear relationship between the reaction rate and the chain length of triglyceride used. Evidently, the length of the chain is affecting the reaction which in principle could be attributed to steric hindrances. Several authors have reported that basic solid catalysts activate the carbonyl groups in compounds containing these groups [19]. For this reason, the reaction conditions were modified to those that favor an adecuate conversion. The established conditions were: 60°C, a Ral/tr of 10 and 3% CaO. As it was observed, the long-chain triglycerides require drastic reaction conditions in order for the reaction to be carried out at shorter times.
Figure 7 shows methyl oleate yield vs time at 60°C, a Ral/tr of 10 and 3% of CaO. As can be observed, large triglycerides require more drastic reaction conditions in order for the transesterification to occur in a viable time. An increase in temperature favors the formation of methyl esters because the reaction rate increases, and when the higher the temperature the viscosity decreases and the solubility increases, which contributes to favor the homogenization process and the diffusion of triglyceride molecules towards the active sites. A reaction time of 5 h reached a methyl oleate yield of approximately 93%
Conclusions
Experimental results show that CaO, with a basic strength of 7.2 <H_<9.3 and 9.3 <H_<15.0 and without any additional treatment, is an active selective and stable catalyst for the transesterification of triglycerides or biodiesel production. Factors such as the catalyst amount and molar ratio alcohol/triglyceride lead to significant variations on the reaction rate of the triglyceride transesterification. Furthermore, it was demonstrated that the alcohol nature affects the reaction and that the larger the chain length of alcohol, the lower the final conversion of triacetin. Undoubtedly, the triglyceride size used for transesterification plays an important role in the catalytic activity. When working with large triglycerides, the steric hindrances and the diffusional limitations presented by the reaction of transesterification become much more evident.
]]> AcknowledgmentsThe authors express their sincere thanks to VIE- UIS for the financial support through the project 5423. Mónica Becerra thanks COLCIENCIAS for the financial support of MSc studies through the program: "Jóvenes Investigadores".
References
1. T. Ebiura, T. Echizen, A. Ishikawa, K. Murai, T. Baba. "Selective transesterification of triolein with methanol to methyl oleate and glycerol using alumina loaded with alkali metal salt as a solid-base catalyst". Appl. Catal. A. Vol. 283. 2005. pp. 111-116. [ Links ]
2. S. Gryglewicz. "Rapeseed oil methyl esters preparation using heterogeneous catalysts". Bioresource Technol. Vol. 70. 1999. pp. 249-253. [ Links ]
3. Y. Xi, R. J. Davis. "Influence of water on the activity and stability of activated Mg-Al hydrotalcites for the transesterification of tributyrin with methanol". J. Catal. Vol. 254. 2008. pp. 190-197. [ Links ]
4. G. Arzamendi, E. Arguiñarena, I. Campo, S. Zabala, L. Gandía. "Alkaline and alkaline-earth metal compounds as catalysts for methanolysis of sunflower oil". Catal. Today. Vol. 133. 2008. pp. 305-313. [ Links ]
5. H. Zhu, Z. Wu, Y. Chen, P. Zhang, S. Duan, X. Liu, Z. Mao. "Preparation of biodiesel catalyzed by solid super base of calcium oxide and its refining process". China J. Catal. Vol. 27. 2006. pp. 391-396. [ Links ]
6. C. Reddy, R. Oshel, J. Verkade. "Room-temperature conversion of soybean oil and poultry fat to biodiesel catalyzed by nanocrystalline calcium oxides". Energy & Fuels. Vol. 20. 2006. pp. 1310-1314. [ Links ]
7. M. Kouzu, T. Kasuno, M. Tajika, S. Yamanaka, J. Hidaka. "Active phase of calcium oxide used as solid base catalyst for transesterification of soybean oil with refluxing methanol". Appl. Catal. A. Vol. 334. 2008. pp. 357-365. [ Links ]
8. K. Tanabe, M. Misono, Y. Ono, H. Hattori. "Determination of acidic and basic properties on solid surfaces". Studies in Surface Science and Catalysis. Vol. 51. 1989. pp. 14-25. [ Links ]
9. D. E. López, J. G. Goodwin Jr., D. A. Bruce, E. Lotero. "Transesterification of triacetin with metanol on solid acid and base catalysts". Appl. Catal. A. Vol. 295. 2005. pp. 97-105. [ Links ]
10. A. Kawashima, K. Matsubara, K. Honda. "Development of heterogeneous base catalysts for biodiesel production". Bioresource Technol. Vol. 99. 2008. pp. 3439-3443. [ Links ]
11. B. C. Gates, T. K. Cheung. "Sulfated zirconia and iron- and manganese-promoted sulfated zirconia: do they protonate alkanes?" Top. Catal. Vol. 6. 1998. pp. 41-47. [ Links ]
12. J. M. Marchetti, A. F. Errazu. "Comparison of different heterogeneous catalysts and different alcohols for the esterification reaction of oleic acid". Fuel. Vol. 87. 2008. pp. 3477-3480. [ Links ]
13. U. Schuchardt, R. Serchelia, R. M. Vargas. "Transesterification of Vegetable Oils: a Review". J. Braz. Chem. Soc. Vol. 9. 1998. pp. 199-210. [ Links ]
14. Y. Asakuma, K. Maeda, H. Kuramochi, K. Fukui. "Theoretical study of the transesterification of triglycerides to biodiesel fuel". Fuel. Vol. 88. 2009. pp.786-791. [ Links ]
15. C. Stavarache, M. Vinatoru, R. Nishimura, Y. Maeda. "Fatty acids methyl esters from vegetable oil by means of ultrasonic energy". Ultrason. Sonochem. Vol. 12. 2005. pp. 367-372. [ Links ]
16. X. Liu, X. Piao, Y. Wang, S. Zhu, H. He. "Calcium methoxide as a solid base catalyst for the transesterification of soybean oil to biodiesel with methanol". Fuel. Vol. 87. 2008. pp. 1076-1082. [ Links ]
17. L. C. Meher, D. Vidya Sagar, S. N. Naik. "Technical aspects of biodiesel production by transesterification - a review". Renew. Sust. Energ. Rev. Vol. 10. 2006. pp. 248-268. [ Links ]
18. S. Yan, M. Kim, S. O. Salley, K. Y Simon Ng. "Oil transesterification over calcium oxides modified with lanthanum". Appl. Catal. A. Vol. 360. 2009. pp. 163-170. [ Links ]
19. Y. Ono. "Solid base catalysts for the synthesis of fine chemicals". J. Catal. Vol. 216. 2003. pp. 406-415. [ Links ]
(Recibido el 03 de febrero de 2010. Aceptado el 15 de octubre de 2010)
*Autor de correspondencia: teléfono: 57 + 7 + 634 47 46, fax: 57 + 7 + 634 46 84, correo electrónico: sgiraldo@uis.edu.co (S. Giraldo)
]]>