www.pubmed.com; y los términos utilizados en la búsqueda fueron: antiagregantes plaquetarios, aspirina, dipiridamol, clopidogrel, abciximab, entre otros; luego se buscaron los textos completos de los artículos que interesaban y además se utilizaron algunos libros de hematología.]]>
www.pubmed.com. The terms used in the search were: platelet antiaggregant, aspirin, dipyridamole, clopidogrel, abciximab, among others, then we searched the full texts of articles of interest and also used some hematology books.]]>
Antiagregantes plaquetarios: mecanismos de acción y riesgos asociados al uso
Platelet antiaggregants: mechanisms of action and use asocied risks
Iván F. PALOMO G.1*; Constanza I. TORRES U.1*; Rodrigo E. MOORE-CARRASCO.1; Marcelo A. ALARCÓN L.1; Patricio J. MARAGAÑO L.1, 2
1Programa de Investigación en Factores de Riesgo de Enfermedad Cardiovascular (PIFRECV), Departamento de Bioquímica Clínica e Inmunohematología, Facultad de Ciencias de la Salud, Universidad de Talca. P.O. box: 747. Talca, Chile
2Unidad de Cardiología, Hospital Regional de Talca, Chile
]]>
RESUMEN
Las Enfermedades Cardiovasculares son la principal causa de muerte en el mundo. Junto con los esfuerzos en disminuir los factores de riesgo cardiovascular, se recurre a diversos medicamentos para disminuir la morbimortalidad asociada a estas enfermedades, entre otros fármacos se utilizan, antihipertensivos, antidiabéticos, hipolipimiantes y antiagregantes plaquetarios. Dada la participación de las plaquetas, tanto en las etapas precoces de la aterogénesis como en la trombosis arterial, el uso de antiagregantes plaquetarios es muy relevante en la prevención primaria y secundaria de las trombosis arteriales. Los antiagregantes plaquetarios son utilizados en prevención primaria y secundaria de ECV. En esta revisión, se abordan diversos aspectos relativos a los antiagregantes, especialmente mecanismo de acción y riesgos asociados al uso durante los últimos 10 años. Como fuente bibliográfica se utilizó principalmente www.pubmed.com; y los términos utilizados en la búsqueda fueron: antiagregantes plaquetarios, aspirina, dipiridamol, clopidogrel, abciximab, entre otros; luego se buscaron los textos completos de los artículos que interesaban y además se utilizaron algunos libros de hematología.
Palabras clave: antiagregantes plaquetarios, enfermedades cardiovasculares.
ABSTRACT
Cardiovascular diseases are the main cause of death in the world. Next to efforts to reduce the cardiovascular risk factors, several drugs are resorted to decrease morbimortality associated to these diseases: antihypertensive, antidiabetic, hypolipemiant and platelet antiaggregant drugs are used among others. Due to the participation of platelets, not only in the early stages of the atherogenesis but also in the artery thromboses, the use of platelet antiaggregant drugs is very relevant in the primary and secondary prevention of the artery thrombosis. In this review, after a brief description of the primary hemostasis, aspects of antiplatelet like mechanisms of action and use asocied risks during the last 10 years are adressed. Primarily literature source was www.pubmed.com. The terms used in the search were: platelet antiaggregant, aspirin, dipyridamole, clopidogrel, abciximab, among others, then we searched the full texts of articles of interest and also used some hematology books.
Key words: platelet antiaggregants, cardiovascular diseases.
INTRODUCCIÓN
]]> Los antiagregantes plaquetarios son utilizados en la prevención secundaria de las enfermedades cardiovasculares (ECV) (1). En esta revisión, después de una breve descripción de la hemostasia primaria, se abordan diversos aspectos relativos a los antiagregantes, fundamentalmente su mecanismo de acción y sus efectos adversos. Como fuentes bibliográficas se utilizó principalmente www.pubmed.com; los términos utilizados en la búsqueda fueron: antiagregantes plaquetarios, aspirina, dipiridamol, clopidogrel, abciximab, entre otros; luego se buscaron los textos completos de los artículos que interesaban y además se utilizaron algunos libros de hematología.Hemostasia primaria
La hemostasia es un proceso complejo que previene la pérdida espontánea de sangre y detiene la hemorragia. Clásicamente se ha clasificado este proceso en hemostasia primaria, coagulación y fibrinólisis.
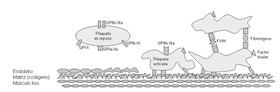
En la hemostasia primaria las plaquetas cumplen un rol fundamental. Éstas son elementos celulares anucleados de 1.5-3 μm de diámetro que se originan por fragmentación del citoplasma de los megacariocitos (2-4). El proceso en el que participan se puede separar en adhesión, secreción y agregación plaquetaria (Véase figura 1). Además de participar en la hemostasia primaria, las plaquetas activadas presentan actividad procoagulante al exponer fosfolípidos aniónicos (5), y sintetizar y expresar factor tisular (6). En los últimos años se ha demostrado que las plaquetas no sólo participan en las complicaciones trombóticas de la aterosclerosis, sino también en la iniciación y progresión de la placa ateromatosa (7). En este sentido, las plaquetas representan el puente de unión entre inflamación y trombosis, procesos fundamentales en el desarrollo de la aterotrombosis.
Figura 1. Representación esquemática de la participación de las plaquetas en la hemostasia primaria. Se muestran las principales moléculas de las plaquetas, de la matriz subendotelial y plasma que participan en los procesos de adhesión, secreción y agregación plaquetaria. (Adaptado de Aldunate y Vial) (18).
Enzimas plaquetarias
La activación plaquetaria es un proceso modulado dinámicamente por señales activantes e inhibidoras a las que se encuentra expuesta la superficie de la célula (8).
]]> Las plaquetas poseen diversos mecanismos y estrategias por los que los estímulos extracelulares se transmiten al interior de la célula, lo que resulta en una respuesta funcional apropiada. Esto se lleva a cabo con la participación de mecanismos complejos y altamente coordinados de transmisión de señales que incluyen: receptores, activación de proteínas que unen nucleótidos de guanina (proteínas G), metabolismo del fosfatidílinositol, metabolismo del ácido araquidónico, movimientos de calcio, reorganización del citoesqueleto y diversos procesos de fosforilación de proteínas (9).Entre otras enzimas participan: (a) fosfolipasa C, que desdobla el fosfatidilinositol 4,5 difosfato (PIP2) en diacilglicerol (DAG) e inositol trifosfato (IP3); (b) fosfolipasa A2, que libera ácido araquidónico (AA) de los fosfolípidos de la membrana plaquetaria; (c) ciclooxigenasa (COX-1), que metaboliza el AA a endoperóxidos cíclicos PGG2/PGH2; (d) tromboxano sintetasa, que transforma los endoperóxidos en tromboxano A 2 (TXA2); (e) adenilciclasa, que genera adenosin monofosfato (AMP) cíclico (cAMP) a partir de adenosin trifosfato (ATP); (f) fosfodiesterasa, que transforma cAMP en AMP; y (g) cinasas (proteína cinasa C, MAP cinasas, tirosina cinasas y cinasas lipídicas) que participan en los procesos de transducción de señales en el citosol plaquetario (5, 10).
Glicoproteínas plaquetarias
Muchos de los procesos en que participan las plaquetas son mediados por las glicoproteínas (GP) de membrana (5, 8). Las glicoproteínas GPIa-IIa, GPIV y GPVI, participan en la adhesión plaquetaria al colágeno de la matriz subendotelial (5, 11, 12). El complejo GPIb-IX-V (5, 13) actúa como receptor del Factor von Willebrand (FVW), interacción esencial en el fenómeno de adhesión de las plaquetas a la pared del vaso dañado y también juega un papel importante en la activación de las plaquetas por la trombina. El complejo GPIIb-IIIa (5, 14) es receptor para el fibrinógeno, FVW, fibronectina y vitronectina. Una vez activadas las plaquetas, la GPIIb-IIIa sufre un cambio conformacional que permite la exposición de un dominio que reconoce la secuencia RGD (Arg-Gly-Asp) presente en fibrinógeno lo que permite la agregación plaquetaria.
Receptores no glicoproteicos. Desde un punto de vista funcional, a los receptores no glicoproteicos se les puede clasificar en receptores activadores (receptores de ADP: P2Y1 y P2Y12, receptores activados por proteasas, trombina: PAR1 y PAR4, y receptor de TXA2) y receptores inhibidores (receptores para prostaglandinas I2 o prostaciclina, E1 y D2, y adenosina (5, 15).
ANTIAGREGANTES PLAQUETARIOS
El uso más frecuente de los fármacos antiplaquetarios es la prevención primaria o secundaria de la trombosis arterial, preferentemente el infarto agudo de miocardio (IAM), en el que las plaquetas, ante la ruptura o erosión de las placas ateroscleróticas en las arterias coronarias, se agregan formando un trombo que obstruye la circulación sanguínea. Un balance favorable entre los efectos benéficos y las complicaciones de la terapia antiplaquetaria se alcanza al tratar pacientes en los cuales el riesgo trombótico supera los riesgos de complicaciones hemorrágicas (16, 17). A continuación se mencionan los principales fármacos antiplaquetarios clasificados en dos grupos según su mecanismo de acción: inhibidores de enzimas plaquetarias e inhibidores de receptores plaquetarios (Véanse tabla 1 y figura 2).
]]> Tabla 1. Clasificacion de los principales antiagregantes plaquetarios según mecanismo de acción.
Figura 2. Representación esquemática de los mecanismos de acción de los fármacos antiplaquetarios en uso. Se muestran las enzimas (ciclooxigenasa y fosfodiesterasa) y receptores (P2Y1, P2Y12; GPIIb-IIIa; PAR1, PAR4) inhibidos por los antiagregantes plaquetarios. Adaptado de Santos et al (5).
Inhibidores de enzimas plaquetarias
Inhibidores de ciclooxigenasa
Aspirina (Véase figura 3). La aspirina (ácido acetil salicílico) actúa inhibiendo la acción de la ciclooxigenasa (COX-1 y COX-2) (Véase figura 2). Al inhibir la COX-1 disminuye la síntesis de TXA2 y, como consecuencia, la agregación plaquetaria. Por su parte, la inhibición de la COX-2, con su efecto anti-inflamatorio, disminuye la inflamación vascular en el sitio de la placa ateromatosa y eso, a su vez, reduce la infiltración de células mononucleares en la placa ateromatosa (15, 18). La terapia con aspirina ha sido reconocida como una herramienta efectiva en el manejo de pacientes con angina inestable. Estudios han mostrado un 40% en la reducción de IAM fatales y no fatales (17, 19). Los efectos adversos más comunes de la aspirina son los trastornos gastrointestinales (náuseas, ardor gastrointestinal, dolor epigástrico, etc), que generalmente son dosis-dependiente (20). Además puede provocar hemorragias, siendo la más frecuente a nivel digestivo alto (21). Por otra parte, a nivel de la mucosa gástrica inhibe la síntesis de prostaglandinas, moléculas que disminuyen la secreción ácida (20). La aspirina se ha convertido en el antiplaquetario de referencia y su tratamiento debe iniciarse tan pronto como sea posible después de la aparición de un evento agudo y continuarse indefinidamente, salvo que esté contraindicado por alergia, complicaciones gastrointestinales o hemorragia (22). La aspirina debe ser usada con cautela en pacientes con antecedentes de úlcera péptica, gastritis y alteraciones de la hemostasia. También debe limitarse su uso en enfermos con trastornos hepáticos o renales y en estados de hipovolemia, pues se incrementa el riesgo de manifestaciones tóxicas, así como su interacción con otros medicamentos que se unen a proteínas (ej. tiroxina, penicilina sódica, fenitoína), ya que la dosis requerida de estos fármacos se ve modificada (18). La respuesta a la aspirina presenta variabilidad interindividual, lo que se explica entre otras razones por las siguientes: hiperlipidemia, hiperglicemia, tabaquismo, ejercicio, síndromes coronarios agudos (SCA), falla cardíaca, absorción, otros medicamentos y polimorfismos (23, 24). Por “resistencia a la aspirina” se ha entendido que ésta no es capaz de: (a) proteger a los individuos de las complicaciones trombóticas, (b) causar una prolongación del tiempo de sangría, (c) reducir la producción de TXA2 o (d) producir efecto antiplaquetario en uno o más ensayos de función plaquetaria in vitro (25-28). Algunos autores han llegado a establecer un muy elevado porcentaje de “resistencia a la aspirina”, siendo ésta más frecuente en mujeres y ancianos (29).
]]> Figura 3. Estructura de inhibidores de enzimas plaquetarias. De ciclooxigenasa (Aspirina, Trifusal y Nitro-aspirina) y fosfodiesterasa (Dipiridamol y Cilostazol). Adaptado de referencias 16, 33, 34 y 41.
Triflusal (Trifluorosalicílico) (30). Presenta similitudes estructurales con los salicilatos (Véase figura 3) (31, 32), pero no es derivado de la aspirina. Trifusal bloquea irreversiblemente la COX-1 (Véase figura 2) e inhibe sólo ligeramente la COX-2, de manera que la síntesis de prostaciclina sólo se reduce levemente (33-35). Además, tanto triflusal como su metabolito activo, el ácido 2-hidroxi-4-trifluorometilbenzoico (HTB), inhiben la degradación de cAMP plaquetario y endotelial disminuyendo la activación plaquetaria e interacción con células endoteliales (36-38).
Triflusal tiene un rol en la prevención primaria de eventos cerebrovasculares, en fibrilación atrial y en la prevención secundaria de infartos cerebrales e IAM, principalmente como alternativa a la aspirina (34). Entre las complicaciones del triflusal se ha informado que tiene efectos fotoalérgicos (39).
Molécula dual en estudio. NCX-4016 (Nitro-aspirina) es un compuesto dual que consiste en una molécula de aspirina unida covalentemente con -ONO2. Su fórmula química es 2-(acetiloxi) ácido benzoico 3-[(nitrooxi)metil]fenil ester. Corresponde a una molécula de aspirina capaz de donar óxido nítrico (NO) (40). Entre sus ventajas se incluyen la combinación de todos los beneficios asociados a la administración de aspirina con las propiedades cardioprotectoras del NO (antitrombóticas, antiaterogénicas y vasodilatadoras) (41). Estudios en animales de experimentación han mostrado que NCX-4016 previene la reestenosis (42).
Inhibidor de fosfodiesterasa
Dipiridamol. Este medicamento (Véase figura 3) inhibe la enzima fosfodiesterasa que inactiva al cAMP, aumentando así la concentración intracitoplasmática de este último (Véase figura 2). El incremento intraplaquetario de cAMP reduce la activación de segundos mensajeros citoplasmáticos. El dipiridamol también estimula la liberación de prostaciclina e inhibe la formación de TXA2 (16, 18, 20). La cefalea es el efecto secundario más común asociado al tratamiento con dipiridamol (25, 43).
Cilostazol. Esta molécula (Véase figura 3) es un inhibidor oral selectivo de nucleótidos cíclicos de la fosfodiesterasa 3 (PDE3) (Véase figura 2), con efectos antiplaquetario, vasodilatador y antimitogénico (44-47). Cilostazol inhibe selectivamente la isoenzima PDE3 y a PDE3A (subtipo cardiovascular), a través de un mecanismo cAMP competitivo (48). La inhibición de la función plaquetaria por cilostazol parece obedecer a la disminución de los niveles de calcio intracelular, resultantes del incremento de los niveles de cAMP (45, 48); además se sugiere que el cilostazol inhibe la agregación plaquetaria suprimiendo la activación de la GPIIb-IIIa (49). Según lo reportado en el estudio CSPS (Cilostazol Stroke Prevention Study) los efectos adversos con este fármaco son infrecuentes, e incluyen cefalea, palpitaciones y diarrea, y está contraindicado en pacientes con falla cardíaca congestiva, (50, 51).
Inhibidores de receptores plaquetarios
Inhibidores de receptores de ADP
]]> Las tienopiridinas (ticlopidina, clopidogrel y prasugrel) son profármacos que una vez metabolizados se transforman en fármacos activos y como tales inhiben los receptores de ADP (P2Y1, P2Y12) (Véase figura 2).Ticlopidina. Este fármaco (Véase figura 4) representa una alternativa a la aspirina en aquellos pacientes con resistencia a esta última (15, 16, 18). La ticlopidina fue el primer agente antiplaquetario de la familia de las tienopiridinas que se usó. Dos importantes ensayos clínicos, CATS (Canadian American Ticlopidine Study) (52) y TASS (Aspirin Stroke Study) (53), demostraron la eficacia de la ticlopidina en la reducción de eventos trombóticos en pacientes con enfermedad aterosclerótica. Sin embargo, presenta un inicio tardío de su efecto, lo cual ha limitado su uso en SCA. Su efecto adverso más importante es la neutropenia marcada, la que generalmente desaparece una vez suspendido el medicamento (25, 54). Además, con baja frecuencia la ticlopidina ha sido asociada a trombocitopenia grave pero reversible, anemia aplásica, hipercolesterolemia y púrpura trombótico trombocitopénica (PTT) (20, 25, 43).
Figura 4. Estructura de inhibidores de receptores plaquetarios. De P2Y1, P2Y12 (Ticlopidina, Clopidogrel y Prasugrel) y de GPIIb-IIIa (Eptifibatide, Tirofiban). Adaptado de referencias 34 y 73.
Clopidogrel. Este medicamento (Véase figura 4) está relacionado química y farmacológicamente con la ticlopidina, pero presenta un perfil de efectos secundarios más favorable (15-17, 55), motivo por el que la ha reemplazado. En el estudio CAPRIE (Clopidogrel Versus Aspirin in Patients at Risk of Ischemic Events), la presentación de rash marcado fue más frecuente con clopidogrel que con aspirina (0.26% vs 0.1%) (56). Basándose en la agregación plaquetaria inducida por ADP, algunos pacientes han sido considerados “no respondedores o resistentes” al clopidogrel (25). Incluso se ha identificado un número no despreciable de estos pacientes que presentan un incremento en la incidencia de trombosis subaguda tras la implantación de un stent (57). Se ha sugerido que el tratamiento concurrente de estatinas que son sustratos del citocromo P450 3A4 (CYP3A4) (ej, atorvastatina y simvastatina), puede interferir con el efecto antiagregante del clopidogrel modificando su biodisponibilidad sistémica y su eficacia clínica (25). Finalmente, además de sus propiedades antiagregantes, se ha demostrado que clopidogrel reduce la formación de conjugados trombocito-leucocito en pacientes con SCA (58) y reduce la expresión de marcadores inflamatorios en plaquetas activadas como CD40L y P-selectina en pacientes sometidos a intervención coronaria percutánea (59).
Prasugrel (CS-747, LY640315). Este fármaco (Véase figura 4), también es un profármaco de tipo tienopiridina (60, 61); es activo por vía oral (62) y posee un espectro de actividades biológicas similares a las que presenta clopidogrel (62). La estructura química de prasugrel le permite una eficiente conversión a su metabolito activo con dependencia menos rigurosa de las enzimas específicas de la vía del citocromo P-450 a nivel hepático, donde es metabolizado (33), ya que in vivo se convierte rápidamente a un metabolito activo (R-138727) que se une específicamente y de forma irreversible al receptor P2Y12 (60). Estas ventajas farmacológicas probablemente se explican por una mejor farmacocinética, es decir, una más alta tasa de conversión del profármaco en el metabolito activo (62). Debido a que las tienopiridinas no inhiben el metabolismo del ácido araquidónico, permite una interacción sinérgica con la aspirina (60, 63-65).
Antagonistas de P2Y12 en estudio. Algunos compuestos antiplaquetarios que inhiben en forma reversible P2Y12 y que se encuentran en estudios de fase I-III, son los siguientes: (a) AZD6140 (AstraZeneca) es un análogo de ATP que no requiere conversión a un metabolito activo y actúa como antagonista directo de P2Y12, (33, 66, 67), (b) Cangrelor (AR-C69931MX) (68, 69) y (c) PRT060128 (Portola) (70).
Inhibidores de GPIIb-IIIa
]]> Varios antiagregantes actuán como bloqueadores de la GPIIb-IIIa, algunos de uso parenteral: Eptifibatide, Tirofiban y Abciximab (Véase figura 2).Eptifibatide. Este fármaco es un péptido cíclico (Véase figura 4) que se une reversiblemente al complejo GPIIb-IIIa (20). Tanto en el estudio PURSUIT (Platelet Glycoprotein IIb/IIIa in Unstable Angina: Receptor Suppression Using Integrilin Therapy), como en el ESPRIT (Enhanced Supresión of the Platelet IIb-IIIa Receptor with Integrilin Therapy), pacientes tratados con eptifibatide, mostraron un incremento en las complicaciones hemorrágicas (71, 72) en algunos casos asociada a trombocitopenia, posiblemente inmunomediada (25).
Tirofiban. Este medicamento corresponde a una molécula pequeña no peptídica (Véase figura 4) que también se une reversiblemente al complejo GPIIb-IIIa (20). En un pequeño porcentaje de pacientes se ha reportado trombocitopenia marcada, probablemente de tipo inmune (18, 25, 43). En los pacientes con insuficiencia renal, el clearance plasmático del tirofiban se encuentra disminuido y la vida media plasmática aumentada (72).
Abciximab. Este fármaco es un anticuerpo monoclonal con especificidad anti-GPIIb-IIIa (17, 20). Su principal complicación es la presentación de eventos hemorrágicos (73). En el proyecto GUSTO IV ACS (Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries), abciximab respecto a placebo se asoció con un incremento de eventos hemorrágicos (73). Aproximadamente el 1-2% de los pacientes tratados con abciximab desarrolla trombocitopenia, al parecer inmunomediada y reversible si se suspende la administración del fármaco (25). Entre los efectos secundarios de este fármaco, además del riesgo hemorrágico por el gran efecto inhibidor de la agregación plaquetaria, destaca su capacidad inmunogénica, a pesar de ser inferior que la descrita con los anticuerpos murinos, y la trombocitopenia (1,6-5%) (74).
Otros anti-GPIIb-IIIa. Varios fármacos antagonistas de la GPIIb-IIIa activos por vía oral han sido descritos: Xemilofiban, Sibrafiban, Orbofiban y Lotrafiban. Sin embargo, los resultados clínicos no han sido alentadores (75).
Antagonistas de receptores PAR
E5555 y SCH530348. Los receptores PAR1 y PAR4 de las plaquetas pueden unir trombina (Véase figura 2) (76), serino proteasa que produce liberación del segmento aminoterminal de los receptores PAR y genera nuevas secuencias aminoacídicas terminales, SFLLRN para PAR1 y GYPGKF para PAR4; éstas autoactivan otras regiones de los receptores PAR e inician la activación plaquetaria vía proteínas G (77). Se han evaluado en estudios clínicos dos antagonistas orales de PAR1: E5555 y SCH530348. La eficacia y la seguridad de E5555 están siendo evaluadas en pacientes con enfermedad arterial coronaria (41).
Pepducinas. Son péptidos basados en la secuencia intracelular de los PAR que interacciona con las proteínas G acopladas. Actúan intracelularmente bloqueando la interacción entre las proteínas G y los PAR. Se ha observado que P1 pal-12 inhibe la activación plaquetaria mediada por PAR1 (78) y P4 pal10 inhibe la activación mediada por PAR4 (79, 80).
Aprotinina. Si bien este medicamento no es un inhibidor de PAR, indirectamente está relacionado con PAR-1 al inhibir serino proteasas (ej. trombina) y así afectar la función plaquetaria. La aprotinina es usada durante las cirugías cardíacas (81-83).
Inhibidor del receptor de tromboxano
]]> S18886. Esta molécula es un inhibidor reversible del receptor del tromboxano (TXA2R) (84) y por ello no sólo inhibe la acción del TXA2, sino también la activación del TXA2R derivada de otros eicosanoides no inhibidos por la aspirina (HETE e isoprostanos) (85). Se ha demostrado la rápida y eficaz actividad de S18886 en inhibir la trombosis inducida por stent sin la concomitante aparición de un mayor riesgo de sangrado (86). También se ha demostrado que S18886 previene la aterogénesis, causa regresión de la placa aterosclerótica y mejora la función endotelial en pacientes con enfermedad arterial coronaria, propiedades relativamente independientes de los efectos derivados del TXA2 en las plaquetas (87, 88).
ANTIPLAQUETARIOS DESDE UNA PERSPECTIVA CLÍNICA
A continuación se comentan algunos aspectos sobre los antiagregantes plaquetarios en uso, desde un punto de vista clínico.
Aspirina. Administrada una vez al día, está recomendada en todas las condiciones clínicas en las que la profilaxis antiplaquetaria tiene un perfil beneficio/riesgo favorable. La evidencia disponible apoya el uso de dosis diarias de aspirina en el rango de 75-100 mg para la prevención a largo plazo de episodios vasculares graves en pacientes de alto riesgo (es decir, de un 3% o más por año). En las situaciones clínicas en que se requiera un efecto antitrombótico inmediato, como en los SCA o en los accidentes cerebrovasculares isquémicos agudos, debe administrarse una dosis inicial de 160-300 mg en el momento del diagnóstico para asegurar una inhibición completa y rápida de la agregación plaquetaria dependiente del TXA2 (89).
Ticlopidina. El papel de la ticlopidina en el repertorio terapéutico actual es incierto. Aunque no hay comparaciones directas suficientemente grandes entre las dos tienopiridinas, las comparaciones indirectas sugieren que el clopidogrel se asocia a una menor carga de toxicidad grave de la médula ósea. Además, a diferencia del clopidogrel, la ticlopidina no tiene una indicación aprobada para el caso de pacientes con IAM reciente (43, 90).
Clopidogrel. El clopidogrel, a una dosis diaria de 75 mg, es una alternativa adecuada en pacientes de alto riesgo con enfermedad coronaria, enfermedad cerebrovascular o enfermedad arterial periférica que tengan alguna contraindicación para tomar aspirina a bajas dosis.
Los resultados del estudio clínico CURE (Clopidogrel in Instable Angina to Prevent Recurrente Events) han llevado a la aprobación de una nueva indicación para el clopidogrel en pacientes con un SCA sin elevación del segmento ST. En este contexto clínico, se recomienda usar una dosis inicial de clopidogrel de 300 mg, seguida de una dosis diaria de 75 mg. La revisión de las guías de práctica clínica existentes requerirá un acuerdo por consenso por parte de los expertos respecto al momento de realizar la intervención coronaria percutánea, la duración del tratamiento con clopidogrel y la combinación con antagonistas de los receptores de la GPIIb/IIIa (91).
Abciximab, Eptifibatide y Tirofiban. El perfil beneficio/riesgo de estos fármacos es muy incierto para aquellos pacientes que cursan con un SCA, que no han sido programados rutinariamente para recibir una revascularización temprana (73). Por el contrario, en el caso de pacientes que se sometan a una intervención coronaria percutánea, la intensificación de la terapia antiplaquetaria añadiendo un antagonista de los receptores de la GPIIb/IIIa por vía intravenosa es una estrategia adecuada para reducir el riesgo de las complicaciones trombóticas asociadas al procedimiento (92-94).
]]> COMENTARIO FINAL
Los antiagregantes en uso han sido útiles en la prevención de eventos cardiovasculares primarios en la población de alto riesgo y eventos secundarios en individuos que ya han presentado uno
o más eventos cardiovasculares trombóticos. Sin embargo, presentan algunos efectos adversos, entre otros hemorragia, hipersensibilidad y trastornos gastrointestinales, y además algún grado de resistencia a su efecto. Dada la frecuencia con las que las ECV se presentan en el mundo desarrollado y en desarrollo, es necesaria la búsqueda de nuevos antiplaquetarios, los que podrán tener como blanco proteínas sobre las que ya otros antiplaquetarios actuan y otras proteínas que aún no son targets de este tipo de fármacos. Entre los blancos seguramente estarán enzimas y proteínas citoplasmáticas, como también glicoproteínas plaquetarias específicas y otros receptores de membrana. Los nuevos antiplaquetarios deben ser más efectivos a menos dosis para disminuir la toxicidad. Respecto a la aspirina un par de comentarios finales: (a) a pesar de sus beneficios aún existe un importante grupo de paciente que no la reciben; al respecto alguien dijo “en prevención secundaria cardiovascular, se debería justificar por qué no dar aspirina en vez de pensar si el paciente la necesita o no” y (b) a pesar de sus efectos adversos y resistencia descrita, considerando sus ventajas y bajo costo comparado con otros antiagregantes plaquetarios, seguramente pasará mucho tiempo antes que otro fármaco la pueda reeemplazar.
REFERENCIAS BIBLIOGRÁFICAS
1. Tendera M, Wojakowski W. Role of antiplatelet drugs in the prevention of cardiovascular events. Thromb Res. 2003; 110 (5-6): 355-359. [ Links ]
2. Schulze H, Shivdasani RA. Mechanisms of thrombopoiesis. J Thromb Haemost. 2005; 3 (8): 1717-1724. [ Links ]
3. Italiano J. The structure and production of blood platelets. En: Gresele P, Fuster V, López J, et al., editores. Platelets in hematologic and cardiovascular disorders- A clinical handbook. New York: Cambridge University Press; 2008. p. 1-20. [ Links ]
4. Italiano J, Hartwig J. Megakaryocyte development and platelet formation. En: Michelson AD, editor. Platelets. EEUU: Elsevier; 2007. p. 1327. [ Links ]
5. Santos MT, Aranda E, Vallés J, Palomo I. Hemostasia primaria. En: Palomo I, Pereira J, Palma J, editores. Hematología: fisiopatología y diagnóstico. Talca: Universidad de Talca; 2005 [ Links ]
6. Panes O, Matus V, Saez CG, Quiroga T, Pereira J, Mezzano D. Human platelets synthesize and express functional tissue factor. Blood. 2007; 109 (12): 5242-5250. [ Links ]
7. Siegel-Axel D, Langer H, Lindemann S, Gawaz M. Role of platelets in atherosclerosis and inflammation. Med Klin. 2006; 101 (6): 467-475. [ Links ]
8. White J. Platelet structure. En: Platelest. Michelson AD, editor. Platelets. 2007. California Elsevier Science (USA); 2007. [ Links ]
9. Stalker T. Mechanisms of platelet activation. En: Gresele P, Fuster V, López J, Page C, Vermylen J, et al, editores. Platelets in hematologic and cardiovascular disorders. A clinical handbook. New York: Cambridge University Press; 2008. [ Links ]
10. Brass L, Stalker T. Mechanisms of platelet activation. En: Gresele P, Fuster V, López J, Page C, Vermylen J, et al, editores. Platelets in hematologic and cardiovascular disorders- A clinical handbook. New York: Cambridge University Press; 2008. [ Links ]
11. Moroi M, Jung SM. Integrin-mediated platelet adhesion. Front Biosci. 1998; 3: d719-728. [ Links ]
12. Moroi M, Jung SM. Platelet glycoprotein VI: its structure and function. Thromb Res. 2004; 114 (4): 221-233. [ Links ]
13. Rivera J, Lozano ML, Corral J, González-Conejero R, Martínez C, Vicente V. Platelet GP Ib/IX/V complex: physiological role. J Physiol Biochem. 2000; 56 (4): 355-365. [ Links ]
14. Bennett JS. Structure and function of the platelet integrin alphaIIbbeta3. J Clin Invest. 2005; 115 (12): 3363-3369. [ Links ]
15. Rezkalla SH, Benz M. Antiplatelet therapy from clinical trials to clinical practice. Clin Med Res. 2003; 1 (2): 101-104. [ Links ]
16. Born G, Patrono C. Antiplatelet drugs. Br J Pharmacol. 2006; 147 Suppl 1: S241-251. [ Links ]
17. Palomo I, Torres C, Moore-Carrasco R, Alarcón M. Mecanimos de acción de los principales antiagregantes plaquetarios. Rev Med Maule. 2008; 26 (1): 50-55. [ Links ]
18. Aldunate J, Vial M. Tratamiento antitrombótico. En: Palomo I, Pereira J, Palma J, editores. Hematología: fisiopatología y diagnóstico. Talca: Universidad de Talca; 2005. [ Links ]
19. Abrams J. Medical therapy of unstable angina and non-Q-wave myocardial infarction. Am J Cardiol. 2000; 86 (8B): 24J-33J; discussion 33J-34J. [ Links ]
20. HankeyGJ,EikelboomJW.Antiplateletdrugs.MedJAust.2003; 178 (11): 568-574. [ Links ]
21. Harrington RA, Becker RC, Ezekowitz M, Meade TW, O"Connor CM, Vorchheimer DA, et al. Antithrombotic therapy for coronary artery disease: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004; 126 (3 Suppl): 513S-548S. [ Links ]
22. Antman EM, Anbe DT, Armstrong PW, Bates ER, Green LA, Hand M, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1999 Guidelines for the Management of Patients with Acute Myocardial Infarction). Circulation 2004; 110 (9): e82-292. [ Links ]
23. Gladding P, Webster M, Ormiston J, Olsen S, White H. Antiplatelet drug nonresponsiveness. Am Heart J. 2008; 155 (4): 591-599. [ Links ]
24. Coma-Canella I, Velasc A. Variability in individual responsiveness to aspirin: clinical implications and treatment. Cardiovasc Hematol Disord Drug Targets. 2007; 7(4): 274-287. [ Links ]
25. Patrono C, Coller B, FitzGerald GA, Hirsh J, Roth G. Platelet-active drugs: the relationships among dose, effectiveness, and side effects: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004; 126 (3 Suppl): 234S-264S. [ Links ]
26. Dussaillant G, Zapata M, Fardella P, Conte G, Cuneo M. Frecuencia y características de la resistencia a aspirina en pacientes cardiovasculares chilenos. Rev Med Chile. 2005; 133 (4): 409-417. [ Links ]
27. Buchanan MR, Brister SJ. Individual variation in the effects of ASA on platelet function: implications for the use of ASA clinically. Can J Cardiol. 1995; 11 (3): 221-227. [ Links ]
28. Berglund U, Wallentin L. Persistent inhibition of platelet function during long-term treatment with 75 mg acetylsalicylic acid daily in men with unstable coronary artery disease. Eur Heart J. 1991; 12 (3): 428-433. [ Links ]
29. Hankey GJ, Eikelboom JW. Aspirin resistance. BMJ. 2004; 328 (7438): 477-479. [ Links ]
30. McNeely W, Goa KL. Triflusal. Drugs. 1998; 55 (6): 823-833; discussion 834-835. [ Links ]
31. Falco A, Salvati F, Vitacolonna E, Avellone G, Pinto A, Di Febbo C, et al. Inhibition of thromboxane biosynthesis by triflusal in type 2 diabetes mellitus. Atherosclerosis. 2005; 183 (2): 329-335. [ Links ]
32. Fernández de Arriba A, Cavalcanti F, Miralles A, Bayon Y, Alonso A, Merlos M, et al. Inhibition of cyclooxygenase-2 expression by 4-trifluoromethyl derivatives of salicylate, triflusal, and its deacetylated metabolite, 2-hydroxy-4-trifluoromethylbenzoic acid. Mol Pharmacol. 1999; 55 (4): 753-760. [ Links ]
33. Michelson AD. P2Y12 antagonism: promises and challenges. Arterioscler Thromb Vasc Biol. 2008; 28 (3): s33-38. [ Links ]
34. Murdoch D, Plosker GL. Triflusal: a review of its use in cerebral infarction and myocardial infarction, and as thromboprophylaxis in atrial fibrillation. Drugs. 2006; 66 (5): 671-692. [ Links ]
35. Cruz-Fernández JM, López-Bescos L, García-Dorado D, López García-Aranda V, Cabades A, Martin-Jadraque L, et al. Randomized comparative trial of triflusal and aspirin following acute myocardial infarction. Eur Heart J. 2000; 21 (6): 457-465. [ Links ]
36. De la Cruz JP, Villalobos MA, García PJ, Smith-Agreda JM, Sánchez de la Cuesta F. Effects of triflusal and its main metabolite HTB on platelet interaction with subendothelium in healthy volunteers. Eur J Clin Pharmacol. 1995; 47 (6): 497-502. [ Links ]
37. Sánchez de Miguel L, Casado S, Farre J, García-Durán M, Rico LA, Monton M, et al. Comparison of in vitro effects of triflusal and acetysalicylic acid on nitric oxide synthesis by human neutrophils. Eur J Pharmacol. 1998; 343 (1): 57-65. [ Links ]
38. Sánchez de Miguel L, Monton M, Mar Arriero M, Guerra JI, Jiménez A, González-Fernández F, et al. The effect of triflusal on human platelet aggregation and secretion: the role of nitric oxide. Rev Esp Cardiol. 2000; 53 (2): 205-211. [ Links ]
39. Nagore E, Pérez-Ferriols A, Sánchez-Motilla JM, Serrano G, Aliaga A. Photosensitivity associated with treatment with triflusal. J Eur Acad Dermatol Venereol. 2000; 14 (3): 219-221. [ Links ]
40. GreseleP,MomiS.Pharmacologicprofileandtherapeuticpotential of NCX 4016, a nitric oxide-releasing aspirin, for cardiovascular disorders. Cardiovasc Drug Rev. 2006; 24 (2): 148-168. [ Links ]
41. Badimon L, Vilahur G. Coronary atherothrombotic disease: progress in antiplatelet therapy. Rev Esp Cardiol. 2008; 61 (5): 501-513. [ Links ]
42. Napoli C, Aldini G, Wallace JL, de Nigris F, Maffei R, Abete P, et al. Efficacy and age-related effects of nitric oxide-releasing aspirin on experimental restenosis. Proc Natl Acad Sci USA. 2002; 99 (3): 1689-1694. [ Links ]
43. Patrono C, Coller B, Dalen JE, FitzGerald GA, Fuster V, Gent M, et al. Platelet-active drugs: the relationships among dose, effectiveness, and side effects. Chest. 2001; 119 (1 Suppl): 39S-63S. [ Links ]
44. Inoue T, Sohma R, Morooka S. Cilostazol inhibits the expression of activation-dependent membrane surface glycoprotein on the surface of platelets stimulated in vitro. Thromb Res. 1999; 93 (3): 137-143. [ Links ]
45. Ito H, Miyakoda G, Mori T. Cilostazol inhibits platelet-leukocyte interaction by suppression of platelet activation. Platelets. 2004; 15 (5): 293-301. [ Links ]
46. Kariyazono H, Nakamura K, Shinkawa T, Yamaguchi T, Sakata R, Yamada K. Inhibition of platelet aggregation and the release of P-selectin from platelets by cilostazol. Thromb Res. 2001; 101 (6): 445-453. [ Links ]
47. Yamazaki M, Uchiyama S, Xiong Y, Nakano T, Nakamura T, Iwata M. Effect of remnant-like particle on shear-induced platelet activation and its inhibition by antiplatelet agents. Thromb Res. 2005; 115 (3): 211-218. [ Links ]
48. Sudo T, Tachibana K, Toga K, Tochizawa S, Inoue Y, Kimura Y, et al. Potent effects of novel anti-platelet aggregatory cilostamide analogues on recombinant cyclic nucleotide phosphodiesterase isozyme activity. Biochem Pharmacol. 2000; 59 (4): 347-356. [ Links ]
49. Ikeda Y, Toshiki S, Kimura Y. Cilostazol. En: Michelson AD, editor. Platelets. EEUU: Elsevier; 2007. p. 1327 [ Links ]
50. Gotoh F, Tohgi H, Hirai S, Terashi A, Fukuuchi Y, Otomo E, et al. Cilostazol stroke prevention study: A placebo-controlled double-blind trial for secondary prevention of cerebral infarction. J Stroke Cerebrovasc Dis. 2000; 9 (4): 147-157. [ Links ]
51. Gotoh F, Ohashi Y. Design and organization of the cilostazol stroke prevention study. J Stroke Cerebrovasc Dis. 2000; 9 (1): 36-44. [ Links ]
52. Gent M, Blakely JA, Easton JD, Ellis DJ, Hachinski VC, Harbison JW, et al. The Canadian American Ticlopidine Study (CATS) in thromboembolic stroke. Lancet. 1989; 1 (8649): 1215-1220. [ Links ]
53. Hass WK, Easton JD, Adams HP Jr., Pryse-Phillips W, Molony BA, Anderson S, et al. A randomized trial comparing ticlopidine hydrochloride with aspirin for the prevention of stroke in high-risk patients. Ticlopidine Aspirin Stroke Study Group. N Engl J Med. 1989; 321 (8): 501-507. [ Links ]
54. Hirsh J, Dalen JE, Fuster V, Harker LB, Patrono C, Roth G. Aspirin and other platelet-active drugs. The relationship among dose, effectiveness, and side effects. Chest. 1995; 108 (4 Suppl): 247S-257S. [ Links ]
55. Savi P, Herbert JM. Clopidogrel and ticlopidine: P2Y12 adenosine diphosphate-receptor antagonists for the prevention of atherothrombosis. Semin Thromb Hemost. 2005; 31 (2): 174-183. [ Links ]
56. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). CAPRIE Steering Committee. Lancet. 1996; 348 (9038): 1329-1339. [ Links ]
57. Gurbel PA, Bliden KP, Hiatt BL, O"Connor CM. Clopidogrel for coronary stenting: response variability, drug resistance, and the effect of pretreatment platelet reactivity. Circulation. 2003; 107 (23): 2908-2913. [ Links ]
58. Xiao Z, Theroux P. Clopidogrel inhibits platelet-leukocyte interactions and thrombin receptor agonist peptide-induced platelet activation in patients with an acute coronary syndrome. J Am Coll Cardiol. 2004; 43 (11): 1982-1988. [ Links ]
59. Quinn MJ, Bhatt DL, Zidar F, Vivekananthan D, Chew DP, El-lis SG, et al. Effect of clopidogrel pretreatment on inflammatory marker expression in patients undergoing percutaneous coronary intervention. Am J Cardiol 2004; 93 (6): 679-684. [ Links ]
60. Jakubowski JA, Winters KJ, Naganuma H, Wallentin L. Prasugrel: a novel thienopyridine antiplatelet agent. A review of preclinical and clinical studies and the mechanistic basis for its distinct antiplatelet profile. Cardiovasc Drug Rev. 2007; 25 (4): 357-374. [ Links ]
61. Farid NA, Payne CD, Ernest CS, 2nd, Li YG, Winters KJ, Salazar DE, et al. Prasugrel, a new thienopyridine antiplatelet drug, weakly inhibits cytochrome P450 2B6 in humans. J Clin Pharmacol. 2008; 48 (1): 53-59. [ Links ]
62. Schror K, Huber K. Prasugrel, a new thienopyridine. Hamostaseologie. 2007; 27 (5): 351-355. [ Links ]
63. Wiviott SD, Antman EM, Winters KJ, Weerakkody G, Murphy SA, Behounek BD, et al. Randomized comparison of prasugrel (CS-747, LY640315), a novel thienopyridine P2Y12 antagonist, with clopidogrel in percutaneous coronary intervention: results of the Joint Utilization of Medications to Block Platelets Optimally (JUMBO)-TIMI 26 trial. Circulation. 2005; 111 (25): 3366-3373. [ Links ]
64. Jernberg T, Payne CD, Winters KJ, Darstein C, Brandt JT, Jakubowski JA, et al. Prasugrel achieves greater inhibition of platelet aggregation and a lower rate of non-responders compared with clopidogrel in aspirin-treated patients with stable coronary artery disease. Eur Heart J. 2006; 27 (10): 1166-1173. [ Links ]
65. Tantry US, Bliden KP, Gurbel PA. Prasugrel. Expert Opin Investig Drugs. 2006; 15 (12): 1627-1633. [ Links ]
66. van Giezen JJ, Humphries RG. Preclinical and clinical studies with selective reversible direct P2Y12 antagonists. Semin Thromb Hemost. 2005; 31 (2): 195-204. [ Links ]
67. Husted S, Emanuelsson H, Heptinstall S, Sandset PM, Wickens M, Peters G. Pharmacodynamics, pharmacokinetics, and safety of the oral reversible P2Y12 antagonist AZD6140 with aspirin in patients with atherosclerosis: a double-blind comparison to clopidogrel with aspirin. Eur Heart J. 2006; 27 (9): 1038-1047. [ Links ]
68. Greenbaum AB, Grines CL, Bittl JA, Becker RC, Kereiakes DJ, Gilchrist IC, et al. Initial experience with an intravenous P2Y12 platelet receptor antagonist in patients undergoing percutaneous coronary intervention: results from a 2-part, phase II, multicenter, randomized, placebo-and active-controlled trial. Am Heart J. 2006; 151 (3): 689 e681-689 e610. [ Links ]
69. Storey RF, Oldroyd KG, Wilcox RG. Open multicentre study of the P2T receptor antagonist AR-C69931MX assessing safety, tolerability and activity in patients with acute coronary syndromes. Thromb Haemost. 2001; 85 (3): 401-407. [ Links ]
70. Gretler DD, Conley PB, Andre P, Jurek M, Pandey A, Ronanko K, et al. "First in human" experience with PRT060128, a new direct-acting, reversible, P2Y12 inhibitor for IV and oral use. J Am Coll Cardiol. 2007; 49 (9 (supl 2)): 326A. [ Links ]
71. Inhibition of platelet glycoprotein IIb/IIIa with eptifibatide in patients with acute coronary syndromes. The PURSUIT Trial Investigators. Platelet glycoprotein IIb/IIIa in unstable angina: receptor suppression Using Integrilin therapy. N Engl J Med. 1998; 339 (7): 436-443. [ Links ]
72. Novel dosing regimen of eptifibatide in planned coronary stent implantation (ESPRIT): a randomised, placebo-controlled trial. Lancet. 2000; 356 (9247): 2037-2044. [ Links ]
73 Simoons ML. Effect of glycoprotein IIb/IIIa receptor blocker abciximab on outcome in patients with acute coronary syndromes without early coronary revascularisation: the GUSTO IV-ACS randomised trial. Lancet. 2001; 357 (9272): 1915-1924. [ Links ]
74. Dasgupta H, Blankenship JC, Wood GC, Frey CM, Demko SL, Menapace FJ. Thrombocytopenia complicating treatment with intravenous glycoprotein IIb/IIIa receptor inhibitors: a pooled analysis. Am Heart J. 2000; 140 (2): 206-211. [ Links ]
75. Gowda RM, Khan IA, Vasavada BC, Sacchi TJ. Therapeutics of platelet glycoprotein IIb/IIIa receptor antagonism. Am J Ther. 2004; 11 (4): 302-307. [ Links ]
76. Demolis JL, Robert A, Mouren M, Funck-Brentano C, Jaillon P. Pharmacokinetics and platelet antiaggregating effects of beraprost, an oral stable prostacyclin analogue, in healthy volunteers. J Cardiovasc Pharmacol. 1993; 22 (5): 711-716. [ Links ]
77. Kahn ML, Nakanishi-Matsui M, Shapiro MJ, Ishihara H, Coughlin SR. Protease-activated receptors 1 and 4 mediate activation of human platelets by thrombin. J Clin Invest. 1999; 103 (6): 879-887. [ Links ]
78. Kubo S, Ishiki T, Doe I, Sekiguchi F, Nishikawa H, Kawai K, et al. Distinct activity of peptide mimetic intracellular ligands (pepducins) for proteinase-activated receptor-1 in multiple cells/tissues. Ann NY Acad Sci. 2006; 1091: 445-459. [ Links ]
79. Wielders SJ, Bennaghmouch A, Reutelingsperger CP, Bevers EM, Lindhout T. Anticoagulant and antithrombotic properties of intracellular protease-activated receptor antagonists. J Thromb Haemost. 2007; 5 (3): 571-576. [ Links ]
80. Covic L, Misra M, Badar J, Singh C, Kuliopulos A. Pepducin-based intervention of thrombin-receptor signaling and systemic platelet activation. Nat Med. 2002; 8 (10): 1161-1165. [ Links ]
81. Levy JH, Sypniewski E. Aprotinin: a pharmacologic overview. Orthopedics. 2004; 27 (6 Suppl): s653-658. [ Links ]
82. DayJR,PunjabiPP,RandiAM,HaskardDO,LandisRC,Taylor KM. Clinical inhibition of the seven-transmembrane thrombin receptor (PAR1) by intravenous aprotinin during cardiothoracic surgery. Circulation. 2004; 110 (17): 2597-2600. [ Links ]
83. Poullis M, Manning R, Laffan M, Haskard DO, Taylor KM, Landis RC. The antithrombotic effect of aprotinin: actions mediated via the proteaseactivated receptor 1. J Thorac Cardiovasc Surg. 2000; 120 (2): 370-378. [ Links ]
84. Kakkos SK, Nicolaides AN. S-18886 Servier. Curr Opin Investig Drugs. 2002; 3 (9): 1324-1327. [ Links ]
85. Morrow JD, Hill KE, Burk RF, Nammour TM, Badr KF, Roberts LJ, 2nd. A series of prostaglandin F2-like compounds are produced in vivo in humans by a non-cyclooxygenase, free radical-catalyzed mechanism. Proc Natl Acad Sci U S A. 1990; 87 (23): 9383-9387. [ Links ]
86. VilahurG,CasaniL,BadimonL.AthromboxaneA2/prostaglandin H2 receptor antagonist (S18886) shows high antithrombotic efficacy in an experimental model of stent-induced thrombosis. Thromb Haemost. 2007; 98 (3): 662-669. [ Links ]
87. Belhassen L, Pelle G, Dubois-Rande JL, Adnot S. Improved endothelial functionby the thromboxane A2 receptor antagonist S 18886 in patients with coronary artery disease treated with aspirin. J Am Coll Cardiol. 2003; 41 (7): 1198-1204. [ Links ]
88. Viles-González JF, Fuster V, Corti R, Valdivieso C, Hutter R, Corda S, et al. Atherosclerosis regression and TP receptor inhibition: effect of S18886 on plaque size and composition--a magnetic resonance imaging study. Eur Heart J. 2005; 26 (15): 1557-1561. [ Links ]
89. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 2002; 324 (7329): 71-86. [ Links ]
90. Bertrand ME, Rupprecht HJ, Urban P, Gershlick AH. Double-blind study of the safety of clopidogrel with and without a loading dose in combination with aspirin compared with ticlopidine in combination with aspirin after coronary stenting: the clopidogrel aspirin stent international cooperative study (CLASSICS). Circulation. 2000; 102 (6): 624-629. [ Links ]
91. YusufS,ZhaoF,MehtaSR,ChrolaviciusS,TognoniG,FoxKK. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N Engl J Med. 2001; 345 (7): 494-502. [ Links ]
92. Cannon CP, Weintraub WS, Demopoulos LA, Vicari R, Frey MJ, Lakkis N, et al. Comparison of early invasive and conservative strategies in patients with unstable coronary syndromes treated with the glycoprotein IIb/IIIa inhibitor tirofiban. N Engl J Med. 2001; 344 (25): 1879-1887. [ Links ]
93. Peterson ED, Pollack CV, Jr., Roe MT, Parsons LS, Littrell KA, Canto JG, et al. Early use of glycoprotein IIb/IIIa inhibitors in non-ST-elevation acute myocardial infarction: observations from the National Registry of Myocardial Infarction 4. J Am Coll Cardiol. 2003; 42 (1): 45-53. [ Links ]
94. Topol EJ, Lincoff AM, Kereiakes DJ, Kleiman NS, Cohen EA, Ferguson JJ, et al. Multi-year follow-up of abciximab therapy in three randomized, placebo-controlled trials of percutaneous coronary revascularization. Am J Med. 2002; 113 (1): 1-6. [ Links ]
]]>
Recibido: Junio 22 de 2008.
Aceptado: Noviembre 4 de 2008.
* Autor a quien debe dirigir la correspondencia: ipalomo@utalca.cl
]]>