 MATERIALS AND METHODS
MATERIALS AND METHODS  RESULTS ]]>
RESULTS ]]>
 DISCUSSION
DISCUSSION  ACKNOWLEDGMENTS
ACKNOWLEDGMENTS  BIBLIOGRAPHY
BIBLIOGRAPHYUSE OF A MICRO TITLE PLATE DILUTION ASSAY TO MEASURE ACTIVITY OF ANTIFUNGAL COMPOUNDS AGAINST Mycosphaerella Fijiensis, MORELET.
Jorge Enrique Peláez Montoya1; Luz Estella Vásquez David2; Thais Judith Díaz Brito3; Darío Antonio Castañeda Sánchez4; Esperanza Rodríguez Beltrán5 and Rafael Eduardo Arango Isaza6
1 Investigador. Unidad de Biotecnología Vegetal, Corporación para Investigaciones Biológicas, CIB, Carrera 72ª No. 78B- 141, Medellín, Colombia. <cib@cib.org.co>
2 Investigadora. Unidad de Biotecnología Vegetal, Corporación para Investigaciones Biológicas, CIB, Carrera 72ª No. 78B- 141, Medellín, Colombia. <lvasquez@cib.org.co>
3 Investigadora. Unidad de Biotecnología Vegetal, Corporación para Investigaciones Biológicas, CIB, Carrera 72ª No. 78B- 141, Medellín, Colombia. <cib@cib.org.co>
4 Investigador. Centro de Investigaciones del Banano CENIBANANO, Asociación de Bananeros de Colombia AUGURA, Conjunto residencial Los Almendros, Urabá, Colombia.<dacastanedas@gmail.com>
5 Investigadora. Unidad de Biotecnología Vegetal, Corporación para Investigaciones Biológicas, CIB, Carrera 72ª No. 78B- 141, Medellín, Colombia. <erodriguez@cib.org.co> ]]>
6 Profesor Asociado. Universidad Nacional de Colombia, Sede Medellín. Escuela de Biociencias. Facultad de Ciencias. A.A. 3840, Medellín, Colombia. <rafaelarango@une.net.co>
Recibido: Junio 14 de 2005; aceptado: Julio 11 de 2006.
Black Sigatoka, caused by the fungus Mycosphaerella fijiensis is the most important disease in banana plantations. The fungus is controlled mainly by fungicide applications with an annual cost of about 350 million dollars in Latin and Central America. Due to the appearance of resistant strains and to the economical and environmental impact caused by the extensive use of fungicides, accurate methods are necessary for monitoring the fungal sensitivity to these agents. In this paper we describe the standarization of a method based on microplate dilutions that measures IC50 of different antifungal compounds against single ascospore cultures of M. fijiensis. The method was used to measure the sensitivity of 30 strains collected from different regions in Colombia against Propiconazol, Benomyl and Azoxystrobin. Used at a larger scale, this method could be useful to monitor M. fijiensis sensitivity against fungicides and to search for new compounds with activity against the fungus.
Key words: Mycosphaerella fijiensis, antifungal activity assay, fungicide resistance.
RESUMEN
La Sigatoka negra, causada por el hongo Mycosphaerella fijiensis, es la enfermedad más importante que afecta plantaciones de banano y plátano. El hongo es controlado principalmente mediante fungicidas químicos con un costo anual de cerca de 350 millones de dólares para América latina y 25 millones de dólares para Colombia. Debido al desarrollo de cepas del hongo resistentes a los fungicidas y a las consecuencias que para el medio ambiente tiene su uso intensivo, es importante el desarrollo de métodos que permitan monitorear de una manera precisa la aparición de resistencia. También es de gran utilidad disponer de un método que permita evaluar en forma sencilla la actividad de nuevas sustancias contra el hongo. En este artículo describimos la estandarización de un método basado en diluciones en microplatos que permite determinar la IC50 de diversos compuestos antifungicos contra cultivos monospóricos de M. fijiensis. Para su validación, el método fue utilizado para medir la sensibilidad de 30 aislamientos, obtenidos de diversas regiones en Colombia, a los fungicidas Propiconazol, Benomyl y Azoxystrobin. Este método es útil para supervisar sensibilidad del M. fijiensis. a los fungicidas químicos y para buscar nuevos compuestos con actividad contra el hongo.
Palabras claves: Mycosphaerella fijiensis, ensayo de la actividad antifungica, resistencia del fungicida.
 MATERIALS AND METHODS
MATERIALS AND METHODS
 RESULTS ]]>
RESULTS ]]>
 DISCUSSION
DISCUSSION
 ACKNOWLEDGMENTS
ACKNOWLEDGMENTS
 BIBLIOGRAPHY
BIBLIOGRAPHY
Bananas and plantains constitute an important source of food for million of people in the world, including Africa, Southeast Asia, South and Central America. These crops are constantly affected by black sigatoka, disease caused by the fungus Mycosphaerella fijiensis (Fullerton 1995, Stover and Simmonds 1987) which causes loses from 18 to 50% of the total production costs (Gaulh 1994, Pérez 1996, Stover 1980). Black sigatoka is controlled mainly by fungicides representing costs of about 350 million dollars per year, in Central and South America (Molina and Krausz 1989, Stierle et al. 1991). In Colombia , an average of 20 - 30 cycles fungicide applications per year, which correspond to about 2.200.000 gallons and a cost of about US$ 30 million a year (Augura 2000). This situation is becoming worse since the number of chemical groups used to control the disease is low, allowing the fungus to develop resistance against such chemical compounds. Due to the economical and environmental impact caused by the extensive use of fungicides, sensitive and accurate methods are necessary for monitoring the M. fijiensis sensitivity against fungicides and to find new compounds with activity against the fungus.
The main systemic fungicides used to control black sigatoka disease have been Bencimidazols, Triazoles, strobirulines and Morfolines. In 1991- 1992 a wide resistance to Benomyl was reported in Costa Rica , situation which increased the use of Propiconazol (Guzman and Romero 1997). Loss of sensitivity to Triazols was found in Belize, Guatemala, Honduras, Costa Rica, Mexico, Panamá and Cameroon (Cronshaw, Lorenz and Mappes 1994). More recently, widespread loss of sensitivity to Azoxystrobin (Sierotzki 2000) has been found. Until now, no loss of sensitivity has been reported for Tridemorph.
Methods for monitoring resistance to fungicides, to control black sigatoka, were initially developed for Bencimidazols and were later modified for Triazols and Morpholines (Anon 1998). Basically they consist of the measurement of ascospore germination after discharge from M. fijiensis infected leaves on agar water plates, supplemented with different fungicide concentrations (DuPont Latin America, 1983). Another method uses monosporic cultures made in PDA from ascospores discharged on agar water and then subcultured in PDA plus different fungicide concentrations; fungus growth is determined by measuring the colony diameter (Romero 1995). Although these methods are easy to carry out and give results relatively fast, they are tedious to perform and inaccurate because the data collection mainly depends on the criteria of the technician.
In this work we described an automated and semiquantitative method to estimate M. fijiensis sensitivity to different antifungal compounds. The fungus is cultured in liquid media amended with different concentrations of the compound and incubated in ELISA 96 wells microplates. The growth of the fungus is monitored at different times by measuring its OD595ηm in an ELISA reader. This method offers an alternative way to monitor resistance and is useful to corroborate results found with other techniques as it can give more accurate results when a strain is suspected to be resistant.
Isolation of M. fijiensis. Fresh and dry leaf samples from banana plants on different stages of infection with M. fijiensis were collected during 1998 and 1999 from several Colombian Bananas and plantains producer regions ( Santa Marta, Tame and Urabá). The leaf samples were allowed to discharge ascospores on water agar plates to produce monosporic cultures according to the method described by Stover 1969. Single germinated ascospores were transferred to potato dextrose agar (PDA) (BBL- Becton Dickinson USA ) and incubated at 27 ºC for 20 days.
Antifungal agents. The following three antifungal agents were used: Propiconazol (Tilt® 25EC; Ciba, Basel, Switzerland ), Benomyl (Benlate®, E. I. DuPont de Nemours) and Azoxystrobin (Bankit® Syngenta). Benomyl was supplied by the manufacturer as standard powder and a stock solution at 5000 mg/l was prepared with sterile distilled water. Stock solutions of Propiconazol, Azoxystrobin and Benomyl were prepared from liquid suspensions, at 50, 100 mg/l and 1000 mg/l in sterile distilled water respectively. ]]> From each compound, several dilutions were prepared in sterile distilled water as follows: 0, 0,01; 0,1; 0,5; 1; 5; and 10 mg/l of Benomyl, 0, 0,01; 0,03; 0,1; 0,3; 1 and 10 mg/l of Propiconazol, 0; 0,005; 0,01; 0,1; 1,0; 3,0 and 10 mg/l of Azoxystrobin. Inoculum preparation. Fresh monosporic subcultures of different M. fijiensis strains grown for 15 20 days at 27 °C in Potato Dextrose Agar PDA (BBL, Becton Dickinson Cockeysville, USA) were used as source to obtain the inoculum. Micelium from those subcultures were resuspended in sterile distilled water. The suspension was then fragmented by vortexing with glass beans of 6mm diam. (Schott, Wertheim Germany) and vortexed during 1- 2 min. followed by filtration with sterile "etamine" cloth (100μm pore) to obtain uniform miceliar fragments. Concentration of miceliar fragments was measured by Neubaver chamber (1/10 mm deep, bright line - Boeco, Germany) and adjusted to 2 x 104, 2 x 105 and 2 x 106 miceliar fragments /ml with water, to test the best working inoculum dilution. Microplate technique. Standardization of the technique was performed with strain No. 981111 chosen at random, in sterile, flat- bottomed 96- well microplates with low evaporation lid (Falcon- Becton Dickinson). Each well was filled with 50 μl of Sabouraud broth (BBLTM Becton Dickinson, Sparks, MD, USA ), 50 μl of the fungal inoculum and 50 μl of the different drug dilutions. Wells filled with 50 μl of Sabouraud medium, 50 μl H2O and 50 μl inoculum were used as positive control. Blanks consisted of 50 μl of Sabouraud medium and 100 μl H20. The microplates were incubated at 27˚C for several days. Miceliar growth was measured by spectrophotometer (Biorad model 550). Readings of the OD595 ηm were taken for the suspensions in each well were taken daily and up to 17 days. Analysis of the results. Growth percentage was determined using a control that did not receive antifungal treatment and its growth was considered as 100 %. Growth percentage was calculated with the formula: % Growth = (mean growth in each sample/ mean growth in the control)x 100. Data was plotted as percentage of growth inhibition against the logarithm of the fungicide concentration. IC50 was calculated using a curve fitting program (Kyplot version 1.0 beta 8 program) with the formula Y = 100x (A1/X+A1) where Y = % of fungal growth, X = fungicide concentration and A1 = IC50. Restrictive random blocks with different repetition numbers and three repetition per each fungicide product was used as experimental design. Different concentrations (seven for each one) were used in these experiments, a blank and a control, all of them with four repetitions.Statistical Analysis. Statistical analysis of samples was done with a one way analysis of variance with significance level of 95 %.
RESULTS
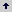
M. fijiensis strains were obtained from single ascospores collected after discharge from infected leaves on a Petri dish and subcultured on PDA. All isolates used were confirmed as M. fijiensis by a PCR based method as previously described (Johanson 1993). Cultures were grown on solid PDA from approximately 20 days until enough mycelium was available for performing micro plate assays.
Inuoculum preparation. We tested two different methods for inoculum preparation. The first method was based on a spectrophotometric assay as has been recommended for filamentous fungi (EspinelIngroff and Kerkering 1991). However this method proved difficult to implement for M. fijiensis and showed a high degree of variability of the inoculum size (data not shown). The second method consisted in counting mycelial fragments with a Neubaver chamber and adjusting fragment concentrations to a desired number (2,5 x 105). This method gave consistent results and was therefore adopted for all the assays.Fungal Growth curves. Several growth curves of the fungus with inoculum sizes of 2 x 102 2 x 104 2 x105 5 x 105 and 15 x 105 hyphal fragments /ml were performed in order to determine which was the best one. As seen in Figure 1A, adequate growth is seen from inoculum sizes of 2 x 105 and up while there is very little or no growth with 2 x 104 and 2 x 102. Fungal growth is rather slow, with an initial lag phase of about 5 days, followed by a long log phase from the 5th to the 12th day approximately and reaching a stationary phase from the day 17. Based on these data we decided to use a 12 day incubation time for growth inhibition assays.
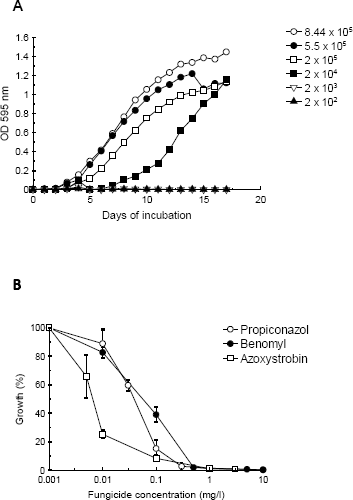
Figure 1. A. growth curve of M. fijiensis in microtiter wells using Sabouraud broth and different inoculum concentrations. Each microtiter well was filled with 50 μl of culture media, and 50 μl of the fungal inoculum. O.D. readings were taken daily at 595 nm. B. growth inhibition curves of M. fijiensis with different concentrations of antifungal agents. Each well was filled with 50 μl of Sabouraud broth, 50 μl of the fungal inoculum and 50 μl of the different drug dilutions. O.D. readings were taken after 8 days of incubation at 27 ˚C.
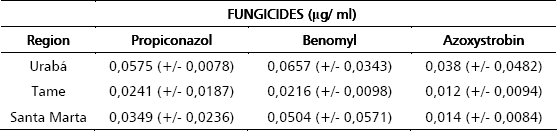
Growth inhibition assays. Assays to determine growth inhibition by the antifungal agents were setup using different concentrations of the chemical agents and performing O.D. readings at fixed incubation times. The concentrations chosen for each compound varied and were based on concentrations recommended by manufacturers for ascospore germination inhibition assays. As seen in figure 1B all compounds tested gave clear inhibition curves which allowed determination of IC50s. Measurement of IC50 values of isolates collected at different geographical locations. In order to test the microplate dilution assay and see if we could find significant differences among strains, we measured IC50 values for 30 M. fijiensis isolates collected in two different years from several regions in Colombia . Overall, IC50 values were 0,038 mg/l (+/- 0,023) for propiconazol, 0,045 mg/l (+/-0,022) for benzymidazole and 0,021 (+/- 0,014) for strobirulin. As seen in Table 1 no statistical difference was found in IC50 values of isolates from different geographic regions or from isolates collected at different years (Table 2).
Table 1. IC50 (mg/ml) averages obtained by the microplate technique of Benomyl, Propiconazol and Azoxystrobin from all the strains collected in different Colombian regions. In parenthesis the Standard deviations of each value.

Regular methods for determination of fungicide sensitivity to M. fijiensis are time consuming and some times inaccurate due to the ability and interpretation by the technician. The method described here offers the advantage of accuracy in the results because it avoids the subjectivity involved in deciding the presence of spore germination inhibition. Additionally, it allows determination of IC50 values for individual singleascospore, cultures which would permit accurate determination of variation sensitivities within populations. Another method that determines IC50 values based on measurement of young colony diameter, has been used to study susceptibility of M. fijiensis to propiconazol (Romero 1997). However, this method has the limitation that size and viability of mycelial fragments are difficult to standardize and M. fijiensis colonies grow very slowly and are difficult to measure.
Errors on readings of the automated method presented here can occur due to the nature of the fungal suspension used as inoculum. It is possible that using conidia instead of mycelial fragments could diminish the assays variability. Nevertheless, fidelity of the results increases when the number of replica wells is increased.
Using the microplate dilution method we demonstrated the effect on fungal growth of three fungicides (Benomyl, Propiconazol and Azoxystrobin) commonly used to control M. fijiensis in banana and plantain fields. In general, all the three fungicides showed consistent dose dependent growth inhibition which allowed the determination of IC50 values. The results of the IC50s (Table 1) shows that all the fungicides used had good growth inhibitory activity against M. fijiensis. These results are in agreement with the IC50s obtained from M. fijiensis isolated form different Colombian regions (Table 1) in two years (Table 2).
The number of samples used in this study does not allow making accurate conclusions about the resistance status of the fungal populations studied in the different regions. However, as the method can be easily scaled up, it has the potential to be used to follow fungal sensitivities of populations if an appropriate number of fungal strains and good sampling strategies are used. Furthermore, once a strain is identified as less sensitive or resistant to a compound, the method allows to accurately measure the level of resistance of the individual strain. This method also allows the screening of new antifungal compounds against the fungus.
ACKNOWLEDGMENTS
This work was supported in part by the Fondo Colombiano de Investigaciones Científicas (Colciencias), Colombia grant No. 2213-05-157-97, The Corporación para Investigaciones Biológicas, CIB and the Postgraduate Program in Biotechnology of The Universidad Nacional de Colombia, Sede Medellín. We thanks Dr. Lucia Afanador Kafuri and Dr. Nadya Cardona Bustos for reviewing and improving the manuscript.
]]>Carta informativa Augura (Colombia). Boletín no. 210 Medellín: Augura. p.18. [ Links ]
5a Reunión del Grupo de Trabajo de Banano de FRAC. 1998. Miami, 21-23 de Enero de 1998. Organizada por Rohm and Haas [ Links ]
Cronshaw, K; Lorenz G. and Mappes, D. 1994. Monitoring results of Mycosphaerella fijiensis to tridemorph. p. 315-321. En: Heany, S., Slawson, D., Holloman, D. W., Smith, M., Russell, P. E. and Parry, D. W., eds. Fungicide resistance. En: British Crop Protection Council Monograph. No. 60. [ Links ]
Du Pont. 1983. Black and yellow sigatokas: improved identification and management techniques. Florida, U.S.: Du Pont Latin America, Coral Gables. 17 p. [ Links ]
Espinel-Ingroff, A. and Kerkering, T. M. 1991. Spectrophotometric method of inoculom preparation for the in vitro susceptibility testing of filamentous fungi. En: Journal of Clinical Microbiology. Vol. 29, no. 2; p. 393-394. [ Links ]
Fullerton, R. H. and Olsen, T. I. 1995. Pathogenic variability of Mycosphaerella fijiensis Morelet, cause of black sigatoka in banana and plantain. En: New Zealand Journal of Crop and Horticultural Science. Vol. 23, no. 1; p. 39-48. [ Links ]
Gaulh, F. 1994. Epidemiology and ecology of black sigatoka (Mycosphaerella fijiensis Morelet) in plantain and banana in Costa Rica, Central America. Montpellier, France : INIBAP. 120 p. [ Links ]
Guzmán, M. y Romero, R. 1997. Comparación de los fungicidas azoxistrobina, propiconazole y diefeconazole en el control de la sigatoka negra (Mycosphaerella fijiensis Morelet), en banano (Musa AAA). En: Corbana. Vol. 22, no. 47; p. 49-59. [ Links ]
Johanson A. and Jeger M. 1993. Use of PCR for detection of Mycosphaerella fijiensis and Mycosphaerella musicola, the causal agents of sigatoka leaf spot in banana and plantain. En: Mycology Research. Vol. 97, no.6; p. 670-674. [ Links ]
Molina, G. and Kraus, J. P. 1989. A phytotoxic activity in extracts of cultures of Mycosphaerella fijiensis var. difformis and its use to evaluate host resistance to black sigatoka. En: Plant Disease. Vol. 73, no. 2; p. 142-143. [ Links ]
Pérez L. 1996. Manual para el control integrado de la sigatoka negra (Mycosphaerella fijiensis) y sigatoka amarilla (Mycosphaerella musicola Leach Mulder) en banano y plátano. FAO. Ministerio de Agricultura de la República de Cuba. 54 p. [ Links ]
Romero, R. A. and Sutton, T. B. 1997. Sensitivity of Mycosphaerella fijiensis, causal agent of black Sigatoka of banana, to propiconazole. En: Phytopathology. Vol. 87; p. 96-100. [ Links ]
Sierotzki, H., Parisi, S., Steinfeld, U., Tenzer, I., Poirey, S. and Gisi, U. 2000. Mode of resistance to respiratory inhibitors at the cytocrhome Bc1 enzyme complex of Mycosphaerella Fijiensis field isolates. En: Pest Management Science. Vol. 56, no. 10; p. 833-841. [ Links ]
Stierle, A. A., Upadhyay, R., Hershenhorn, J., Strobel, G. A. and Molina, G. 1991. The phytotoxins of Mycosphaerella fijiensis, the causative agent of black sigatoka disease of bananas and plantains. En: Experientia Vol. 47, no. 8; p. 853-895. [ Links ]
Stover, R. H. 1969. The Mycosphaerella species associated with banana leaf spots. En: Tropical Agriculture ( Trinidad). Vol. 46; p. 325-332. [ Links ]
________. 1980. Sigatoka leaf spot of banana and plantains. En: Plant Disease. Vol. 64, no. 8; p. 750-755. [ Links ]
________ and Simmonds, N. W. 1987. Bananas 3ed. New York: Longman Scientific and Technical. 468 p. [ Links ] ]]>