TRANSFORMACIÓN ENZIMÁTICA DE LA ESTRUCTURA CRISTALINA DEL COPOLÍMERO POLIETIL-VINIL ALCOHOL (EVOH)
Carolina ARBOLEDA E.1,2, Freimar SEGURA S.1,2 y Amanda I. MEJÍA G.1,2*
1 Grupo Biodegradación y Bioconversión de Polímeros - BIOPOLIMER, Facultad de Química Farmacéutica, Universidad de Antioquia. A.A. 1226. Medellín, Colombia
2 Grupo Ciencia de los Materiales, Instituto de Química, Facultad de Ciencias Exactas y Naturales, Universidad de Antioquia. A.A. 1226. Medellín, Colombia
* Autor a quien se debe dirigir la correspondencia: amejia@quimbaya.udea.edu.co
Recibido: Septiembre 21 de 2006 Aceptado: Abril 17 de 2007
ABSTRACT
Changes in the crystalline structure of the copolymer poly(ethylene-co-vinyl alcohol) (EVOH) that occurs during the enzymatic fermentation system with the fungus Phanerochaete chrysosoporium were studied. This fungus has a powerful group of oxidative enzymes that had been used for several processes of environmental pollutants degradation. In order to use the enzymatic system of the fungus to improve degradation, films of the copolymer are inoculated and maintained in a solid state fermentation (SSF) system using as substrate a mixture of agroindustrial corn wastes with an initial humidity of 90%, during forty days. Analyses of the films were made at different times of fermentation. The effect on the chemical structure of the polymer was studied by using FTIR-spectroscopy with attenuated total reflection (ATR) and changes in the crystalline structure were determined by differential scan calorimetry DSC and X-ray diffraction technique. The lignin peroxidase (LiP) activity was followed by UV-spectrophotometry. We concluded that after 20 days of fermentation the copolymer has lost 50 percent of the initial crystalline structure.
Keywords: poly(ethylene-co-vinyl alcohol), lignin peroxidase, Phanerochaete chrysosporium, crystalline structure.
RESUMEN
Se estudia el cambio en la estructura cristalina del copolímero polietilvinil alcohol (EVOH) que tiene lugar durante el sistema de fermentación enzimática con el hongo Phanerochaete chrysosporium. El hongo cuenta con un poderoso grupo de enzimas oxidativa, que han sido utilizadas en la degradación de varios contaminantes ambientales. Con el fin de utilizar el sistema enzimático de este microorganismo en la degradación del EVOH, las películas del copolímero fueron inoculadas y mantenidas bajo un sistema de fermentación en estado sólido, utilizando como sustrato la mezcla de residuos agroindustriales de maíz, con una humedad inicial del 90%, durante 40 días. El análisis de las películas se llevó a cabo en diferentes tiempos de fermentación; los cambios en la estructura cristalina fueron determinados por calorimetría diferencial de barrido DSC y difracción de rayos X. La actividad de la enzima ligninoperoxidasa fue seguida por espectrofotometría ultravioleta. Concluimos que después de 20 días de fermentación, el copolímero pierde el 50% de la cristalinidad inicial.
Palabras clave: Polietilvinil alcohol, ligninoperoxidasa, Phanerochaete chrysosporium, estructura cristalina.
INTRODUCTION
]]> With more and more plastics being employed in human lives and increasing pressure being placed on the capacities available for plastic waste disposal, the need for biodegradable plastics and the biodegradation of plastic wastes has assumed increasing importance in the last few years.Recent research has shown that thermoplastics derived from polyolefin are traditionally considered resistant to environmental degradation. In this way some biodegradable plastics have been marketed, nevertheless present limitations and obstacles for use.(1)
The EVOH is a copolymer of polyethylene (PE) and polyvinyl alcohol (PVOH); its application has been extended mainly for food packaging because of its excellent gas barrier properties(2), flavors permeability resistance and good mechanical properties, significantly increasing the storage time of foods packed on this material. This polymer, like many others, causes an environmental problem because of the great volume of solid wastes that remain once the foods are consumed (3). It is necessary to find an alternative way for the treatment of those wastes. EVOH is a material which includes a PVOH segment that is susceptible to be degraded and in this way to continue the complete biodegradation of the material It is a good method for the disposition of those wastes used daily in food packaging(4).
The EVOH biodegradation under submerged culture using the enzymatic system of the fungus basydiomycete Phanerochaete chrysosporium has been studied (5).Nevertheless this way turns out to be more expensive. On the other hand, the fermentation in solid state (SSF) simulates better environmental and nutritional fungal conditions (6). The agro-industrial wastes are consider good substrates for the culture of ligninolytic fungi like the Phanerochaete chrysosporium, that act on the lignin structure and release the carbon source. Many of those agro-industrial wastes are also consider pollutants(7). The basydiomycete Phanerochaete chrysosporium is a white rot fungus widely studied, that produces a great number of extracellular oxidative enzymes during the secondary metabolism under limited conditions of nitrogen or carbon(9), among them the lignino peroxidase, (LiP) and the manganese peroxidase (MnP), which can be induced with the aid of co-substrates as the veratrílico alcohol and the manganese(8) We have studied the effect of the Phanerochaete chrysosporium in a system of solid fermentation on corncob and cornhusk, on films of EVOH at different times of fermentation (10). In this study was determinated the effect that has the Phanerochaete chrysosporium in a system of solid fermentation in cornhusk and corncob, on films of EVOH that are determinate at different times of fermentation. The copolymer films were characterized by FTIR-spectroscopy to observe the main functional groups changes, and by differential scan calorimetry (DSC) and X-ray diffraction we determinated changes in the melting and crystallization temperature and changes in the crystalline structure of polymer.
METHODS AND MATERIALS
Polymer
Copolymer Polietil-Co-Vinil Alcohol (EVOH) 22 µm thickness, containing a 29% of polyethylene (Elastochem, Spain, donated by the Agro-alimentary Institute of Valencia) was used.
Fungus
Phanerochaete chrysosporum stock BKM-F-1767 (ATCC 24725) cultivated in YMPG medium. It is incubated for 5 days to 37°C and stored at 4°C. For the experiment, the spores are scraped and dissolved in tween 80 at 0.5 % p/v, and mixed in vortex by 3 minutes. The amount of spores was determined by the spectrophotometric method described by Jiménez ET to (1999). (8)
Substrate
]]> The solid substrate was made from cornhusk and corncob collected in the east of Antioquia at 1.800 meters sea-level at 18° C. Cornhusk and corncob were dried to 55° C during 48 hours and sifted by 0.5cm sieve.Basal Media
Sterile solution sodium tartrate buffer pH 4.5, added with veratrylic alcohol (AV) 2.5mm and tween 80 to 0.05 % (p/v).
Solid state fermentation system SSF
A mixture of equal amounts of cornhusk and corncob adjusted with basal media at initial humidity (HI) of 90%, was inoculated with spores solution 5.4x106 spores/g of substrate. The copolymer films were previously washed with alcohol, dried, weighed and them inoculated. Static beds reactors were maintained in a stable relative humidity 60% at 24°C room temperature. The experiments were made by triplicate and one control under the same conditions without fungus. The conditions of humidity, size of inoculum and temperature was determinated in a previous work by Bumpus (7).
Activity of the LiP
A sample of 200 to 400 μl of extracellular liquid is added into a quartz cell of 1 milliliter. Sodium tartrate buffer solution 0.15M (pH=3) was added until reach 800μl and 50μl of AV 10mM is added. The reaction begins with the addition of 50ml of H2O2 (50μl/50ml H2O recently prepared). The absorbance was measured at 310 nm wavelength by U.V-stpectrophotometry. (11)
U.V.
A scan between 200 and 1000 nm becomes to each film (dry and to constant weight) is made in a spectrophotometer 50 Cary Bio.
FTIR-spectroscopy
]]> The films previously washed with ethanol to 70% and dried at 50° C by 24 hours, were cut as large as ATR quartz cell and were run in a Mattson 5000 with ATR Beijing Madison. Thirty two Interferogram scans were averaged to give spectra from 400 to 5000 cm-1Differential scanning calorimetric (DSC)
The analyses were made in a differential scanning calorimeter DSC TA instrument 2920. Initially to erase the thermal history of polymer, a heating procedure until temperature reached 15° C over the melting temperature of polymer was made, in which an isotherm for 5 minutes became followed by a controlled cooling with helium at a rate 20°C/min and then equilibrated to 50º C, registering crystallization exotherm. A second heating was made at 10 ºC/min to obtain the melting endotherm. The analysis was made under nitrogen atmosphere at 40 ml/min.
X-ray Difraction
A X-ray Difractometer Rigaku Miniflex type with Rx: Cu Source and plate holder was used to analyze the films. Samples were analyzed under the following conditions: 30 KV, 10 mA. A scan was made from 3ºC o 40ºC t at a rate of 2ºC/min at room temperature, λ = 1.5418 A°.
RESULTS
Activity of the lignin peroxidase
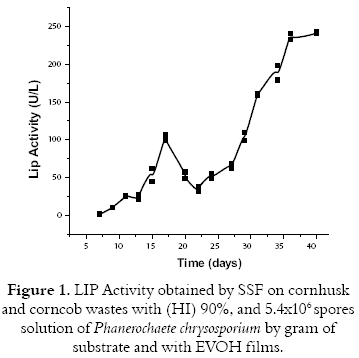
The activity of the enzyme lignin peroxidase was determined every 2 days. (See figure 1). the activity increases reaching the maximum activity corresponding to 243 u/l by the 40th day. Nevertheless the production of the enzyme not always increased as it was observed by the 17th day when it descends strongly. This can be due because the most complex structures of the substrate like the lignin has been degraded into molecules of lower molecular weight and it has been released to new carbon sources like the cellulose and the hemicellulose; they are more assimilable for the microorganism and its enzymatic production diminishes, since the enzymes are produced in limited nutrients conditions.
Analysis of film EVOH
]]> Qualitative analysis of the films of EVOH by ultraviolet spectroscopy: The EVOH shows characteristic bands at 230 and 280 nm, corresponding to the PVOH, and is observed that these bands decrease with degradation time. Which indicates that in the analyzed film of EVOH, are less groups C=O (corresponding to the 280nm band to) and a lower amount of groups C=C corresponding to the 230nm band. However it is observed that those bands move towards smaller wavelengths; these changes may occurr because in treated EVOH film the PE predominates and does not shows characteristic bands in the UV region. These analyses confirm that the part of the EVOH structure that has been biodegraded is the corresponding to PVOH. (Data not shown). The groups OH that are present in the PVOH can facilitate the beginning of oxidative reactions (12) caused by the enzymes that Phanerochaete chrysosporium produces during its secondary metabolism.DSC thermograms analyses: The thermograms of the initial EVOH films (as it is obtained commercially) and the EVOH films exoterms of crystallization and endotherms of melting were obtained at 19, 29 and 36 days of biodegradation (see figure 2 and figure 3). In figure 2 it is observed how crystallization exotherm diminishes 2.55ºC after 36 days of degradation in relation to initial polymer.
In figure 3 the melting temperature (Tm) of the initial film, at 184°C corresponds to EVOH with 29% of ethylene (13). It is observed in the thermograms by the 11th day of incubation an increase in the (Tm).This increase can be due to the molecule breaking that crosses the amorphous regions. Nevertheless, a decrease of the (Tm) is observed from the 184,92 of initial polymer to 183.51ºC EVOH at 36th day of culture, what indicates a loss of crystalline ordering of polymer. Although there was not a great decrease in the temperature, the change in the enthalpy of melting is significant and the percentage in the crystallinity of polymer is lower.
Kinetic of Biodegradation of the EVOH by DSC: In order to determine the kinetic of the reaction, the percentages of crystallinity of each film were obtained. The crystallinity degree is determined by the area under the curve of a first order transition; by measurement of enthalpy in that transition. The crystallinity degree can be calculated determining enthalpy by gram of the area under the curve and divided by enthalpy of melting of the completely crystalline material or the material with greater crystallinity (14). In biodegradation studies, enthalpy is taken from material obtained at time zero of biodegradation, as reference to the higher greater crystallinity. The values of Hm and Hc were obtained using a thermal analysis software (see figures 2 and 3).
In these experiments the loss of crystallinity in the EVOH samples, follows a cero order kinetic law and the half life time - t50- is observed when the EVOH has lost 50% of initial crystallinity. - Co was calculated by the following equation:
t50=0.5 xs Co/k=0.5 58,6124982/1.4967 xs=19.6 days
In which the EVOH film lost the half of its initial crystallinity. The kinetic model of the biodegradation of the EVOH under the conditions studied was order 0. This indicates that the loss of polymer crystallinity is constant in time and does not depend on the concentration of the EVOH resting; it is unimolecular.
The diffraction patterns were taken at 20, 24, 29, 36 and 40 days of degradation (see figure 4). The initial sample corresponds to the commercial EVOH not degraded. Crystalline picks at 19.86º and 29.22º for the initial sample are observed, that corresponds to the reflection planes of (110) and (200) respectively of the orthorhombic form of polyethylene. (15).
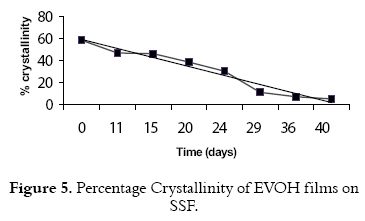
A decrease in the intensity picks is observed with degradation time, indicating a process of deformation of the crystalline zones, due to the attack of the enzymatic system of the fungus and to the expulsion of parts of the chain that constituted the main chain of polymer. From difractograms we also calculated the cristallinities of the samples, comparing the area of the crystalline peaks to the total area. An increase of the area of crystalline picks compared with “background” (that indicates the amorphous portion of polymer) represents the degree of crystallinity of the sample (15). The collected data from ray-x analysis represent the intensity of crystalline area IC, on total intensity IT (see figure 5). It is observed that after 40 days of degradation, the film conserves only 10% of its crystallinity.
]]>
Qualitative analysis of the films by ATR - FTIR: The samples show a difference in the intensity of the band 2920 cm-1 and the band placed at minor ν, that indicated the changes in the relation between stretching -CH groups of saturated bonds and =CH unsaturated chain bonds. In the same way, the appearance of a new band is observed at ν=1502 cm-1, this band is not characteristic of EVOH; it is assignable to α, β diketones groups, that confirms oxidations of the chain in the EVOH (4) and the formation of C=C isolated in the polymer chain. This is similar to the previous experiments. (10) Data not shown The fact that EVOH obtains a greater amount of instaurations and diketones groups confirm that has undergone important changes in its chemical structure. Also OH groups of the PVOH provide a hydrophilic character to the films, allowing a greater mycelia adhesion to the polymer. (12)
DISCUSSION
The solid state fermentation is being used successfully for production of secondary metabolites, like enzymes and others, with the additional advantage to take solid agro-industrial wastes for the process, a great variety of agricultural wastes that in the most part of time are also considered environmental pollutants.(16) In this work it is used to obtain a stable system for the LiP enzymes production, which is an indicator that the enzymatic system of Phanerochaete chrysosporium is active and is able to break the EVOH bonds in the inoculated films. The loss of crystallinity of the films of EVOH followed by DSC and x-rays analysis shows cero order kinetics. This kinetic model indicates that the loss of polymer crystallinity is constant with time, and does not depend on the EVOH concentration. The EVOH films showed, at 36 days of biodegradation, smaller melting points and crystallization points that confirm the ruptures of the polymeric chain, giving therefore an EVOH of smaller molecular weight. The results obtained by DSC give 19.5 days so that the initial EVOH loses half of their crystallinity, whereas by X-ray analysis the time is 20.5 days. The difference between the crystallinity value obtained by different methods is usual in polymers (17,18) due to the applied methodology. It is assumed inherent to the instrumental limitations. The analyses by DSC and X-ray, allow to conclude that the enzymes of the Phanerochaete chrysosporium were able to modify the crystalline structure of polymer, producing a polymer with a more disordered structure. This increases the permeation rate and diminishes its dimensional stability, allowing a greater access to oxidative enzymes of this microorganism. The metabolic system of the Phanerochaete chrysosporium has characteristic aspects as they are the extracellular enzyme production, composed by peroxides and veratrylic alcohol, as well as the colonization of the microorganism in form of hifas, that have a synergic effect on the degradation of the EVOH films, providing a more a oxidized surface more prone to the attack by microorganisms. On the other hand the fraction of polyvinyl alcohol provides a hydrophilic atmosphere to the polymer, allowing a greater adhesion of the microorganism and in this way a greater contact with the enzymatic system that is highly oxidative. The close union that exists between the PE and the PVOH in the EVOH allows the microorganism to have access to the structure of the polyethylene that is in this case the no biodegradable segment.
CONCLUSION
Biodegradation of commercial high molecular weight polyethylene proceeds slowly. Nevertheless the PVOH provides OH groups, so that this degradation may take place. Hydrophilic groups of the co-polymer films facilitate to microorganisms to get attached to the surface of films and may thus accelerate the biodegradation of higher molar mass compounds or polymer chains.
It is also important from the environmental point of view to know that the formed low molar mass compounds are bioassimilated and do not accumulate in the environment.
We concluded that after 20 days fermentation the copolymer has lost 50 percent of the initial crystalline.
ACKNOWLEDGEMENTS
To the Universidad de Antioquia’s Comité para el Desarrollo de la Investigación (CODI) and to the Université Libre de Bruxelles for funding the investigation.
]]> BIBLIOGRAPHIC REFERENCES1. Ying Z, Yanful EK, Amarjeet S. Bassi A. Review of Plastic Waste Biodegradation. Crit Rev Biotechnol 2005; 25(4):243-250. [ Links ]
2. Gaucher-Miri V, Jones GK, Kaas R, Hiltner A.,Baer E. Plastic Deformation of EVA, EVOH and their Multilayers, J Mat Sci 2002; 37(13): 2635-2644 [ Links ]
3. Ferrante MA. Packaging for the Next Millenium. Food Eng Int 1997; 22: 29-34. [ Links ]
4. Katz F. European Packages Continue Tradition of Innovation with a Green Tint. Food Technol 1999; 53 (1): 88. [ Links ]
5. Mejía A, López B, Sierra L. Biodegradation of Polyvynilalcoholco- ethylene with Enzymatic Extract of Phanerochaete chrysosporium. Mater Res Innovat 2001; 4 (2-3):148-154. [ Links ]
6. Robinson T, Singh D, Nigam P. Solid State Fermentation: a Promising Microbial Technology for Secondary Metabolite Production. Appl,Microbiol Biotechnol.2001;55 (3):284-289. [ Links ]
7. Bumpus JA. Biodegradation of Polycyclic Hydrocarbons by Phanerochaete chrysosporium. Appl Environ Microbiol 1989; 55(1):154-158. [ Links ]
8. Malherbe S, Cloete TE. Ligninocellulose Biodegradation. A Review. Environ Sci Biotechnol. 2003; 1(2):105-114. [ Links ]
9. Wainwright M. Introducción a la biotecnología de los hongos. Zaragosa, España: John Wiley; 1995. [ Links ]
10. Arboleda C, Mejía A, López B. Ethyl Vinyl Alcohol Biodegradation on Semi Solid-State Fermentation by the Phanerochaete chrysosporium. Acta Farmacéutica Bonaerense 2004; 23 (2):123-8 [ Links ]
11. Jiménez GA, Mejía AI, López BL. Actividad de las enzimas ligninolíticas del Phanerochaete chrysosporium y su variación con la adición de Mn2+. Rev Acad Colomb Cienc 1999; 23(89):587-594. [ Links ]
12. Gugumus F. Thermoxidative Degradation of Polyolefins in the Solid State: Part 5. Kinetics of Functional Groups Formations in PE-HD and PE-LLD. Polymer Degradation and Stability 1997; 55(1):21-43. [ Links ]
13. Hawkins WL. Polymer Stabilization. New York: Wiley & Sons; 1972. [ Links ]
14. Okaya T, Ikari I. Poly(vinylalcohol) Developments-A Textbook. 2 ed. New York:. Wiley & Sons; 1992.p 195-267. [ Links ]
15. Guadagno L, Naddeo C, Vittoria V, Camino G, Cagnani C. Chemical and Morphological Modifications of Irradiated Linear Low Density Polyethyelene (LLDP). Polymer Degrad Stabi 2001; 72(1):175-186. [ Links ]
16. Manzur A, Cuamatzi F. Effect of the Growth of Phanerochaete chrysosporium a Blend of Low Density Polyethylene and Sugar Cane Bagasse. J Appl Polymer Sci 1997; 66(1):105-111. [ Links ]
17. Kong Y, Hay JN. The Enthalpy of Fusion and Degree of Crystallinity of Polymers as Measured by DSC. Eur Polymer J 2003;39(8):1721-1727 [ Links ]
18. Lagarón JM, Giménez E, Saura J, Gavara R. Phase Phase Morphology, Crystallinity and Mechanical Properties of Binary Blends of High Barrier Ethylene-vinyl Alcohol Copolymer and Amorphous Polyamide and a Polyamide-containing Ionomer. Polymer 2001; 42 (17): 7381-7394. [ Links ] ]]>