Detección de PMTV Utilizando Anticuerpos Policlonales Contra un Péptido Antigénico Derivado de la Cápside Viral
Yuliana Gallo García1; Pablo A. Gutiérrez Sánchez2 and Mauricio Marín Montoya3
1 Biological Engineer. Biochemistry. Universidad Nacional de Colombia - Sede Medellín - Facultad de Ciencias - Laboratorio de Microbiología Industrial. A.A. 3840, Medellín, Colombia <ymgallo@gmail.com>
2 Associate Professor. Universidad Nacional de Colombia - Sede Medellín - Facultad de Ciencias- Escuela de Biociencias-Laboratorio de Microbiología Industrial. A.A. 3840, Medellín, Colombia <paguties@unal.edu.co>
3 Associate Professor. Universidad Nacional de Colombia - Sede Medellín - Facultad de Ciencias- Escuela de Biociencias-Laboratorio de- Biología Molecular y Celular. AA. 3840, Medellín, Colombia <mamarinm@unal.edu.co>
Recibido: Enero 25 de 2013; aceptado: Agosto 04 de 2013.
]]>Abstract. Potato mop-top virus (PMTV; genus Pomovirus; family Virgaviridae) is the causing agent of the spraing disease in potato (Solanum tuberosum). PMTV is transmitted by Spongospora subterranea f. sp. subterranea (Sss). This disease has a widespread distribution in potato growing regions around the world. The possibility of obtaining strain specific antibodies at low cost can greatly increase the sensitivity and use of serological tests in seed certification programs, plant breeding and quarantine regulations to avoid dissemination of this injurious virus. This work presents an alternative procedure for the production of PMTV specific antibodies useful in serological test such as ELISA and lateral flow. In contrast to standard methods requiring the isolation of viral particles or expression of recombinant capsid, this method uses peptides mimicking the N-terminal region of PMTV capsid protein as antigen for the production of specific polyclonal antibodies. The antibodies were tested against bait plants grown in soil infested with viruliferous Sss, as well as potato plants obtained from naturally Sss infested fields in Colombia. PMTV was detected in 9/14 and 24/28 foliage samples of N. benthamiana and S. phureja, respectively. In the case of field plants, the virus was detected in eight out of 12 root tissues evaluated. The minimum peptide concentration detected by ELISA was of the order of 0.1 nM.
Key words: ELISA, RT-PCR, Potato mop-top virus, Solanum tuberosum, Spongospora subterranea.
Resumen. Potato mop-top virus (PMTV; género Pomovirus; familia Virgaviridae) es transmitido por Spongospora subterranea f. sp. subterranea (Sss), agente causal de la sarna polvosa de la papa. Esta enfermedad tiene una amplia distribución en las regiones cultivadoras de papa alrededor del mundo. La posibilidad de obtener anticuerpos específicos contra cepas de este virus, puede incrementar la sensibilidad y la utilización de pruebas serológicas en programas de certificación de semilla, mejoramiento genético y regulaciones cuarentenarias que eviten su diseminación a zonas libres de la enfermedad. En esta investigación, se presenta un procedimiento alternativo para la producción de anticuerpos específicos para PMTV de utilidad en pruebas serológicas como ELISA y flujo lateral. A diferencia de los métodos estándar que requieren del aislamiento de partículas virales o de la expresión de cápside recombinante, este sistema utiliza un péptido que simula la región N-terminal de la proteína de la cápside de PMTV, como antígeno para la producción de anticuerpos policlonales. Dichos anticuerpos fueron evaluados en plantas señuelo sembradas en suelos infestados con quistosoros de Sss, así como en plantas de papa obtenidas de suelos de Colombia con infección natural de Sss. La utilización de estos anticuerpos permitió la detección del PMTV en 9/14 y 24/28 muestras foliares de N. benthamiana y S. phureja, respectivamente. Para el caso de plantas de papa obtenidas del campo, las pruebas detectaron el virus en ocho de 12 raíces evaluadas. La concentración mínima detectada del péptido por ELISA fue de 0,1 nM.
Palabras clave: ELISA, RT-PCR, Potato mop-top virus, Solanum tuberosum, Spongospora subterranea.
Potato mop-top virus (PMTV; genus Pomovirus; family Virgaviridae) (Adams et al., 2009), is the causing agent of the spraing disease in potato (Solanum tuberosum), a pathology characterized by the formation of necrotic arcs and brown flecks inside the tubers (Santala et al., 2010). PMTV is transmitted by the plasmodiophorid Spongospora subterranea f. sp. subterranea (Sss), responsible for powdery scab on potato. The resting spores of Sss remain infective in soil for years, and when hatched they release zoospores that infect plant roots or developing tubers and introduce PMTV to the plant cells (Santala et al., 2010).
PMTV alone can decrease yields in potato fields up to 25% and as high as 80% if damages associated to powdery scab are taken into account (Jones and Harrison, 1972; Guerrero, 2000). PMTV has a widespread distribution in potato growing regions around the world such as Europe, Asia, Australia and the American continent (Latvala-Kilby et al., 2009). In America, PMTV has been detected in Canada and the United States (Xu et al., 2004; Lambert et al., 2003), Peru and Bolivia (Tenorio et al., 2006), Venezuela (Ortega and Rodríguez, 2004), Costa Rica (Vásquez et al., 2006) and Colombia (Vélez, 2007).
The first report of PMTV in Colombia dates from 2007, in potato fields located in the departments of Cundinamarca and Boyacá (Vélez, 2007). The presence of PMTV was also confirmed in the department of Antioquia and Nariño by Gil et al. (2011). Several studies have revealed a dramatic increase of Sss in the principal potato-growing regions of Colombia and it is very likely that incidence of PMTV is also on the rise (Jaramillo and Botero, 2007; Carreño, 2009; Gilchrist et al., 2009; Gil et al., 2011). Unfortunately, foliar symptoms normally associated with PMTV infections are not conspicuous in Andean potato varieties being easily mistaken for physiological disorders and/or infection by other viruses (Tenorio et al., 2006). Moreover, spraing symptoms have never been observed in tubers from PMTV-infected plants, either at harvest or after storage, in this region (Tenorio et al., 2006). Therefore, reliable detection methods are required for the production of PMTV-free seed and to prevent further spread to new geographical areas.
Viral infections are commonly detected using virus-specific methods such as RT-PCR, real-time RT-PCR, immunocapture RT-PCR (IC-RT-PCR) and ELISA (Santala et al., 2010). Although RT-PCR is highly sensitive, it is also prone to false negative results due to the presence of PCR inhibitors in the RNA extracts, and to false positive results due to cross-contamination (Webster et al., 2004; Davey, 2009). The ELISA test is considered by many to be the cheapest and most convenient method for routine analysis of large sets of samples. In the case of PMTV, several mono and polyclonal antibodies have been developed using conventional techniques based on purified viral particles (Arif and Torrance, 1996; Nielsen and Mølgaard, 1997; Cerovska et al., 2006).
]]> Due to their abundance and high conservation, viral coat proteins are ideal antigens for the production of antibodies to be used in serological detection tools. Unfortunately, the purification of viruses can be a tedious and difficult task that requires expensive equipment and specialized staff. It is also common that purification procedures do not provide the virus in a completely pure form and antibodies often develop against contaminating polypeptides. These are important issues with PMTV due to the sporadic and highly irregular distribution of virions in infected plants and its presence as naked RNA (Xu et al., 2004; Wright et al., 2010). This problem could be solved with the use of recombinant proteins; unfortunately previous studies have demonstrated recombinant PMTV CP to be unstable and difficult to produce (Latvala-Kilby et al., 2009).Peptides can be used as surrogates of their parent proteins for antibody production when the amount of target protein is limited and/or not well behaved. Peptide-specific antibodies have a high probability of recognizing the parent protein either in the native or denatured state and therefore can be used reliably in different serological tests such as western-blot and ELISA. Synthetic peptides can be prepared in highly pure form and coupled to carrier proteins such as hemocyanin to produce antibodies against difficult proteins. This paper demonstrates the feasibility of using peptides mimicking the N-terminal region of PMTV capsid-protein as antigen for the production of PMTV-specific polyclonal antibodies. These antibodies were tested against bait plants grown in soil infested with viruliferous Sss, as well as potato plants obtained from naturally Sss infested fields.
MATERIALS AND METHODS
Sequence analysis and homology modeling. The best linear epitope was identified by Hoop and Woods analysis of the PMTV capsid protein of a Colombian isolate (Gil et al., 2011) using BioEdit 5.0 (Hall, 1999). The highest-scoring region was chosen using a sliding window of 15 residues. Multiple alignment of capsid proteins from members of the Virgaviridae family was performed with the program MUSCLE available at www.ebi.ac.uk (Edgar, 2004). The structure of PMTV-CP was built with MODELLER 9.2 (Sali et al., 1995), using the structure of Tobacco mosaic virus (TMV) coat protein as template (PDBid: 2TMV).
Peptide synthesis. Synthesis was performed at Genscript USA Inc (Piscataway, NJ, USA) by manual solid phase method using standard Fmoc Chemistry on 2-Cl-Trityl resin. Side chains were protected using trityl, tert-butyl or Boc group. DIC/HOBt was used as activating reagent and 20% piperidine in DMF as deprotection reagent. Ninhydrin test was employed to monitor coupling reaction. If the result was positive, then double coupling was performed and followed by capping with anhydrous acetic anhydride in DMF. After peptide chain elongation, peptide resin was treated with Reagent K and peptide was precipitated and washed three times by dry diethyl ether. The crude peptide was dissolved in 10% acetonitrile and analyzed by reverse-phase HPLC on a Shimazu 20A system using waters X-bridge column (i.d.4.6 mm x 250 mm). The peptides were purified by semi-preparative HPLC.
Antibody production. Peptide PMTV(p) was coupled to the carrier protein keyhole limpet hemocyanin (KLH). Two rabbits were immunized at GenScript on day 1, 14, 35 and 56 with 50 g peptide-KLH conjugate or KLH alone adsorbed to Complete Freund's Adjuvant (CFA) and Incomplete Freund's Adjuvant (IFA). Serum samples were collected before treatment (preimmune serum) on day 21, 42 and 63. Total IgG was affinity-purified from sera of the immunized rabbits. 27.21 mg of purified antibody were obtained, dissolved in phosphate buffered saline buffer (PBS, pH 7.4) and lyophilized until further use.
Serological tests. The titer of the purified antibody was calculated by ELISA as the reciprocal of the highest serum dilution yielding an optical density two times higher than the blank. Free PMTV peptide was used as coating agent at a concentration of 4 mg mL-1 in PBS buffer at pH 7.4. 100 mL of peptide solution were used per well. AP-conjugated Goat Anti-Rabbit IgG (H+L) was used as secondary antibody. For detection of PMTV in infected plants, 100 mg of either root or leaf tissue macerated in extraction buffer was used in a standard indirect ELISA protocol (Matthews, 1993). Leaf samples were macerated in extraction buffer containing 137 mM NaCl, 3 mM KCl, 2% PVP 24kD, 0.05% Tween 20 and 0.02% NaN3. Root tissue was macerated in extraction buffer supplemented with 1% egg albumin. Extraction buffer and 0.1 mM PMTV(p) were used as negative and positive controls, respectively. Samples with an optical density two times higher than the blank were considered positive (Matthews, 1993). Optical density was measured using a Multiscan plate reader (Labsystems®, Finlandia). Commercial Monoclonal antibodies (MAb) and alkaline phosphatase (AP)-conjugated MAbs to PMTV were purchased from Bioreba® (Reinach, Switzerland).
RT-PCR. RNA was purified from S. tuberosum roots using the RNeasy plant mini kit (Qiagen, California, USA). PMTV-CP was amplified using primers PMTV C819 (5' CTA TGC ACC AGC CCA GCG TAA CC 3') and H360 (5' CAT GAA GGC TGC CGT GAG GAA GT 3') (MacKenzie, 1996). RT-PCR was performed using a previously published protocol by Gil et al. (2011).
Bait plants. Nicotiana benthamiana and Solanum phureja plants were infected with Sss cystosori isolated from soil samples collected in the Colombian departments of Antioquia, Boyacá, Cundinamarca and Nariño. Cystosori were extracted from 1 kg of infected soil using 100 and 25 mm sieves according to the method reported by Jaramillo and Botero (2007). Bait plants were kept in a greenhouse at the Agrarian Station Paysandú located in the district of Santa Elena in the city of Medellín, Antioquia (2,550 masl, average temperature of 14 °C and average annual precipitation of 2,000 mm).
]]> Field test. Samples of Solanum tuberosum ssp. andigena cv Diacol Capiro and Puracé were collected in four fields in flowering stage at the municipality of La Unión (Antioquia). Three random root samples and two leaf specimens were collected at each potato field. The presence of PMTV was evaluated by ELISA as described above. PMTV(p) and Sss cystosori were used as positive controls. The presence of PMTV in cystosori was confirmed by RT-PCR. Results were cross-checked with a commercial DAS-ELISA PMTV detection kit from Bioreba® (ref 113075).
RESULTS AND DISCUSSION
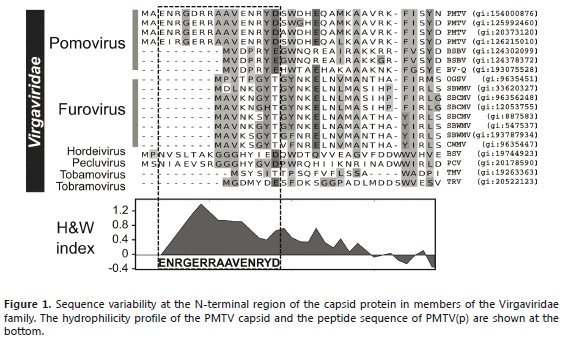
The choice of peptide antigen was based on two criteria: solvent exposure and sequence variability. There are several algorithms available to predict the sequences of potential epitopes on proteins. The most frequently used are those that identify hydrophilic clusters with high probability of being located on the protein surface. In this case, the method developed by Hoop and Woods (1981) was used. This procedure assigns a hydrophilicity score to each amino acid and then locally averages these values along the peptide chain. The point of highest local average hydrophilicity is attributed to an antigenic determinant (Hoop and Woods, 1981). The use of this method on PMTV coat-protein reveals a potential epitope at the N-terminus comprising residues 3-17, PMTV(p), with sequence NRGERRAAVENRYD (Figure 1).
The location of this peptide at the N-terminus is convenient as this region varies significantly between members of the Virgaviridae family such as the genera Pomovirus, Furovirus, Hordeivirus, Pecluvirus, Tobamovirus and Tobamovirus. Regions of high variability can be useful in the detection of strains of the same virus while conserved epitopes could be used to differentiate more distantly related taxa (Andrade et al., 2012). Sequence variants found only in certain members of a protein family can ensure the production of highly specific antibodies. A database search using Blastp and excluding PMTV sequences retrieves a putative transcriptional regulator of the GntR family from Streptomyces sp. (ZP_08451187) as the best hit with only 82% identity against an 11-residue segment of the PMTV peptide. This result suggests that this peptide is highly specific to the PMTV coat protein and will not cross-react with plant proteins.
Hydrophilicity plots should be used with care; however, when combined with structural models they can identify antigenic regions with more accuracy (Idrees and Ashfaq, 2013). Exposed sequences can be found by analyzing the surface properties of crystallographic or NMR structures of proteins and there is a higher probability that an antibody prepared against these sequences will work well in serological tests (Liang et al., 2009). Unfortunately, there are no 3D structures available for the coat protein of PMTV or any virus of the genus Pomovirus. However, a PSI-Blast search suggested that TMV, also a member of the Virgaviridae family, could be used as a template for building-up a model of the PMTV coat protein. Analysis of the antigenic regions in the resulting model, clearly suggests that this N-terminal segment is exposed to the solvent and protrudes to the exterior of the assembled virus (Figure 2). This result reinforced the idea that specific antibodies against PMTV could be produced using an N-terminal peptide mimic.
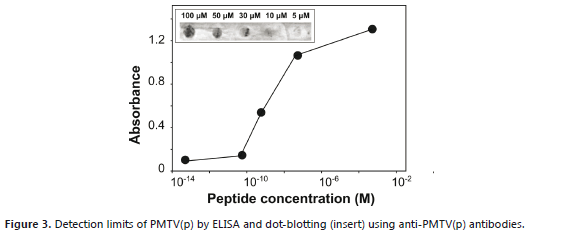
Approximately 27 mg of purified anti-PMTV(p) were obtained with a titer superior to 1:512,000 using a starting dilution of 1:1000 (1 mg mL-1) (Table 1). Antibodies sensitivity was tested by ELISA and dot blotting. The minimum peptide concentration detected by ELISA was of the order of 0.1 nM. Detection of the PMTV peptide was about 10000 times less sensitive by dot-blot, which showed a detection limit of approximately 10 mM (Figure 3). PMTV(p) was not recognized using commercial monoclonal antibodies suggesting that this peptide represents a different epitope of the PMTV capsid.
]]>
A preliminary test of the anti-PMTV(p) antibodies was performed using N. benthamiana and S. phureja baits infected with Sss cystosori isolated from soils in the departments of Antioquia, Boyacá, Cundinamarca and Nariño (Figure 4). Purified cystosori and PMTV(p) were used as positive controls. In N. benthamiana baits, PMTV was detected in nine foliage samples out of fourteen and six root specimens out of fifteen. In the case of S. phureja, twenty-four foliage samples tested positive in contrast to only 9 positive root samples out of twenty-nine. Dependence of PMTV detection on the type tissue analyzed is not surprising as this virus is characterized by a sporadic and highly irregular distribution in infected plants (Wright et al., 2010).
A field test was carried out in potato fields from the municipality of La Unión, Antioquia; a region with one of the highest incidence of powdery scab disease in Colombia (Osorio et al., 2012). In contrast to bait plants, PMTV was most frequently found in potato roots instead of leaves. ELISA was positive in eight out of twelve root samples while only one leaf sample tested positive out of eight. Commercial monoclonal antibodies gave similar results, detecting PMTV in ten root samples and none of the leaf specimens (Table 2). The higher proportion of positive samples in roots as compared to leaf samples can be explained by the role of CP-RT in virus transmission. CP-RT is expected to be more abundant in the basal portion of plants where the Sss vector is present in contrast to aerial plant parts where PMTV can be found in naked form together with movement proteins (Wright et al., 2010; Andrade et al., 2012).
Using the same plants tested by ELISA, the presence of PMTV was confirmed by RT-PCR in eleven samples. These results are in agreement with the high incidence levels of PMTV in the municipality of La Unión (Andrade et al., 2102; Osorio et al., 2012). Detection of PMTV shows slight discrepancies dependent on the method used. However, due to the non uniform distribution of PMTV, presence of the virus may not be detected unless multiple samples from the same plant are tested. Inconsistent ELISA and RT-PCR test results are common in PMTV detection and several cases have been reported in the literature (Sokmen et al., 1998; Santala et al., 2010).
This study shows an alternative procedure for the production of PMTV specific antibodies useful in serological test such as ELISA, in contrast to standard methods requiring the isolation of viral particles or expression of recombinant capsid protein. Latvala-Kilby et al. (2009) demonstrated that purified 6xHis-tagged PMTV CP is not detected by DAS-ELISA. To make matters worse, wild-type PMTV CP could not be detected with the MAb in samples from PMTV-infected potato tubers or from leaves of PMTV-infected N. benthamiana plants using Western blotting (Latvala-Kilby et al., 2009). This indicates that probably conformational changes occur in the CP epitope during both the purification and SDS-PAGE affecting the epitope recognized by the Mab used. Some of these detection problems can be overcome with the use of peptides mimicking linear epitopes of the capsid protein used for the production of antibodies. This strategy requires knowledge of a small portion of the N-terminal amino acid sequence combined with structural information. The variability of these linear epitopes can also be used to design antigens aimed at the detection of virus variants detected by standard amplification and sequencing of nucleic acids. This represents a way to update detection antibodies in the diagnosis of novel variants, which would be difficult to achieve using standard protocols for antibody production based on intact virions or purified capsid proteins.
The possibility of obtaining strain specific antibodies at low cost can greatly increase the sensitivity and use of serological tests in seed certification programs, plant breeding and quarantine regulations to avoid dissemination of injurious plant diseases within and outside the country. Otherwise, the use of symptomless, PMTV-infected seed tubers could possess a risk of contaminating new fields with the virus. Unfortunately, the erratic distribution of the virus still represents one of the biggest challenges in confirming and verifying the accuracy of the test results. It would be interesting to evaluate our antibodies in more detailed studies of PMTV incidence in the principal potato growing regions of Colombia and other Andean countries. Future work should also test the performance of anti-PMTV(p) antibodies in lateral flow immunoassays and determine their suitability for routine detection of PMTV on tubers.
CONCLUSIONS
]]> Using PMTV-specific polyclonal antibodies raised against a synthetic peptide mimicking the N-terminal region of PMTV capsid-protein, allows the detection of the virus in both N. benthamiana and S. phureja bait plants that had been previously inoculated with Sss cytosori. In S. tuberosum plants obtained directly from the field, PMTV was detected more frequently in roots than in leaves. This result agrees with the sporadic and highly irregular distribution of PMTV in infected plants.This study demonstrates the viability of using synthetic peptides for the production of antibodies specific to local variants of plant viruses. These antibodies can be employed in serological tests such as ELISA, dot-blot and lateral flow. Future work should address the effect of point mutations on both sensitivity and specificity of anti-PMTV antibodies.
ACKNOWLEDGMENTS
This study was funded by Ministerio de Agricultura y Desarrollo Rural de Colombia, Grant 090-2007S4527-87-08. We thank José Fernando Gil and Inés Osorio for providing Sss inoculated N. benthamiana plants.
BIBLIOGRAPHY
Adams, M.J., J.F. Antoniw and J. Kreuze. 2009. Virgaviridae: a new family of rod-shaped plant viruses. Archives of Virology 154(12): 1967-1972. [ Links ]
Andrade, D., Y. Gallo, P. Gutiérrez and M. Marín. 2012. Polyclonal antibodies to detect the CP-RT protein of potato mop-top virus. Revista Facultad Nacional de Agronomía Medellín 65(2): 6673-6686. [ Links ]
Arif, M. and L. Torrance. 1996. Detection of potato mop-top virus in potato tubers. Proceedings Crop Protection in Northern Britain 1: 325-330. [ Links ]
Carreño, A.J. 2009. Evaluación de la variabilidad genética de Spongospora subterranea f. sp. subterranea mediante la comparación de regiones ITS del ADN ribosomal de cepas procedentes de las regiones productoras de papa en Colombia. Tesis Magíster en Ciencias Agrarias. Facultad de Agronomía. Universidad Nacional de Colombia. Bogotá. 104 p. [ Links ]
Cerovská, N, M. Filigarová and T. Pecenkevá. 2006. Production of polyclonal antibodies to a recombinant potato mop-top virus non-structural triple gene block protein 1. Journal of Phytopathology 154(7): 422-427. [ Links ]
Davey, T. 2009. The importance of potato mop-top virus (PMTV) in Scottish seed potatoes. Doctoral Theses (Life Sciences). Heriot Watt University & Science and Advice for Scottish Agriculture, Edinburgh, Scotland, UK. 178 p. [ Links ]
Edgar, R.C. 2004. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research 32(5): 1792-1797. [ Links ]
Gil, J.F, P.A. Gutiérrez, J.M. Cotes, E.P. González and M. Marín. 2011. Caracterización genotípica de aislamientos colombianos del potato mop-top virus (PMTV, Pomovirus). Actualidades Biológicas 33(94): 69-84. [ Links ]
Gilchrist, E., S. Jaramillo y S. Reynaldi. 2009. Efecto sobre la sarna polvosa de cuatro aislamientos del hongo Trichoderma asperellum en tres tipos de suelo. Revista Facultad Nacional de Agronomía Medellín 62(1): 4783-4792. [ Links ]
Guerrero, O. 2000. La roña o sarna polvosa en el departamento de Nariño. pp. 127-129. En: Fedepapa (ed.). Papas colombianas 2000, con el mejor entorno ambiental. Fedepapa, Bogotá [ Links ].
Hall, T.A. 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series 41: 95-98. [ Links ]
Hoop, T.P. and R.K. Woods. 1981. Predictions of protein antigenic determinants from amino acid sequences. Proceedings of the National Academy of Sciences of the United States of America 78(6): 3824-3828. [ Links ]
Idrees, S. and U.A. Ashfaq. 2013. Structural analysis and epitope prediction of HCV E1 protein isolated in Pakistan: an in-silico approach. Virology Journal 10(113): 1-8. [ Links ]
Jaramillo, S. y J.M. Botero. 2007. Respuesta de diferentes poblaciones de Spongospora subterranea f. sp. subterranea a la rotación entre dos cultivares de papa (Solanum tuberosum ssp. andigena). Revista Facultad Nacional de Agronomía Medellín 60(2): 3859-3876. [ Links ]
Jones, R.A.C. and B.D. Harrison. 1972. Ecological studies on potato mop-top virus in Scotland. Annals of Applied Biology 71(1): 47-57. [ Links ]
Lambert, D.H., L. Levy, V.A. Mavrodieva, S.B. Jonson, M.J. Babcock and M.E. Vayda. 2003. First Report of potato mop-top virus on potato from the United States. Plant Disease 87(7): 872. [ Links ]
Latvala-Kilby, S., J.M. Aura, N. Pupola, A. Hannukkala and J.P.T. Valkonen. 2009. Detection of potato mop-top virus in potato tubers and sprouts: combinations of RNA2 and RNA3 variants and incidence of symptomless infections. Phytopathology 99(5): 519-531. [ Links ]
Liang, S., D. Zheng, C. Zhang and M. Zacharias. 2009. Prediction of antigenic epitopes on protein surfaces by consensus scoring. BMC Bioinformatics 10(302): 1-10. [ Links ]
MacKenzie, D.J. 1996. Detection of potato mop-top virus in leaf or tuber tissue by reverse transcription-polymerase chain reaction. Document CPHBT96K03, Centre for Plant Health, Agriculture and Agri-Food Canada, Sidney, B.C., Canada. [ Links ]
Matthews, R.E.F. 1993. Diagnosis of plant virus diseases. CRC Press, Boca Raton, FL. 374 p. [ Links ]
Nielsen, S.L. and J.P. Mølgaard. 1997. Incidence, appearance and development of potato mop-top furovirus-induced sprain in potato cultivars and the influence on yield, distribution in Denmark and detection of the virus in tubers by ELISA. Potato Research 40(1): 101-110. [ Links ]
Ortega, E. y Y. Rodríguez. 2004. Virus del mop top: una amenaza para la producción de papas en Venezuela. INIA Divulga 1: 36-40. [ Links ]
Osorio, I., P. Gutiérrez y M. Marín. 2012. Revisión: Spongospora subterranea f. sp. subterranea y su virus asociado potato mop-top virus (PMTV), dos patógenos reemergentes en los cultivos de papa de Colombia. Revista Facultad Nacional de Agronomía Medellín 65(1): 6361-6378. [ Links ]
Santala, J., O. Samuilova, A. Hannukkala, S. Latvala, H. Kortemaa, U. Beuch, A. Kvarnheden, P. Persson, K. Topp, K. Ørstad, C. Spetz, S.L. Nielsen, H.G. Kirk, M. Budziszewska, P. Wieczorek, A. Obrepalska-Steplowska, H. Pospieszny, A. Kryszczuk, J. Sztangret-Wisniewska, Z. Yin, M. Chrzanowska, E. Zimnoch-Guzowska, E. Jackeviciene, L. Taluntyte, N. Pupola, J. Mihailova, I. Lielmane, L. Järvekülg, K. Kotkas, E. Rogozina, A. Sozonov, I. Tikhonovich, P. Horn, I. Broer, S. Kuusiene, J. Staniulis, J.G. Uth, G. Adam and J.P.T. Valkonen. 2010. Detection, distribution and control of potato mop-top virus, a soil-borne virus, in northern Europe. Annals of Applied Biology 157(2):163-178. [ Links ]
Sali, A., L. Potterton, F. Yuan, H. van Vlijmen and M. Karplus. 1995. Evaluation of comparative protein modelling by MODELLER. Proteins 23(3): 318-326. [ Links ]
Sokmen, M.A., H. Barker and L. Torrance. 1998. Factors affecting the detection of potato mop-top virus in potato tubers and improvement of test procedures for more reliable assays. Annals of Applied Biology 133(1): 55-63. [ Links ]
Tenorio, J., Y. Franco, C. Chuquillanqui, R.A. Owens and L.F. Salazar. 2006. Reaction of potato varieties to potato mop-top virus infection in the Andes. American Journal of Potato Research 83(5): 423-431. [ Links ]
Vásquez, V., M. Montero-Astúa y C. Rivera. 2006. Incidencia y distribución altitudinal de 13 virus en cultivos de Solanum tuberosum (Solanaceae) en Costa Rica. Revista de Biología Tropical 54(4): 1135-1141. [ Links ]
Vélez, B. 2007. Detección e identificación del potato mop-top virus (PMTV) en áreas de producción de papa donde se encuentra Spongospora subterranea en dos departamentos de Colombia. Tesis Magíster en Ciencias Agrarias. Facultad de Agronomía. Universidad Nacional de Colombia. Bogotá. 115 p. [ Links ]
Webster, C.G., S.J. Wylie and M.G. Jones. 2004. Diagnosis of plant viral pathogens. Current Science 86(12): 1604-1607 [ Links ]
Wright, K.M., G.H. Cowan, N.I. Lukhovitskaya, J. Tilsner, A.G. Roberts, E.I. Savenkov and L. Torrance. 2010. The N-terminal domain of PMTV TGB1 movement protein is required for nucleolar localization, microtubule association, and long-distance movement. Molecular Plant Microbe Interaction 23(11): 1486-1497. [ Links ]
Xu, H., T.L. Dehaan and S.H. De Boer. 2004. Detection and confirmation of potato mop-top virus in potatoes produced in the United States and Canada. Plant Disease 88(4): 363-367. [ Links ]
]]>