ARTÍCULO ORIGINAL
Advanced oxidation process H2O2/UV combined with anaerobic digestion to remove chlorinated organics from bleached kraft pulp mill wastewater
Proceso de oxidación avanzada H2O2/UV combinado con digestión anaerobia para remoción de compuestos organoclorados presentes en efluentes de papel y celulosa kraft
Diego Botelho Ruas1, Tatiana Rodríguez Chaparro2*, Eduardo Cleto Pires1
1Hydraulics and Sanitary Engineering Department. School of Engineering of Sao Carlos. University of Sao Paulo. Av. Trabalhador Saocarlense, No 400. Zip code: 13566-590. Sao Carlos - SP, Brazil.
]]> 2Civil Engineering Department. Military University. Cra. 11 No 101-80. Bogotá, Colombia.*Autor de correspondencia: teléfono: + 57 + 65 0 00 00 ext 1286, fax: + 57 + 21 47 280 correo electrónico: adela.rodriguez@unimilitar.edu.co (T. Rodríguez)
(Recibido el 21 de junio de 2011. Aceptado el 23 de mayo de 2012)
Abstract
This study investigated the application of an advanced oxidation process combining hydrogen peroxide with ultraviolet radiation (H2O2/UV) to remove recalcitrant compounds from Kraft bleaching effluent. Anaerobic pre-treatment was performed to remove easily degraded organics using a horizontal-flow anaerobic immobilized biomass (HAIB) reactor. Bleaching plant effluent was treated in the HAIB reactor processed over 19 h of hydraulic retention time (HRT), reaching the expected removal efficiencies for COD (61 ± 3%), TOC (69 ± 9%), BOD5 (90 ± 5%) and AOX (55 ± 14%). However, the anaerobic treatment did not achieve acceptable removal of UV254 compounds. Furthermore, there was an increase of lignin, measured as total phenols. The H2O2/UV post-treatment provided a wide range of removal efficiencies depending on the dosage of hydrogen peroxide and UV irradiation: COD ranged from 0 to 11%, UV254 from16 to 35%, lignin from 0 to 29% and AOX from 23 to 54%. All peroxide dosages applied in this work promoted an increase in the BOD5/COD ratio of the wastewater. The experiments demonstrate the technical feasibility of using H2O2/UV for post-treatment of bleaching effluents submitted to anaerobic pre-treatment.
Keywords: Advanced oxidation process, bleaching effluents, recalcitrant organic matter, AOX, biodegradability ratio
Resumen
]]> Este trabajo tuvo como objetivo investigar la aplicación de un proceso de oxidación avanzada basado en la combinación de peróxido de hidrógeno con la radiación ultravioleta (H2O2/UV) para eliminar compuestos recalcitrantes de presentes en los efluentes de blanqueamiento de pulpa celulosita. Con el fin de degradar la materia orgánica fácilmente hidrolizable, se utilizo como pretratamiento el reactor anaerobio horizontal de lecho fijo (RAHLF). El reactor RAHLF operó con un tiempo de detención hidráulica (TRH) de 19 horas, alcanzando eficiencias de remoción de DQO (61 ± 3%), COT (69 ± 9%), DBO5 (90 ± 5%) y AOX (55 ± 14%). Sin embargo, en el tratamiento anaerobio no se observo una reducción considerable de compuestos aromáticos medidos como UV254. Adicional a lo anterior, se presento un aumento de la lignina, medida como fenoles totales. La aplicación del proceso de oxidación avanzada como pos tratamiento del efluente anaerobio, mostró una amplia gama de eficiencias de remoción en función de la dosis aplicada de peróxido de hidrógeno y la radiación ultravioleta: la DQO varió de 0 a 11%, la UV254 del 16 al 35%, la lignina 0-29% y los AOX variaron del 23 al 54%. Todas las dosis de peróxido aplicado en este trabajo promovieron un aumento en la razón de biodegradabilidad aerobia (DBO5/DQO). Con este estudio se demuestra la viabilidad técnica del uso de H2O2/UV como post-tratamiento de efluentes previamente tratados por procesos anaerobios.Palabras clave: Procesos de oxidación avanzada, efluentes de blanqueamiento, materia orgánica recalcitrante, AOX, razón de biodegradabilidad
Introduction
The chlorolignins present in bleached effluents from a Kraft mill have high molecular weights (HWM). They are the source of difficulties in biological wastewater treatment plants since the phenolic byproducts of lignin result in recalcitrance and toxic wastewater characteristics [1-3 ]. Pulp and paper mills typically use aerobic biological systems such as activated sludge and aerated ponds as their main wastewater treatment processes. However, beginning in the 1980s, some pulp and paper mills incorporated anaerobic-aerobic biological sequences, due to several potential advantages over pure aerobic treatment: lower operational cost, sludge production, land, and nutrients requirements; energy recoverability via methane combustion; and a substantial capacity of chlorinated organic matter removal, which is a particularly important issue for bleached pulp production [4-7 ]. Removal of organochlorine compounds under anaerobic conditions has been attributed largely to ''reductive dechlorination'' [8 ]. According to these authors, the electronegative nature of the chlorine substituent increases the oxidation level of organic compounds, making them more susceptible to reduction reactions. Although combined anaerobic-aerobic treatment sequences have been successful in eliminating some chlorinated compounds, a growing concern about the remaining concentration of these compounds drives the continued search for processes that could further reduce their concentration in the discharged effluents. Therefore, several new technologies have been developed and tested in recent decades; among the most promising are advanced oxidation processes (AOPs), [9 ]. The •OH radical can also modify the chemical structure of recalcitrant organic chemicals transforming them into simpler compounds, with lower molecular mass and less toxicity toward microorganisms, which are, therefore, more biodegradable, [3, 10-14 ]. Hydrogen peroxide (H2O2) has been used for many years to remove organic matter from industrial wastewater and supply water [12, 15 ]. It is a versatile oxidizing agent that is more effective than chlorine, chlorine dioxide or potassium permanganate, and it can be converted via catalysis to hydroxyl radicals, either rapidly in the presence of ultraviolet radiation (UV) or more slowly in its absence [16, 17 ]. This research is focused on the application of the H2O2/UV AOP for treating anaerobically pre-treated bleaching plant wastewater from a Kraft pulp mill in preparation for a final aerobic polishing. To date, there have been several studies investigating the application of AOPs combined with aerobic biological treatments in recalcitrant effluents [3, 11, 13, 18 ]. However, investigations into the use of the H2O2/UV process following anaerobic pre-treatment have been scarce.
Materials and methods
Wastewater
Wastewater was obtained from a Brazilian Kraft pulp mill with ECF sequence (Elemental chlorine free).Wastewater was collected from the alkaline and acid step of the fully bleached line. These effluents are discharged by the industrial plant in the ratio of 60% and 40%, respectively. The effluents were collected in two times separately and mixed in the ratio of 60%:40% in the laboratory and storage at 4°C until use. This mixture is the model wastewater for this research. The residual hydrogen peroxide in the bleaching effluent was much lower than the applied dosages (< 3.0 mg L-1).
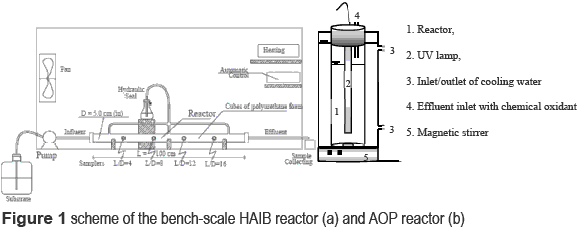
]]> Anaerobic treatmentThe horizontal-flow anaerobic immobilized biomass (HAIB) was selected for the biological treatment. The HAIB bioreactor was comprised of a 1000 mm long (L), 50 mm diameter (D) acrylic cylinder with a total volume of approximately 1964 mL and an L/D of 20. Polyurethane foam cubes (5 mm in size with an apparent density of 23 kg/m3) were used as biomass immobilization support. The reactor was inoculated with granulated sludge from an UASB reactor treating effluent from poultry slaughterhouse (Avicola Dacar S.A., Tiete, state of Sao Paulo, Brazil) (figure 1). The sludge was macerated and immobilized in the foam according to [19 ]. The HAIB reactor was kept in an acclimatized chamber set at 30°C±1°C during 112 days and operated with hydraulic retention time of 19 h based on the net volume of the reactor. Prior to feeding the wastewater into the anaerobic reactor, the pH was adjusted to between 7.0 and 7.5. Nitrogen (N) and phosphorous (P) were added as urea and monobasic sodium phosphate, respectively, to obtain a COD:N:P ratio equal to 500:5:1, as suggested by [20 ].
Post-treatment with the advanced oxidation process H2O2/UV
The HAIB reactor effluent was submitted to post-treatment with hydrogen peroxide and ultraviolet radiation, in an attempt to promote the removal of recalcitrant organic matter and to increase the biodegradability ratio of the remaining compounds. The AOP experiments were carried out in a cylindrical tube reactor made of borosilicate glass, which is mounted inside another glass tube such that a thermostatic bath can enclose the reactor, as shown in figure 1. The reactor was 54 cm high and had an internal diameter of 7 cm, with a net volume of 1000 mL. This experiment was conducted in a batch mode. A 15W low-pressure UV lamp with 254 nm emission, a length of 45 cm long and diameter of 2.5 cm (Starlux G15T8) was inserted vertically into the centre of the reactor.
The hydrogen peroxide was introduced at the beginning of the treatment process by thoroughly mixing the necessary amount of a 35% hydrogen peroxide solution with one litre of the sample to be treated at the start of the treatment. All experiments were performed with analytical reagent grade hydrogen peroxide. The experiments were performed with four sample replicates using 50, 100, 250 and 500 mg L-1 H2O2 and applying ultraviolet radiation for 15, 30, 45 and 60 minutes. Where possible the results are expressed in terms of ''applied energy'' (kWh m-3 of treated effluent) to show the order of magnitude of the energy consumed by the H2O2/UV process. Since the volume treated in the AOP reactor was one litre and the power consumption of the ultraviolet radiation lamp was 15 W, one hour of ultraviolet irradiation corresponds to an applied energy of 15 kWh m-3. The residual H2O2 in the wastewater after AOP treatment was neutralized by adding sodium sulphite (Na2SO3) to avoid any further reaction that could interfere with the interpretation of the experimental results. Prior to neutralization with sodium sulphite a sub-sample was collected for immediate measurement of the residual hydrogen peroxide concentration.
Analytical methods
Analyses of COD, BOD5, TOC, UV254, lignin and pH were performed according to [21 ]. Modifications proposed by [22 ] were applied to determine the total volatile acid concentration and alkalinity. The AOX determinations followed the [23 ] batch method using a Thermo Glass/Dextar apparatus. The chloride ion analysis was carried out using the thiocyanate mercury method. The residual hydrogen peroxide present in the samples was measured in accordance with [24 ] using a DR 4000/HACH spectrophotometer. The presence or absence of H2O2 was also qualitatively tested with Merckoquant kits (MERCK).
]]> Results and discussion
Anaerobic treatment
Reactor stability (less than 10% variation in COD removal efficiency) was achieved after a start-up period of 44 days. The anaerobic reactor was subsequently monitored over 112 days of continuous operation. The removal efficiencies achieved for COD, BOD5, and TOC (seen in table 1) can be considered adequate, because the HAIB reactor would function as an initial treatment for removing easily degradable organics. However, there was only 10% removal of UV254 compounds in the biological system. The anaerobic reactor maintained satisfactory operational stability, with an applied average organic load of 1.47 ± 0.08 kg COD m-3 d-1 over the experimental period. [25 ] compared results from several authors and concluded that the COD removal efficiency is around 60% in anaerobic reactors treating bleaching effluents. Biodegradability, as represented by the BOD5/COD ratio, decreased from the influent (0.29 ± 0.07) to the effluent (0.06 ± 0.03). This indicated that the effluent entering post-treatment with H2O2/UV was of inherently recalcitrant nature [26 ]. The pH values were 7.1 ± 0.2 for influent samples and 8.0 ± 0.1 for the effluent. The bicarbonate alkalinity showed higher values for the effluent (589 ± 53 mg CaCO3 L-1) than for the influent (302 ± 129 mg CaCO3 L-1). The ratio of intermediate alkalinity to partial alkalinity (IA/PA) of the effluent in the anaerobic reactor (0.23 ± 0.08) varied within the range of values found in stable anaerobic conversion processes, as suggested by [22 ].
Volatile acids were successfully removed to a significant extent, with average values in the influent and effluent of 123 ± 17 and 47 ± 7 mg HCHO L-1, respectively. The results show increment of lignin concentration from the influent (20 ± 5.8 mg L-1) to the effluent (30 ± 1.8 mg L-1) of the HAIB reactor. According to [21 ], the lignin analysis via colorimetry method (Method 5550-A; [21 ]) using the folin-phenol reagent is not specific for lignin, but rather takes other phenolic compounds into account; thus the lignin is expressed in terms of ''mg phenol L-1''. Some of these compounds, such as aromatic chemical groups and intermediate chemicals produced by the anaerobic digestion, can affect the chemical analysis of lignin [27 ]. With regard to the chlorinated organic compounds, the HAIB reactor provided an average AOX removal efficiency of 55%, resulting in an average effluent concentration of 10 ± 1.6 mg L-1. [7 ] mentioned that AOX removal efficiency ranges from 40 to 65% in the anaerobic treatment of pulp and paper wastewaters. In fact, aerobic treatment systems also present similar AOX removals in pulp mill effluents. [4 ] noted studies in which the average AOX removal efficiency in activated sludge systems range from 40 to 60%. Because of its similar removal efficiencies, the anaerobic technology may be a better option to remove chlorinated organic matter from bleached Kraft pulp mill effluents, as its operational cost is lower than the costs of activated sludge systems. Another important factor is that anaerobic bacteria can transform chlorinated compounds into compounds that can more easily be degraded by aerobic microorganisms in a subsequent stage [28 ]. Dechlorination occurred during the anaerobic treatment, as indicated by the increase in chloride ion concentration in the effluent (508 ± 48 mg L-1) compared to the influent (431 ± 61 mg L-1). Some chlorinated organic compounds were decomposed by biological oxidation, resulting in the liberation of Cl- ions, a process known as ''reductive dechlorination'' that can decrease the biorefractory and toxic characteristics of the effluents [8, 13, 29 ]. However, the mass balance between the AOX analysis and chloride ions (both parameters are expressed in terms of ''mg Cl- L-1'') was not closed. The sum average of the AOX and chloride parameters in the influent (455 mg Cl- L-1) was lower than that in the effluent (518 mg Cl- L-1). [8 ] also observed this imbalance in the analysis of AOX and chloride in anaerobic fermentation of bleached Kraft mill effluents and proposed two hypotheses to explain their observation. The first hypothesis was the release of intracellular chloride by anaerobic biomass which was not initially detected by the AOX measurement; the second was that the chlorinated compounds modified by the anaerobic digestion influenced the AOX quantification method. In the present case, the explanation may be in the chloride measurement method, since the content of AOX compared with the content of chlorine ions is not high enough to impact the mass balance if experimental uncertainties are taken into consideration.
Post-treatment with H2O2/UV
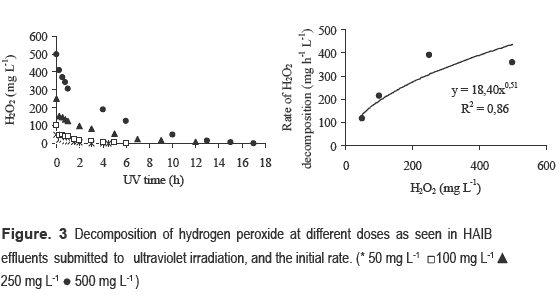
To understand the results of the post-treatment, it is helpful to look first at the H2O2 conversion over time, as seen in table 2. After one hour of reaction, 75% of the H2O2 was consumed when the dosage was 50 mg H2O2 L-1, but only 39% was consumed when 500 mg H2O2 L-1 was applied. This result has important implications when considering specific removal loads, on reactor design, and on treatment cost.
In all tested hydrogen peroxide dosages, the concentrations of COD, TOC, and chloride ions showed minimal changes after AOP treatment (figure 2a, b, and f). This is important since it implies that organic matter was not being completely mineralized, but rather transformed to a more biodegradable state (as shown in figure 2g), which was the goal of the H2O2/UV treatment. An increase in chloride ions was expected for the treated effluents, in parallel with the AOX removal. Such an effect would be caused mainly by the rupture of chlorinated organic compound chains and the resulting liberation of chloride ions, as noted by other authors [13, 29 ]. In fact, this effect was also observed in the anaerobic treatment, which increased the chloride ion concentration with the corresponding AOX removal, although not equally balanced. After one hour of treatment, the UV254 removal efficiency for 50 and 100 mg H2O2 L-1 reached nearly the same level, 16 and 17%, respectively (figure 2c). On the other hand, higher removal efficiencies were achieved for dosages of 250 and 500 mg H2O2 L-1, at 28 and 35%, respectively; however a constant decrease of the UV254 content clearly indicated that more time was necessary to reach the potential removal that these dosages provided. Subsequent experiments, shown in figure 3 and 4, indicated that one hour of treatment was not sufficient to convert all the H2O2 into hydroxyl radicals. Two observations could be made concerning lignin (as seen in figure 2d): (a) the average removal levels with 50 and 100 mg H2O2 L-1 (at the optimal energy application level of 7.5 kWh m-3) were 9 and 20%, respectively, since higher UV doses may seem to result in the formation of phenols and a ''recovery'' of lignin; (b) doses of 250 and 500 mg H2O2 L-1 promoted removal efficiencies that increased simultaneously with the increase in applied energy, reaching values of 29% when 15.0 kWh m-3 was applied. Further investigation is needed to determine the behaviour of lignin under low hydrogen peroxide doses. The AOX removal (figure 2e) was more closely related to UV radiation exposure time, or the applied energy, than to hydrogen peroxide dosage. For different H2O2 doses, there were only minor differences in AOX removal efficiencies, ranging from 45% for 50 mg H2O2 L-1 to 54% when 500 mg H2O2 L-1 was applied under exposure to 15.0 kWh m-3 of UV radiation. When the results of UV254 compound removal (figure 4) are extrapolated to AOX removal two to three hours of treatment are necessary to provide a clear distinction between the removal efficiencies of different hydrogen peroxide doses.
]]>
It seems that for higher H2O2 dosages longer UV exposures may induce the decomposition of biodegradable chemicals, as observed in the decreased biodegradability ratio for 250 and 500 mg H2O2 L-1. However, the results are inconclusive. As in other yet unpublished results from other AOP experiments, some small but consistent fluctuations in the biodegradability ratio seem to occur over time for high hydrogen peroxide doses as time passes. This behaviour is expected since intermediate compounds are formed and consumed until complete mineralization is achieved and some of these intermediate compounds may be more biodegradable than others.
This point will be addressed later on. For instance, for 250 mg H2O2 L-1 there was an increase of 125% with just 3.8 kWh m-3, while applying 15.0 kWh m-3 resulted in a107% biodegradability ratio increase. Thus, in order to minimize chemical and energy consumption H2O2 doses must be optimized. Other studies have also indicated that there are optimal oxidizing agent dosages for treating effluents with advanced oxidative processes [2, 13, 18 ]. In particular, these studies noted that at high concentrations, hydrogen peroxide itself may act as a free-radical scavenger in secondary reactions, causing a decrease in the hydroxyl radical concentration [10, 16, 17 ]. As indicated in figure 3, complete consumption of the hydrogen peroxide can take many hours. To observe the long term behaviour of the residual H2O2 concentration, COD, TOC, lignin, and UV254, the experiments shown in figure 2 were extended until all the hydrogen peroxide was consumed. This required three hours for the lowest dosage (50 mg H2O2 L-1) and 17 h for the highest dosage (500 mg H2O2 L-1). Figure 2 presents the kinetic analysis of the hydrogen peroxide decomposition during the oxidation treatment under ultraviolet radiation, as estimated by the initial rate method using the Microcal Origin 7.5® software. The order of the reaction of hydrogen peroxide in anaerobic effluents under ultraviolet radiation was approximately 0.51. This value differed from that reported by [15 ], who suggested that a pseudo-first order model could describe the reaction between H2O2/UV and bleaching effluents. However, it is noteworthy to highlight that the studied effluents had different characteristics, as the bleaching effluents used in the current work were previously submitted to anaerobic treatment. The figures 2 and 4 show that there was some saturation in the reaction rate of hydrogen peroxide in doses above 250 mg L-1 with the effluent perhaps approaching a reaction of zero order. It is also important to observe that the reaction rate decreases as the H2O2 concentration increases. The correlation coefficients can be considered adequate, (R2 = 0.86), and it is possible to state that the observed behaviour is due to the change in hydrogen peroxide dosage. Figure 4 shows the physicochemical parameters monitored during the kinetic study of the hydrogen peroxide decay. Doses of 50 mg L-1 and 100 mg L-1 provided maximum COD removals of 14% (after two hours of UV irradiation) and 19% (after four hours of UV radiation), respectively. The most extensive removals did not necessarily correspond with the highest exposure to UV radiation. According to some authors [10, 16, 17 ] this can be related either to the effect of •OH radicals that recombine to form hydrogen peroxide or to an excess of H2O2 in the reaction environment. However, the latter hypothesis is not supported by the data of this particular work since higher dosages provided higher removal of TOC and COD, thus showing that our oxidant was not dosed in excess.
TOC was not removed using the two lowest dosages regardless of UV exposure time. In fact, there was a 5 to 24% TOC increase, which may be the result of compounds originally in the colloidal state that became soluble during the advanced oxidation process; this phenomenon has been observed in some other experiments performed during this work as well as by other researchers [27 ]. Higher dosages induced significant COD and TOC removals above a UV exposure threshold. However, exact threshold values were not determined in these experiments. On the other hand, doses of 250 and 500 mg H2O2 L-1 both provided maximum COD removals of 28% after 12 hours of UV irradiation and 59% after 17 hours of UV irradiation, respectively. The maximum TOC removal reached 13% with 12 hours of UV irradiation and 46% with 17 hours of UV irradiation. These hydrogen peroxide dosages also induced TOC increases of up to 16%, mainly in the first hour of the chemical treatment. Nonetheless, after three hours of UV radiation TOC was gradually removed, indicating that the H2O2/UV process induced mineralization of the effluent. [18 ] also observed this behaviour in the TOC removal for the H2O2/UV process as applied to a synthetic effluent containing the recalcitrant compound tetrahydrofuran. The authors concluded that pretreatment of the effluent for a posterior biological treatment could be achieved with H2O2 dosages as low as 1 mol H2O2 per mol TOC, which significantly increased the biodegradability ratio of the effluent. However, to employ the H2O2/UV process as a tertiary treatment, [18 ] proposed a higher hydrogen peroxide dosage of 4 mol H2O2 per mol of TOC to promote effluent mineralization rather than just modifications of the characteristics of organic matter. It should be observed that the dosages proposed by [18 ] are specific to their work and as some simple stoichiometric calculations can demonstrate, they may not necessarily hold true for other compounds. To use their data, it would be necessary to apply approximately 1240 mg H2O2 L-1 just to increase biodegradability ratio, a dosage that is 2.5 times higher than the highest used in this work.
The maximum removal efficiencies of UV254 compounds were similar at 20% after three hours of UV exposure for 50 mg H2O2 L-1 and 22% after six hours of UV exposure for 100 mg H2O2 L-1. However, the two higher dosages applied, 250 and 500 mg H2O2 L-1, resulted in significantly increased removal efficiencies of 50% and 80%, respectively. Oscillations in the lignin removal efficiencies during exposure to ultraviolet radiation appeared during the first two hours. For doses of 50 and 100 mg L-1, the lowest lignin values were reached after 30 minutes of treatment at 26 and 21 mg L-1, respectively. Afterwards there was some increase of lignin, reaching maximum values of 32 mg L-1 after three hours of UV irradiation at 50 mg H2O2 L-1, and 36 mg L-1 after two hours of UV irradiation at 100 mg H2O2 L-1. After this, the lignin concentration maintained a relatively constant level. The other H2O2 dosages tested (250 and 500 mg H2O2 L-1) also resulted in oscillating lignin concentrations over the first two hours of UV exposure. However, in contrast with the lower dose cases, there was a general tendency of lignin reduction with increasing UV radiation exposure. As mentioned previously, the oscillatory behaviour of lignin degradation may be related to intermediate species formation from the decomposition of organic matter, with its subsequent removal, or to colloidal matter that becomes soluble and is then removed.
Conclusions
The horizontal-flow anaerobic immobilized biomass reactor performed effectively in treating effluents from the bleached Kraft pulp Mill. There was suitable removal of organic matter and chlorinated organic compounds. However, post-treatment of the effluent was necessary to achieve discharge requirements. The H2O2/UV post-treatment provided additional AOX removal (up to 54%) and increased the biodegradability ratio (up to 131%) of the anaerobic effluents. A dosage of 100 mg H2O2 L-1 and an energy level of 11.3 kWh per m3 applied by the UV radiation lamp was the optimal combination for increasing the biodegradability ratio and removing chlorinated organic compounds and lignin. The kinetic studies of complete H2O2 decomposition demonstrated good removal efficiencies for both organic matter and lignin, though long ultraviolet irradiation times may be required. It is concluded that the application of H2O2/UV as post-treatment for complex effluents from anaerobic reactors is a technically feasible option.
]]> Acknowledgements
This study was supported by Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP). Grants received from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) are gratefully acknowledged.
References
1. D. Savant, R. Abdul, D. Ranade. ''Anaerobic degradation of adsorbable organic halides (AOX) from pulp and paper industry wastewater''. Bioresource Technology. Vol. 97. 2006. pp. 1092-1104. [ Links ]
2. E. Catalkaya, F. Kargi. ''Color, TOC and AOX removals from pulp mill effluent by advanced oxidation processes: A comparative study''. Journal of Hazardous Materials B139. Vol. 139. 2007. pp. 244253. [ Links ]
3. L. Bijan, M. Mohseni. ''Integrated ozone and biotreatment of pulp mill effluent and changes in biodegradability and molecular weight distribution of organic compounds''. Water Research. Vol. 39. 2005. pp. 3763-3772. [ Links ]
]]>4. D. Pokhrel, T. Viraraghavan. ''Treatment of pulp and paper mill wastewater - a review''. Science of the Total Environment. Vol. 333. 2004. pp. 37-58. [ Links ]
5. G. Vidal, M. Soto, J. Field, R. Mendez, J. Lema. ''Anaerobic biodegradability and toxicity of wastewaters from chlorine and total chlorine-free bleaching of eucalyptus kraft pulps''. Water Research. Vol. 31. 1997. pp. 2487-2494. [ Links ]
6. G. Lettinga. ''Sustainable integrated biological wastewater treatment''. Water Science and Technology. Vol. 33. 1996. pp. 85-98. [ Links ]
7. J. Rintala, J. Puhakka. ''Anaerobic treatment in pulp and paper-mill waste management - a review''. Bioresource Technology. Vol. 47. 1994. pp. 1-18. [ Links ]
8. W. Parker, E. Hall, G. Farquhar. ''Evaluation of dechlorination mechanisms during anaerobic fermentation of bleached kraft mill effluent''. Water Research. Vol. 27. 1993. pp. 1269-1273. [ Links ]
]]>9. J. Carey, E. Hall, N. McCubbin. Review of scientific basis for AOX effluent standard in British Columbia. Minister of Water. Land and Air Protection in British Columbia. Disponible en: http://www.environment.gov.au/epbc/notices/assessments/2007/3385/pubs/20dioxins.pdf. 2002. [ Links ]
10. C. Huang, C. Dong, Z. Tang. ''Advanced chemical oxidation: its present role and potential future in hazardous waste treatment''. Waste Management. Vol. 13. 1993. pp. 361-377. [ Links ]
11. D. Ruas, A. Mounteer, A. Lopes, B. Gomes, F. Brandao, L. Girondoli. ''Combined chemical biological treatment of bleached eucalypt kraft pulp mill effluent''. Water Science and Technology. Vol. 55. 2007. pp. 143-150. [ Links ]
12. P. Gogate, A. Pandit. ''A review of imperative technologies for wastewater treatment I: oxidation technologies at ambient conditions''. Advances in Environment Research. Vol. 8. 2004. pp. 501-551. [ Links ]
13. M. Yeber, J. Rodriguez, J. Freer, J. Baeza, N. Duran, H. Mansilla. ''Advanced oxidation of a pulp mill bleaching wastewater''. Chemosphere. Vol. 39. 1999. pp. 1679-1688. [ Links ]
]]>14. A. Marco, S. Esplugas, G. Saum. ''How and why combine chemical and biological processes for wastewater treatment''. Water Science and Technology. Vol. 35. 1997. pp. 321-327. [ Links ]
15. C. Prat, M. Vicente, S Esplugas. ''Treatment of bleaching waters in the paper-industry by hydrogen- peroxide and ultraviolet-radiation''. Water Research. Vol. 22. 1988. pp. 663-668. [ Links ]
16. J. Crittenden, S. Hu, D. Hand, S. Green. ''A kinetic model for UV/ H2O2 process in a completely mixed batch reactor''. Water Research. Vol. 33. 1999. pp. 2315-2328. [ Links ]
17. O. Legrini, E. Oliveros, A. Braun. ''Photochemical processes for water-treatment''. Chemical Review. Vol. 93. 1993. pp. 671-698. [ Links ]
18. C. Raj, H. Quen. ''Advanced oxidation processes for wastewater treatment: Optimization of UV/H2O2 process through a statistical technique''. Chemical Engineering Science. Vol. 60. 2005. pp. 5305-5311. [ Links ]
]]>19. E. Foresti, M. Zaiat, A. Cabral, V. Nery. ''Horizontal- flow anaerobic immobilized sludge (HAIS) reactor for paper industry wastewater treatment''. Brazilian Journal of Chemical Engineering. Vol. 12. 1995. pp. 157-163. [ Links ]
20. B. Ammary. ''Nutrients requierements in biological industrial wastewater treatment''. African Journal of Biotechnology. Vol. 3. 2004. pp. 236-238. [ Links ]
21. APHA. Standard Methods for the Examination of Water and Wastewater. 21th ed. Ed. American Public Health Association. Washington, D.C. 2005. pp. 1325. [ Links ]
22. L. Ripley, W. Boyle, J. Converse. ''Improved alkalimetric monitoring for anaerobic-digestion of high-strength wastes''. Journal (Water Pollution Control Federation). Vol. 58. 1986. pp. 406-411. [ Links ]
23. Scan, Scandinavian Pulp. Paper and Board Testing Committee. Test Methods. Estocolmo. 1989. [ Links ]
]]>24. N. Klassen, D. Marchington, H. McGowan. ''H2O2 Determination by the I3- Method and by KMnO4 titration''. Analytical Chemistry. Vol. 66. 1994. pp. 2921-2925. [ Links ]
25. G. Vidal, S. Videla, M. Diez. ''Molecular weight distribution of Pinus radiata kraft mill wastewater treated by anaerobic digestion''. Bioresource Technology. Vol. 77. 2001. pp. 183-191. [ Links ]
26. Metcalf & Eddy. Wastewater Engineering treatment Disposal Reuse. 4th ed. Ed. McGraw - Hill Book. New York. 2003. pp. 1815. [ Links ]
27. R. Pereira. Combination of aerobic biological treatment with advanced oxidative process seeking to intensify organic matter removal in textile and kraft pulp mill effluents. Dissertation (Magister Scientiae), Federal University of Vifosa, Vifosa. (MG-Brazil). 2007. pp. 80. [ Links ]
28. J. Field, R. Sierra. ''Microbial transformation and degradation of polychlorinated biphenyls''. Environmental Pollution. Vol. 155. 2008. pp. 1-12. [ Links ]
]]>29. T. Essam, M. Amin, O. El Tayeb, B. Mattiasson, B. Guieysse. Sequential photochemical-biological degradation of chlorophenols''. Chemosphere. Vol. 66. 2007. pp. 2201-2209. [ Links ]
]]>