PHARMACOLOGY AND TOXICOLOGY
HEPATOPROTECTIVE EFFECT OF CALYCES EXTRACT OF Physalis peruviana ON HEPATOTOXICITY INDUCED BY CCl4 IN WISTAR RATS
EFECTO HEPATOPROTECTOR DE UN EXTRACTO DE CÁLICES DE Physalis peruviana EN HEPATOTOXICIDAD INDUCIDA POR CCl4 EN RATAS WISTAR
Reina Marcela TORO A. QF1; Diana Marcela ARAGÓN N. PhD1; Luis Fernando OSPINA G. PhD1*
1 Grupo de Investigación ''Principios Bioactivos de Plantas''; Departamento de Farmacia, Universidad Nacional de Colombia, A.A. 14490, Cra 30 No. 45-03. Bogotá D.C.,Colombia.
]]> * Autor a quien se debe dirigir la correspondencia: lfospinag@unal.edu.co.
Recibido: Noviembre 06 de 2013
Aceptado: Octubre 15 de 2013
ABSTRACT
Background: Physalis peruviana (''uchuva'', Solanaceae) is a widespread species of the South American Andes and widely used in traditional medicine. Its fruits are consumed as food and for the treatment of diabetes. The juice of Physalis peruviana fruits is topically applied in the eyes for pterigyum treatment. Previous works reported that the fruit extracts has modulating activity of oxidative stress in experimental diabetes models induced by streptozotocin. It has been attributed antipyretic, antimicrobial, analgesic and anti-inflammatory properties to the calyces enveloping the fruit. Reported literature demonstrates in vivo and in vitro that different calyx's extracts have antioxidant and anti-inflammatory activities. Objectives: To evaluate the in vivo hepatoprotective effect of the extract of Physalis peruviana calyces, involving inflammation and oxidative stress models at hepatic level. Methods: Hepatotoxicity was induced by single oral administration of CCl4 (2 mL / Kg in olive oil) in Wistar rats. Physalis peruviana extract (250 mg/Kg) and silymarin (200 mg/Kg), used as control drug, were administrated twice a day for five days. At the end of the experiment, animals were euthanized and the liver enzymes alanine aminotransferase, aspartate aminotransferase, and alkaline phosphatase were measured as well as some parameters of hepatic antioxidant status like superoxide dismutase and catalase activities, protein oxidation and lipid peroxidation. Results: Extract of Physalis peruviana calyces inhibited significantly (p < 0.001) liver oxidative stress caused by CCl4, maintaining superoxide dismutase and catalase activities close to normal. Studied extract also reduced significantly liver enzymes levels increased by CCl4 administration. Conclusion: It was suggested that the extract of Physalis peruviana calyces presents a hepatoprotective effect related to its antioxidant activity, especially regarding to lipid peroxidation inhibition.
Keywords: Physalys peruviana, hepatotoxicity, oxidative stress, antioxidant.
RESUMEN
Antecedentes: Physalis peruviana (''uchuva'', Solanaceae) es una especie distribuida en los Andes suramericanos y de amplio uso a nivel etnofarmacológico. Sus frutos, además de ser usados como alimento, también son consumidos para el tratamiento de la diabetes. Por su parte, el zumo del fruto es aplicado localmente para el tratamiento de pterigios. En modelos de diabetes experimental inducidos por estreptozotocina, trabajos previos han reportado que los extractos de los frutos poseen actividad moduladora del estrés oxidativo. A los cálices que envuelven el fruto, se les atribuyen propiedades antipiréticas, antimicrobianas, analgésicas y antiinflamatorias, entre otras. Estudios previos han demostrado en modelos in vivo e in vitro que diferentes extractos de los cálices presentan actividad antioxidante y antiinflamatoria. Objetivo: Evaluar el posible efecto hepatoprotector de un extracto de cálices de Physalis peruviana en un modelo experimental in vivo que involucra procesos de inflamación y de estrés oxidativo a nivel hepático. Métodos: Se empleó un modelo de hepatotoxicidad inducida por CCl4 en ratas Wistar, mediante una única administración oral de CCl4. Tanto el extracto de Physalis peruviana (250 mg/Kg) como la silimarina (200 mg/Kg), empleada como patrón de referencia, fueron administradas dos veces al día durante cinco días. Al final del experimento, los animales fueron sacrificados y se evaluaron los niveles hepáticos de las enzimas alanina-aminotranferasa, aspartato-transaminasa y fosfatasa alcalina, al igual que algunos parámetros del estado antioxidante hepático como actividad de superóxido dismutasa y catalasa, oxidación proteica y peroxidación lipídica. Resultados: El extracto evaluado de Physalis peruviana inhibió significativamente (p < 0,001) el estrés oxidativo causado a nivel hepático por el CCl4, manteniendo las actividades de superóxido dismutasa y catalasa con valores cercanos a los normales. El extracto también redujo significativamente los niveles de las enzimas hepáticas que fueron incrementados después de la administración de CCl4. Conclusión: Estos resultados sugieren que el extracto de cálices de Physalis peruviana posee un efecto hepatoprotector relacionado con su actividad antioxidante, especialmente en lo referente a la inhibición de la peroxidación lipídica.
]]> Palabras clave: Physalys peruviana, hepatotoxicidad, estrés oxidativo, antioxidante.
INTRODUCTION
Oxidative stress is employed to indicate the oxidative damage produced to biological molecules such as nucleic acids, proteins, lipids, and carbohydrates (1). This phenomenon occurs when reactive oxygen species (ROS), including superoxide anion, hydrogen peroxide (H2O2) and hydroxyl radical (•OH) exceed the cellular antioxidant defense. This imbalance produces adverse effects on the tissues and eventually cardiovascular or liver diseases, inflammatory processes and other chronic diseases such as hepatic fibrosis (2, 3).
The administration of carbon tetrachloride (CCl4) in rodents is the most studied experimental model of liver inflammation. The CCl4 is biotransformed in the liver by cytochrome P450 and produces trichloromethyl radical (CCl3•) and ROS, initiating a lipid peroxidation leading to hepatocyte death and subsequent inflammation of the organ (4). These changes modify the normal performance of liver and alter serum levels of liver alanine aminotransferase (ALT), aspartate aminotransferase (AST) and alkaline phosphatase (AP).
Cape gooseberry (Physalis peruviana L., Solanaceae) is an abundant specie of the South American Andes that has been widely used and studied for medicinal purposes; this specie has been used for its anti-mycobacterial, antipyretic, analgesic, diuretic, and anti-inflammatory properties (5, 6). Extracts from the calyces of Physalis peruviana have demonstrated anti-inflammatory (7), immunomodulatory (8) and anti-nociceptive activities (9). Furthermore, it has been reported antioxidant and hypoglycemic activity in fruit extracts (10), and effectiveness in the treatment of pterygium, due to the inhibition of fibroblasts proliferation (11). Therefore, the aim of the present study was to evaluate the possible hepatoprotective effect of calyces extract of P. peruviana in CCl4 hepatotoxicity model.
METHODS AND MATERIALS
Plant Material and extraction
]]> Physalis peruviana calyces were obtained from a local market of Bogota city (voucher specimen was deposited at The Herbario Nacional Colombiano -COL- 51200, identified by the Botanic C.I. Orozco-). Fresh calyces were selected and dried in air circulating oven at 50°C for 36 h. Dried material was milled and then was extracted by exhaustive percolation with petroleum ether according to other works (7). The extract in petroleum ether was fractionated by a liquid - liquid partition with methanol-water (9:1). The methanol-water fraction was dried in a rotary evaporator and used for this work.Animals
Male Wistar rats (220-250 g) were bred and housed under standard conditions (12 h light/12 h dark cycles, temperature 22 ± 2 °C). Food and water were available ad libitum. The protocol was approved by the Ethics Committee of Science Faculty of the National University of Colombia. The animals were cared for in accordance with the international ethical guidelines (12).
Treatment
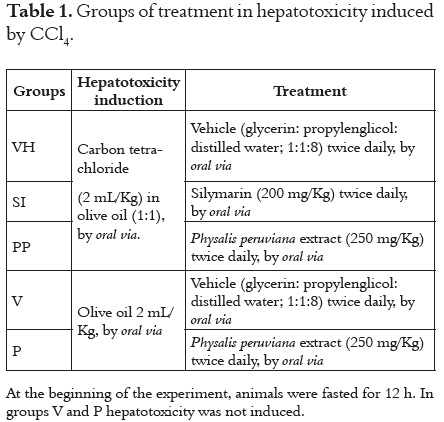
The rats were randomly divided into five groups of 10 animals each one. Table 1 describe the treatments.
At the end of the experiment, all animals were anesthetized for cardiac puncture in order to obtain blood for ferric reduction ability of plasma (FRAP) and biochemical determinations (alanine aminotransferase-ALT, alanine aminotransferase- AST, and alkaline phosphatase-AP). Finally, the rats were sacrificed by cervical dislocation and the livers were quickly removed, place in ice cold, rinsed, cut and homogenized (Polytron® PT-2100) in cold Tris–HCl (5 mM, pH 7.4). Homogenates were centrifuged (10000 rpm, 4°C, 10 min), and supernatants were used to evaluate the oxidative stress parameters. The protein concentration was determined using Bradford reagent.
Histopathological studies
Sections of liver samples were washed and fixed with 10% neutralized formalin (pH 7.4) and then stained with hematoxylin and eosin, and were kept under observation for pathological changes.
]]> Ferric reduction ability of plasma (FRAP)This method measure the antioxidants ability in biological sample by reducing the 2,4,6-tripyridyls- triazine (Fe3+-TPTZ) to the ferrous form (Fe2+) (13). The FRAP was expressed as mg FeSO4/mg protein.
Index of lipid peroxidation
This parameter was evaluated by the measure of thiobarbituric acid reactive substances (TBARs). Briefly, 450μL of homogenated were added to 50μL of phosphate buffers (50 mM, pH 7,4) and 1 mL of trichloroacetic acid (10%). The mix was centrifuged (2000 rpm, 10 min, 4°C) and 1 mL of supernatant was added to 1 mL of thiobarbithuric acid (0.67%). This mixture was boiled for 30 min, with the subsequent absorbance measurement at 532 nm. Concentration of the TBARs were expressed as (mmol/mL)/mg protein, using the expression ε = A/c*h, where ε = 153000 M–1cm–1.
Protein carbonyl content (COP)
This parameter was measured using 2,4-dinotrophenylhydrazine (DNPH) according to methods previously described (14). Briefly, 50 μL of homogenate was added to 250 μL of 2,4-dinitrophenylhydrazine (DNPH, 10 mM in HCl 2M) and left to stand at room temperature for 1h stirring every 15 min. Thereafter, 500 μL of cold trichloroacetic acid (TCA, 20%) were added and left to stand for 15 min at 4°C. Then, the mixture was centrifuged at 11000 rpm for 5 min, the supernatant was removed and the pellet was rinsed three times with ethanol: ethyl acetate (1:1), and centrifuged after each rinse for 7 min at 3000 rpm. Finally, the pellet was dissolved in 250 μL of Guanidine 6 M and incubated at 37°C for 10 min. The absorbance was measured at 360 nm. The carbonyl content was calculated using the expression ε = A/c*h, where ε = 22000 M–1cm–1 and expressed as nmol/mg protein.
Superoxide dismutase activity (SOD)
The Cu-SOD and Mn-SOD were determined by the riboflavin-o-dianisidine test (13). 4 μL of homogenate was added to 200 μL of the reaction mixture (0,1 mL of o-dianisidine 6 mM; 0,1 mL of riboflavin 0.29 mM and phosphate buffer, 50 mM, pH 7.8). The mixture was incubated at dark for 5 min at 25°C and then exposed for 8 min to fluorescent light; the absorbance was measured at 415 nm before and after the light exposition. The SOD concentration was extrapolated from a calibration curve and expressed as U/mg protein.
Catalase activity (CAT)
The method is based on the H2O2 decomposition and the rate constant determination k (s–1) (15). Briefly, 250 μL of H2O2 (10 mM) were added to 5 μL of homogenate and 45 μL of phosphate buffer (50 mM, pH 7.0). Immediately, the change in the absorbance was measured at 240 nm for 30 sec. The enzyme activity was expressed as k (s mg protein)–1.
]]> Statistical AnalysisThe results were expressed as means ± SD and were analyzed by one-way analysis of variance (ANOVA), followed by Dunnett's test. Statistical differences were considered (*) when p < 0.05.
RESULTS
Significant increases of enzymes AST, ALT and ALP (more than 5 times baseline levels) were found in the control group (VH) (figure 1). These results are similar to those reported by other authors and were consistent to the mode of action of CCl4, which induces a large lipid peroxidation in the endoplasmic reticulum of hepatocytes (16, 17).
Histopathological studies showed that rats without CCl4 had a normal hepatic architecture, indicating that the vehicle and the extract of Physalis peruviana do not have hepatotoxic effect (figure 2). The administration of CCl4 induced sub-massive hepatic necrosis and a pronounced hepatic microvacuolar steatosis (figure 2).
Regarding to oxidative stress parameters, a significant increase (around 50%) in SOD and CAT activities [1.87 ± 0.13 units/ mg protein and 3.16 ± 0.04 k/(s* mg protein), respectively] in the group VH respect to group V was found [0.60± 0.16 units/ mg protein and 1.15 ± 0.17 k/(s* mg protein), respectively], while silymarin and P. peruviana maintain enzyme activities near to normal (figures 3A y 3B).
Furthermore, a high level of carbonyl (figure 3C) was found in group VH (5.78 ± 0.94 nmol/mg protein) being three times more than in group V (1.45 ± 0.06 nmol/mg protein). Similar to the other parameters evaluated, silymarin and the extract of P. peruviana showed a decrease in the levels of carbonyl groups. Lipid peroxidation, expressed as nmoles of malondialdehyde (MDA), were increased almost twice in the group VH (9.29 ± 0.19 nmol MDA/mg protein) (figure 3D). In this case, silymarin prevented lipid peroxidation produced in the liver by CCl4 in a 50%, while the extract of P. peruviana reduced significantly this damage, in a 30% respect to group VH.
DISCUSSION
]]> Serum levels of liver enzymes such as ALT, AST and ALP, and other compounds such as urea, are important markers of liver function and the possible damage to hepatocytes. These enzymes are released from the cytosol and mitochondria, therefore lesions that alter the permeability of hepatocytes elevate the normal levels of ALT, AST and ALP more than three times, allowing the passage of these enzymes to peripheral blood (16, 18).Treatment with silymarin (SI) and P. peruviana (PP) prevent a significant increase in the levels of ALT, AST and ALP. This could be due to the antioxidant activity of these treatments. Silymarin is a mixture of flavonolignans extracted from Silybum marianum, which had reported protective effect against various liver diseases (19), including those induced by oxidative stress like cirrhosis, diabetes and liver fibrosis (20-22). Its mechanism of action is still unknown; however, two possible mechanisms proposed are through free radical scavenging and regulation of immune functions modulated by cytokines (23, 24). The decrease in enzyme levels (AST, ALT and ALP) in silymarin group (SI) is probably due to the antioxidant activity of silymarin, demonstrated against DPPH, ROS such as •OH, HOCl and against lipid peroxidation. A similar effect can be attributed to the extract of P. peruviana which had demonstrated superoxide radical scavenging activity, inhibition of lipid peroxidation in vitro and scavenging of ROS like H2O2 (6).
Microvacuolar steatosis found in histopalogical studies is associated with a state of oxidative stress generated, since the trichloromethyl radical (•CCl3) covalently binds to cellular components such as glutathione reductase, inhibiting the secretion of lipoproteins causing the subsequent steatosis. On the other hand, after the •CCl3 reaction with molecular oxygen forms the peroxyl radical (•OOCCl3), the process of lipid peroxidation initiates generating disruption of membrane cell of the hepatocytes (25, 26). Furthermore, histopathological findings in liver tissues of rats with silymarin and P. peruviana presented a slight attenuation of liver steatosis, and absence of hepatic necrosis in the treatment with P. peruviana (figure 2).
As mentioned before, after CCl4 administration diverse reactive oxygen species (ROS) are generated, which must be neutralized by the antioxidant defense system of the organism like the enzymes SOD and CAT. The role of SOD is the dismutation of the superoxide radical to hydrogen peroxide, which is converted in water by CAT . For this reason, in most events of oxidative stress these two enzymes are affected (27). Previous studies have fully described the alteration; generally, increase in the activity of antioxidant enzymes such as SOD and CAT after administration of CCl4 to Wistar rats (28). Results found in this investigation with P. peruviana were similar to those obtained from silymarin and other plants such as Solanum tuberosum (29), Phyllanthus amarus (30) and Hippophae rhamnoides (31).
Recently, it was studied the possibility that free radicals induced not only lipid peroxidation but also oxidation of carbonyl groups of proteins. In this way, it was established that •OH radicals can extract an H of any carbon of amino acids, forming a carbonyl radical which subsequently can be converted to an alkyl peroxyl radical. The radical formed can break peptide bonds, generating hydrolysis and oxidation of other proteins (18). Finally, free radicals can cause significant alterations of cell function, (including death) causing oxidation and inactivation of enzymes and accumulation of other oxidized proteins. The effect of CCl4 on protein oxidation is not fully elucidated, but recent studies demonstrated that in the presence of this toxic the liver shows an increase (about 50%) in the levels of carbonyl groups (32). The effect of silymarin and the extract of P. peruviana in the decreasing of carbonyl groups levels could be associated with the antioxidant activity previously described.
Lipid peroxidation results are similar to those reported for other antioxidants such as carotenoids and tocopherols (33) in models of hepatotoxicity induced by CCl4. Clearly, there is a strong relationship between in vivo antioxidant activity and in vitro inhibition of lipid peroxidation reported for the extracts of Physalis peruviana (6).
CONCLUSIONS
In conclusion, the evaluated calyces extract of Physalis peruviana has a significant hepatoprotective activity that could be related to its antioxidant capacity, especially by the inhibition of lipid peroxidation.
]]> ACKNOWLEDGEMENTS
This work was supported by the National University of Colombia, CIAT (International Center of Tropical Agriculture) and the Ministerio de Agricultura y Desarrollo Rural of Colombian government.
Conflict of interest: The author reports no declaration of interest.
REFERENCES
1. Cadenas E. Biochemistry of oxygen toxicity. Ann Rev Biochem. 1989 Jun; 58 (1): 79-110. [ Links ]
2. Halliwell B. Antioxidants and human disease: A general introduction. Nutrition Rev. 1997 Jan; 55 (1): 44-52. [ Links ]
3. Parola M, Robino G. Oxidative stress-related molecules and liver fibrosis. J Hepatol. 2001 Aug; 35 (2): 297-306. [ Links ]
4. Recknagel RO, Glende EA, Dolak JA, Waller RL. Mechanisms of carbon tetrachloride toxicity. Pharmacol Ther. 1989 Jan; 43 (1): 139-154. [ Links ]
5. Fischer G, Miranda D, Piedrahita W, Romero J. Avances en cultivo poscosecha y exportación de la uchuva, Physalis peruviana L., en Colombia. Bogotá, Colombia: Unibiblios; 2008. 325p. [ Links ]
6. Wu SJ, Ng LT, Huang YM, Lin DL, Wang SS, Huang S, et al. Antioxidant activities of Physalis peruviana. Biol Pharm Bull. 2005 Jun; 28 (6): 963-966. [ Links ]
7. Franco LA, Matiz GE, Calle J, Pinzón R, Ospina LF. Actividad antiinflamatoria de extractos y fracciones obtenidas de cálices de Physalis peruviana L. Biomedica. 2007 Mar; 27 (1): 110-115. [ Links ]
8. Martínez W, Ospina LF, Granados D, Delgado G. In vitro studies on the relationship between the anti-inflammatory activity of Physalis peruviana extracts and the phagocytic process. Immunopharmamacol Immunotoxicol. 2010 Mar; 32 (1): 63-73. [ Links ]
9. Muñoz CE, Vergel NE, Aragón DM, Ospina LF. Efecto antinociceptivo de Critoniella acuminata, Physalis peruviana y Salvia rubescens. Rev Col Cienc Quím Farm. 2009 Jan- Jun; 38 (1): 31-41. [ Links ]
10. Mora AC, Aragón DM, Ospina LF. Effects of Physalis peruviana fruit extract on stress oxidative parameters in streptozotocindiabetic rats. Lat Am J Pharm. 2010 Jul; 29 (7): 1132-1136. [ Links ]
11. Pardo JM, Fontanilla MR, Ospina LF, Espinosa L. Determining the pharmacological activity of Physalis peruviana fruit juice on rabbit eyes and fibroblast primary cultures. Invest Ophtalmol Vis Sci. 2008 Jul; 49 (7): 3074-3079. [ Links ]
12. National Research Council. Guide for the Care and Use of Laboratory Animals. Washington, D.C: National Academy Press; 1996. 98 p. [ Links ]
13. Paynter J. Changes in activity of the manganese superoxide dismutase enzyme in tissues of the rat with changes in dietary manganese. J Nutr. 1980 Mar; 110 (3): 437-447. [ Links ]
14. Baltacioğlu F, Akalin E, Alver A, Değer O, Karabulut E. Protein carbonyl levels in serum and gingival crevicular fluid in patients with chronic periodontitis. Arch Oral Biol. 2008 Aug; 53 (8): 716-722. [ Links ]
15. Aebi H. Catalase in vitro. Meth Enzymol. 1981 Jan; 105 (1): 121-126. [ Links ]
16. Wang PY, Kaneko T, Tsukada H, Nakano M, Nakajima T, Sato A. Time courses of hepatic injuries induced by chloroform and by carbon tetrachloride: comparison of biochemical and histopathological changes. Arch Toxicol. 1997 Oct; 71 (10): 638-645. [ Links ]
17. Basu S. Carbon tetrachloride-induced lipid peroxidation: eicosanoid formation and their regulation by antioxidant nutrients. Toxicology. 2003 Jul; 189 (1-2): 113-127. [ Links ]
18. Smart R, Hodgson E. Molecular and biochemical toxicology. 3rd ed. New York, USA: John Wiley and Sons; 2008, 535 p. [ Links ]
19. Shaker E, Mahmoud H, Mnaa S. Silymarin, the antioxidant component and Silybum marianum extracts prevent liver damage. Food Chem Toxicol. 2010 Mar; 48 (3): 803-806. [ Links ]
20. Lieber CS, Leo MA, Cao Q, Ren C, DeCarli LM. Silymarin retards the progression of alcohol-induced hepatic fibrosis in baboons. J Clinic Gastroenterol. 2003 Oct; 37 (4): 336-339. [ Links ]
21. Soto C, Pérez J, García V, Uría E, Vadillo M, Raya L. Effect of silymarin on kidneys of rats suffering from alloxan-induced diabetes mellitus. Phytomedicine 2010 Dec; 17 (14): 1090-1094. [ Links ]
22. Shaker ME, Zalata KR, Mehal WZ, Shiha GE, Ibrahim TM. Comparison of imatinib, nilotinib and silymarin in the treatment of carbon tetrachloride-induced hepatic oxidative stress, injury and fibrosis. Toxicol Appl Pharmacol. 2011 Apr; 252 (2): 165-175. [ Links ]
23. Ferenci P, Dragosics B, Dittrich H, Frank H, Benda L, Lochs H, et al. Randomized controlled trial of silymarin treatment in patients with cirrhosis of the liver. J Hepatol. 1989 Jul; 9 (1): 105-113. [ Links ]
24. Luper S. A review of plants used in the treatment of liver disease: Part 1. Altern Med Rev. 1989 Dec; 3 (6): 410-421. [ Links ]
25. Boll M, Weber LW, Becker E, Stampfl A. Mechanism of carbon tetrachloride induced hepatotoxicity. Hepatocellular damage by reactive carbon tetrachloride metabolites. Z Naturforsch C. 2001 Jul-Aug; 56 (7-8): 649-659. [ Links ]
26. Weber LW, Boll M, Stampfl A. Hepatotoxicity and mechanism of action of haloalkanes: carbon tetrachloride as a toxicological model. Crit Rev Toxicol, 2003 Mar; 33 (2): 105-136. [ Links ]
27. Halliwell B, Gutteridge J. Free radical in biology and medicine. 4 ed. New York, USA: Oxford University Press; 2007. 851 p. [ Links ]
28. Szymonik-Lesiuk S, Czechowska G, Stryjecka-Zimmer M, Siomka M, Madro A, Celinski K, et al. The in vitro and in vivo antioxidant properties of seabuckthorn (Hippophae rhamnoides L.) seed oil. Food Chem. 2011 Mar; 125 (2): 652-659. [ Links ]
29. Singha N, Kamatha V, Narasimhamurthy M, Rajini PS. Protective effect of potato peel extract against carbon tetrachlorideinduced liver injury in rats. Environ Toxicol Pharmacol. 2008 Aug; 26 (8): 241-246. [ Links ]
30. Bhattacharjee R, Sil PC. Protein isolate from the herb, Phyllanthus niruri L. (Euphorbiaceae), plays hepatoprotective role against carbon tetrachloride e induced liver damage via its antioxidant properties. Food Chem Toxicol. 2007 May; 45 (5): 817-826. [ Links ]
31. Maheshwari DT, Yogendra Kumar MS, Verma SK, Singh VK, Singh SN. Antioxidant and hepatoprotective activities of phenolic rich fraction of Seabuckthorn (Hippophae rhamnoides L.) leaves. Food Chem Toxicol. 2011 Sept; 49 (9): 2422-2428. [ Links ]
32. Sundari P, Banumath W. Does oxidative protein damage play a role in the pathogenesis of carbon tetrachloride-induced liver injury in the rat?. Biochim Biophys Acta. 1997 Dec; 162 (2-3): 169-176. [ Links ]
33. Krinsky NI. Antioxidant functions of carotenoids. Free Radic Biol Med. 1989 Jun; 7 (6): 617-635. [ Links ]
]]>