Eficacia del aprepitant para la prevención de la náusea y el vómito postoperatorios. Revisión sistemática y metaanálisis de ensayos clínicos
Efficacy of aprepitant for prevention of postoperative nausea and vomiting. Systematic review and meta-analysis of randomized clinical trials
Eficácia do aprepitanto (Emend) para a prevenção da náusea e vómito pós-operatórios. Revisão sistemática e meta-análises de ensaios clínicos
Marta Inés Berrío Valencia1; Juan Pablo Botero Aguirre2
]]>1 Médica anestesióloga, M. Sc., en Epidemiología, Hospital Pablo Tobón Uribe, Medellín, Colombia. martaberrio@gmail.com
2 Químico Farmacéutico, M. Sc., en Epidemiología, Hospital Pablo Tobón Uribe, Medellín, Colombia.
Recibido: noviembre 02 de 2013
Aceptado: marzo 04 de 2014
RESUMEN
]]> Objetivo: evaluar la eficacia del aprepitant, comparada con la de otros antieméticos, para la prevención de la náusea y el vómito postoperatorios en adultos que recibieron anestesia general.Métodos: revisión sistemática de ensayos clínicos con asignación aleatoria que evaluaron comparativamente la eficacia del aprepitant con la de otros antieméticos para la prevención de la náusea y el vómito postoperatorios, el uso de antiemético de rescate y los efectos adversos. La búsqueda se hizo en The Cochrane Library, EBSCO, Embase, LILACS, OVID, PubMed, SciELO, ScienceDirect, Scopus y Google Scholar. Se definió la heterogeneidad con la Q de Cochran y el estadístico I2, se usó el modelo de efectos fijos y aleatorios, se empleó el método de Mantel- Haenszel con el riesgo relativo de cada desenlace y su respectivo intervalo de confianza al 95%.
Resultados: se encontró diferencia significativa a favor del aprepitant para la prevención del vómito a las 24 (RR 0,52; IC 95%: 0,38-0,7) y a las 48 horas (RR 0,51; IC 95%: 0,39-0,67), pero no de la náusea a las 24 horas (RR 1,16; IC 95%: 0,85-1,6).
Conclusiones: el aprepitant previene el vómito postoperatorio, pero no la náusea, a las 24 y 48 horas.
PALABRAS CLAVE
Antieméticos, Aprepitant, Metaanálisis, Náusea y Vómito Postoperatorios
SUMMARY
Objective: To evaluate the efficacy of aprepitant compared with other antiemetics for the prevention of postoperative nausea and vomiting in adults who underwent general anesthesia.
Methods: Systematic review of randomized clinical trials with meta-analysis, that evaluated the efficacy of aprepitant in comparison with other antiemetics for the prevention of postoperative nausea and vomiting, antiemetic rescue and adverse effects. The search was done in The Cochrane Library, EBSCO, EMBASE, LILACS, OVID, PubMed, SciELO, ScienceDirect, Scopus and Google Scholar. Heterogeneity was defined with the Cochran Q and I2 statistic; the model of fixed and random effects, and the Mantel-Haenszel method for relative risk of each outcome and its respective confidence interval 95% were used.
Results: There was significant difference in favor of aprepitant for the prevention of vomiting at 24 (RR 0.52; 95% CI: 0.38-0.7) and at 48 hours (RR 0.51; 95% CI: 0.39 to 0.67), but not for nausea at 24 hours (RR 1.16; 95% CI: 0.85-1.6).
]]> Conclusions: Aprepitant prevents postoperative vomiting, but not nausea, at 24 and 48 hours.KEY WORDS
Antiemetics, Aprepitant, Meta-analysis, Postoperative Nausea and Vomiting
RESUMO
Objetivo: avaliar a eficácia do aprepitanto, comparada com a de outros antieméticos, para a prevenção da náusea e vómito pós-operatórios em adultos que receberam anestesia geral.
Métodos: revisão sistemática de ensaios clínicos com atribuição aleatória que avaliaram comparativamente a eficácia do aprepitanto com a de outros antieméticos para a prevenção da náusea e vómito pós-operatórios, o uso de antiemético de resgate e os efeitos adversos. A busca se fez em The Cochrane Library, EBSCO, Embase, LILACS, OVID, PubMed, SciELO, ScienceDirect, Scopus e Google Scholar. Definiu-se a heterogeneidade com o Q de Cochran e o estatístico I2, usou-se o modelo de efeitos fixos e aleatórios, empregou- se o método de Mantel-Haenszel com o risco relativo de cada desenlace e seu respectivo intervalo de confiança a 95%.
Resultados: encontrou-se diferença significativa a favor do aprepitanto para a prevenção do vómito às 24 (RR 0,52; IC 95%: 0,38-0,7) e às 48 horas (RR 0,51; IC 95%: 0,39-0,67), mas não da náusea às 24 horas (RR 1,16; IC 95%: 0,85-1,6). Conclusões: o aprepitanto previne o vómito pós -operatório, mas não a náusea, às 24 e 48 horas.
PALAVRAS IMPORTANTES
Antieméticos, Aprepitanto, Meta-análises, Náusea e vómito pós-operatórios
]]>
INTRODUCCIÓN
La náusea y el vómito postoperatorios (NVPO) son dos de los efectos secundarios más indeseables de la anestesia general y pueden ser, incluso, más incómodos para los pacientes que el dolor postoperatorio (1). Su fisiopatología no se ha comprendido completamente (2). El pequeño gran problema (the big little problem), como algunos autores han calificado la NVPO, sigue siendo una dificultad en el perioperatorio, en especial en la cirugía de corta estancia y se ha posicionado como un marcador de calidad en anestesiología (3,4).
Los pacientes con NVPO se demoran una hora más en la unidad de cuidado postanestésico y el tiempo de enfermería es mayor con un aumento en el costo de la recuperación postoperatoria (5). Sin profilaxis, hasta 30% de los pacientes con anestesia general presentan NVPO (6). Cuando un paciente tiene múltiples factores de riesgo la incidencia aumenta hasta 70% a 80% (6-8). La NVPO impacta negativamente la morbilidad y la satisfacción del paciente y es la causa más común de readmisión hospitalaria en las cirugías ambulatorias (9,10). Aunque generalmente se presentan eventos no fatales y autolimitados, el control inadecuado de la NVPO puede traer complicaciones serias como deshidratación, trastornos hidroelectrolíticos, hipertensión venosa, dehiscencia de suturas, sangrado, hematoma (11), ceguera (12), aspiración (13), compromiso de la vía aérea, ruptura esofágica (síndrome de Boerhaave) y hasta enfisema (14).
La NVPO puede ocurrir en el postoperatorio inmediato o más tardíamente y en la actualidad la náusea y el vómito postoperatorios después del alta (NVPA) han ganado interés entre el personal de la salud porque hasta 36% de quienes la presentan niegan haber tenido episodios de NVPO en el postoperatorio inmediato (15); además, la NVPA causa retardo en reasumir las actividades cotidianas y un tiempo de recuperación más prolongado (15).
Los cuatro factores de riesgo para la NVPO son: el sexo femenino, el antecedente de NVPO o cinetosis, ser no fumador y el uso postoperatorio de opioides (8). Con un factor predictor el riesgo de NVPO es del 10%; con dos, tres y cuatro factores, del 39%, 61% y 79%, respectivamente (8). Este modelo fue desarrollado para el paciente adulto bajo anestesia general con halogenados (8).
Los antagonistas del receptor 5-hidroxitriptamina tipo 3 (5-HT3) han permanecido como el principal tratamiento, pero ahora el manejo multimodal y los antagonistas del receptor de la neuroquinina 1 (NK-1, por su sigla en inglés) han ganado importancia principalmente en los pacientes con alto riesgo de NVPO (9), en cuya profilaxis se recomienda la terapia multimodal, que consiste en administrar dos o más medicamentos de diferentes grupos con el fin de lograr un efecto aditivo (10). Los antagonistas del receptor NK-1 son la clase más reciente de antieméticos; de ellos, el aprepitant es la primera formulación disponible y se plantea su uso en los pacientes con alto riesgo de náusea y vómito, como los que reciben quimioterapia en quienes se han hecho muchos estudios (10,16-18).
Existe un metaanálisis de varios antieméticos para la profilaxis de la NVPO (19), que no incluye los antagonistas del receptor NK-1, y se debe actualizar la revisión sistemática existente sobre el aprepitant (20) ya que se dispone de nuevas evidencias (21-25); además, no se ha comparado sistemáticamente su eficacia con la de otros antagonistas del receptor de la NK-1 y existe controversia para la prevención de la náusea postoperatoria con el aprepitant (22), por lo que se llevaron a cabo una nueva revisión sistemática y un metaanálisis.
MATERIALES Y MÉTODOS
]]> Protocolo y registroEl protocolo de la revisión sistemática fue registrado en PROSPERO 2013: CRD42013005005. Disponible en http://www.crd.york.ac.uk/PROSPERO/display_record.asp?ID=CRD42013005005
Criterios de elegibilidad
Los estudios elegibles fueron ensayos clínicos controlados con asignación aleatoria que evaluaron la eficacia clínica para la prevención de la NVPO y las reacciones adversas producidas por el aprepitant comparado con otros antieméticos en pacientes adultos que recibieron anestesia general. Se usó la estrategia PICOS (por la sigla en inglés de patient, intervention, comparison, outcome, strategy), así:
P: pacientes adultos con riesgo de náusea y vómito postoperatorios que recibieron anestesia general.
I: aprepitant profiláctico
C: otros antieméticos
O: 1. Incidencia de náusea a las 24 horas del postoperatorio
2. Incidencia de vómito a las 24 y 48 horas del postoperatorio
3. Necesidad de antiemético de rescate a las 24 horas del postoperatorio
]]> 4. Efectos adversos del aprepitantS: ensayos clínicos controlados
Fuentes de información
Se hizo la búsqueda de los estudios en las siguientes bases de datos: The Cochrane Library, EBSCO, Embase, LILACS, OVID, National Library of Medicine's Pubmed database, SciELO, ScienceDirect, Scopus y Google Scholar. También se consultaron las bases de registro de ensayos controlados The Cochrane Central y clinicaltrials. gov. Para identificar estudios adicionales se llevó a cabo una búsqueda manual en la lista de referencias de las publicaciones elegidas. Se complementó con una búsqueda en bola de nieve y en fuentes de literatura gris y se hizo contacto con clínicos expertos.
Búsqueda
Se usó la siguiente terminología Medical Subject Headings (MeSH): postoperative nausea and vomiting; aprepitant; receptors, neurokinin-1 antagonism; clinical trials. La estrategia de búsqueda en la National Library of Medicine's Pubmed database por MEDLINE fue:
(''aprepitant''[Supplementary Concept] OR ''aprepitant'' [All Fields]) AND (''ondansetron'' [MeSH Terms] OR ''ondansetron'' [All Fields])
''aprepitant''[Supplementary Concept] OR ''aprepitant'' [All Fields]) AND (''ondansetron'' [MeSH Terms] OR ''ondansetron'' [All Fields]) AND (''random allocation'' [MeSH Terms] OR (''random'' [All Fields] AND ''allocation'' [All Fields]) OR ''random allocation'' [All Fields] OR ''randomized''[All Fields])
((''receptors, neurokinin-1''[MeSH Terms] OR (''receptors'' [All Fields] AND ''neurokinin-1'' [All Fields]) OR ''neurokinin- 1 receptors'' [All Fields] OR ''neurokinin 1 receptor'' [All Fields]) AND antagonist [All Fields]) AND (''ondansetron'' [MeSH Terms] OR ''ondansetron'' [All Fields])
((''receptors, neurokinin-1'' [MeSH Terms] OR (''receptors'' [All Fields] AND ''neurokinin-1'' [All Fields]) OR ''neurokinin-1 receptors'' [All Fields] OR ''neurokinin 1 receptor'' [All Fields]) AND antagonism [All Fields]) AND (''nausea'' [MeSH Terms] OR ''nausea'' [All Fields])
]]> (''aprepitant'' [Supplementary Concept] OR ''aprepitant'' [All Fields]) AND (''postoperative nausea and vomiting'' [MeSH Terms] OR (''postoperative'' [All Fields] AND ''nausea'' [All Fields] AND ''vomiting'' [All Fields]) OR ''postoperative nausea and vomiting'' [All Fields] OR (''postoperative'' [All Fields] AND ''nausea'' [All Fields]) OR ''postoperative nausea'' [All Fields])Los filtros fueron: custom date range (1990 to 2013- 07), species (humans), article types (randomized controlled trial) y ages (Adult: 18+ years).
Selección de los estudios
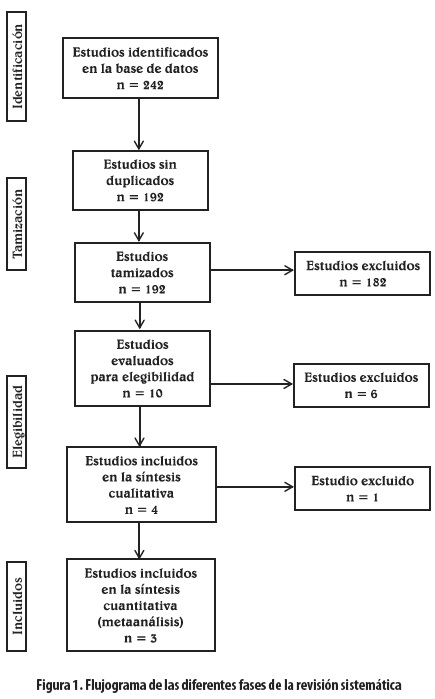
Los estudios incluidos en el metaanálisis se detallan en el flujograma de la figura 1 y los excluidos (17,21-33), así como los motivos de exclusión, se presentan en la tabla 1.
Proceso de extracción de datos
Los autores (MIBV y JPBA) de forma independiente fueron los encargados de evaluar los títulos y resúmenes para excluir los estudios que no cumplieran los criterios de inclusión, previa eliminación de los estudios duplicados. No se hizo cegamiento de los evaluadores respecto a la procedencia de los artículos como nombres de los autores, afiliaciones, revista de publicación y resultados de los estudios. Se resolvieron por consenso las discrepancias entre los investigadores. Se obtuvieron los textos completos de todos los artículos que ameritaran ser revisados a la luz de los criterios de inclusión, título, resumen o ambos. Se contactó a los autores vía email en caso de requerir alguna información.
Lista de datos
La información recolectada fue la siguiente: autor principal, año de publicación, nombre de la revista, nombre del artículo, patrocinadores, lenguaje de publicación, realizado en un centro o multicéntrico, criterios de inclusión y exclusión de los participantes, desenlaces, total de participantes, pérdidas, tiempo de seguimiento de los pacientes, tiempo de duración del estudio, dosis y tiempo de administración del aprepitant, dosis del antiemético de control, tiempo y vía de administración del antiemético de control y cointervenciones. Los desenlaces primarios fueron: náusea a las 24 horas del postoperatorio y vómito a las 24 y 48 horas del postoperatorio. Los desenlaces secundarios fueron: antieméticos de rescate a las 24 horas del postoperatorio y efectos adversos.
Riesgo de sesgo en los estudios individuales
]]> Se utilizaron la lista de chequeo del Consolidated Standards of Reporting Trials (CONSORT) para evaluar los criterios de calidad estructural de los estudios (34) y la escala de Jadad para evaluar la calidad del contenido (35). Se evaluó el riesgo de sesgos de los estudios incluidos con la herramienta de The Cochrane Collaboration.Medidas de resumen
La medida resumen del efecto de la intervención fue una razón de riesgos relativos (RR) con su respectivo intervalo de confianza (IC) al 95% calculada con el método de Mantel-Haenszel. Se reportó el número necesario a tratar (NNT) con su respectivo IC al 95% como medida de eficacia para la prevención del vómito postoperatorio a las 24 y a las 48 horas.
Síntesis de resultados
Se hicieron gráficos de bosque (forest plot) estratificados según el tamaño de los estudios para examinar los sesgos potenciales que pudieran afectar los resultados. Se identificó la heterogeneidad de los estudios incluidos con el índice I2 en el que los valores de 25%, 50% y 75% corresponden a heterogeneidad baja, mediana y alta, respectivamente; además, se usó chi2 con n-1 grados de libertad y un alfa menor de 0,1 para la significancia estadística. Para el metaanálisis se usaron los modelos de efectos fijos y de efectos aleatorios de acuerdo con el grado de heterogeneidad encontrado. Los paquetes estadísticos utilizados fueron Review Manager (RevMan) versión 5.2 (Copenhaguen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2012) y Stats- Direct Software Versión. 2.7.9 (9 July 2012) (StatsDirect Ltd, England) para el IC 95% de I2 y la prueba de Egger.
Riesgo de sesgo entre los estudios
Debido a los pocos estudios incluidos en el metaanálisis no se pudo hacer la prueba de Egger y se decidió no realizar el gráfico de embudo (funnel plot).
Análisis adicionales
Se hicieron análisis de subgrupos para el efecto de la dosis de aprepitant utilizada sobre los resultados. Se efectuaron los análisis de sensibilidad con el fin de explorar el origen de la heterogeneidad y se excluyeron los estudios de más peso para establecer su efecto dominante sobre los resultados.
]]> RESULTADOS
De los diez estudios analizados, se excluyeron cuatro que no permitían comparar directamente el aprepitant con otro antiemético (22-24,33); se excluyó otro por no tener control (32) y uno más por ser el control placebo (21). Quedaron cuatro estudios, de los cuales uno se excluyó por su baja calidad estructural con el CONSORT y metodológica con un puntaje de Jadad de 2 (25). Los tres estudios remanentes compararon el aprepitant con el ondansetron para un total de 1.803 pacientes (36-38). Dos estudios fueron multicéntricos en diferentes países (36,37), y el otro fue de un centro único en Estados Unidos (38). Todos los estudios tuvieron alta calidad estructural por CONSORT y metodológica por puntaje de 5 de Jadad. Los tres estudios fueron publicados en inglés, con un seguimiento de 48 horas de los pacientes ingresados y fueron financiados por Merck and Co., Inc. Dos de ellos se hicieron en pacientes con cirugía abdominal abierta; el otro fue en pacientes neuroquirúrgicos y tuvo como cointervención la dexametasona para disminuir el edema cerebral (38). Todos los estudios se llevaron a cabo en pacientes con riesgo alto de NVPO a la luz de los criterios de inclusión y la mayoría de los pacientes eran de sexo femenino con un promedio de edad en la quinta década de la vida para todos los estudios. No hubo diferencias estadísticamente significativas en los antecedentes de NVPO, cinetosis o tabaquismo entre los diferentes grupos de los estudios. Ninguno de estos presentó pérdidas mayores del 10% durante la ejecución. La medicación de los grupos de tratamiento (aprepitant) en cada ensayo clínico con asignación aleatoria (ECA) se administró en diferentes esquemas de dosificación: 40 mg y 125 mg en dosis únicas que se compararon con ondansetron 4 mg en dosis única. La tabla 2 muestra las características de los estudios incluidos.
Se encontraron tres ECA (1.803 participantes) que compararon el aprepitant con el ondansetron. Solo un estudio, con un total de 107 participantes, clasificó para hacer el forest plot de náusea a las 24 horas, y no se encontró diferencia estadísticamente significativa entre la formulación de aprepitant 40 mg y ondansetron 4 mg con un RR 1,16 (IC 95%: 0,85-1,6) (figura 2).
Se encontró que el aprepitant 40 mg previene el vómito a las 24 horas del postoperatorio comparado con el ondansetron 4 mg con un RR 0,52 (IC 95%: 0,38-0,7) (figura 3); no se encontró heterogeneidad: I2 0% y chi2 0,61, df 1 (p = 0,43) por lo que se usó el modelo de efectos fijos. Para el vómito a las 48 horas del postoperatorio, se encontró que el aprepitant 40 mg lo previene con un RR 0,51 (IC 95%: 0,39-0,67), comparado con el ondansetron 4 mg (figura 4); no se encontró heterogeneidad: I2 0% y chi2 0,29, df 1 (p = 0,59) por lo que se usó el modelo de efectos fijos. En cuanto al antiemético de rescate a las 24 horas no hubo diferencias entre el grupo de aprepitant 40 mg y el de ondansetron 4 mg con un RR 1,02 (IC 95%: 0,88-1,19); se encontró heterogeneidad moderada: I2 33%; (IC 95%: 0%-80,9%) y chi2 2,99, df 2 (p = 0,22) por lo que se usó el modelo de efectos aleatorios.
El análisis por subgrupos de acuerdo con la dosis administrada de aprepitant tampoco afectó la necesidad de antiemético de rescate a las 24 horas; se hizo con aprepitant a la dosis de 40 mg y posteriormente con dosis de 125 mg. No hubo diferencias entre el grupo de aprepitant 125 mg y el control: RR 0,98 (IC 95%: 0,86-1,12) y no se encontró heterogeneidad: I2 0% y chi2 0,09, df 1 (p = 0,76).
El análisis de sensibilidad mostró cómo incluso retirando el estudio de mayor peso y luego el de menor peso, no se afectó la estimación final combinada del efecto para vómito postoperatorio a las 24 y 48 horas ni el resultado del antiemético de rescate a las 24 horas del postoperatorio. Los efectos adversos (constipación, pirexia y QT mayor de 60 milisegundos) con aprepitant 40 mg fueron menos que con el ondansetron: RR 0,75 (IC 95%: 0,58-0,96); no se encontró heterogeneidad: I2 0%, chi2 0,03, df 1 (p = 0,86). Con dosis de 125 mg también fueron menos los efectos adversos comparados con el ondansetron: RR 0,68 (IC 95%: 0,48-0,97); se encontró heterogeneidad moderada: I2 43% y chi2 1,75, df 1 (p = 0,19).
La diferencia de riesgo de vómito a las 24 horas fue de 0,14 (IC 95%: 0,08-0,20), con un NNT del aprepitant 40 mg para prevenir vómito a las 24 horas del postoperatorio de 7,04 (IC 95%: 4,9-12,4). La diferencia de riesgo de vómito a las 48 horas fue de 0,166 (IC 95%: 0,10-0,20) con un NNT de 6 (IC 95%: 4,34-9,75). No se encontraron estudios que compararan el aprepitant con otros antagonistas del receptor de la NK-1, específicamente: fosaprepitant, casopitant, rolapitant y neuropitant.
DISCUSIÓN
El aprepitant previene el vómito a las 24 horas y 48 horas del postoperatorio. En la mayoría de los estudios se comparó con el ondansetron, pero hay que tener en cuenta que la administración de este último fue antes de la inducción (36,37), lo que puede generar sesgo por no ser el período en que se ha evaluado su mejor eficacia (8,39,40). Además, la vida media larga del aprepitant comparada con la del ondansetron favorece su efecto antiemético más prolongado: el aprepitant tiene una vida media de 9-13 horas (16,17) y la del ondansetron es de 4-6 horas (41); probablemente la mejor comparación sería con el palonosetron, como el antagonista de serotonina con vida media larga (42- 44), pero hasta el momento no hay estudios que comparen estos dos medicamentos. El aprepitant mostró un buen perfil de seguridad al compararlo con el ondansetron probablemente debido al aumento de la potencia con el metaanálisis. El NNT del aprepitant 40 mg es de 7 para prevenir vómito a las 24 horas comparado con el ondansetron 4 mg en pacientes con alto riesgo de NVPO; este resultado se debe evaluar en términos económicos al no mostrar superioridad notoria. Sin embargo, no se encontraron diferencias con el empleo de antieméticos de rescate hasta las 24 horas del postoperatorio. La actualización de las guías para el tratamiento de la NVPO menciona los antagonistas del receptor NK-1 y, aunque el aprepitant cuenta con el mayor número de estudios dentro de este grupo, la experiencia clínica aún es limitada y no se ha establecido su uso como profilaxis rutinaria (45).
]]> Las limitaciones de este trabajo se deben a los pocos estudios actuales que comparan el aprepitant como profilaxis de la NVPO y a la baja calidad de algunos de ellos, que no permitió su inclusión en el metaanálisis; además, puede existir sesgo de publicación por ser estudios financiados por una casa farmacéutica y así tener la tendencia a publicar resultados positivos; por otra parte, son estudios en pacientes con riesgo alto de NVPO que deberían tener la combinación de dos o tres antieméticos (8,40).El alto costo del aprepitant puede ser un factor que disminuya su uso en nuestro medio y se requerirían estudios de costo-efectividad para evaluar su aplicación en pacientes seleccionados. El costo aproximado del aprepitant 40 mg es de $55.000 pesos colombianos (COP) y el del vial de palonosetron, de $107.000 COP; en contraste, la ampolla de ondansetron de 4 mg cuesta $4.000 COP. pero hay que tener en cuenta el costo de la hora en la unidad de recuperación postanestésica, que es de aproximadamente $55.000 COP y los costos de las complicaciones potenciales de la NVPO.
Aunque se requieren más estudios clínicos con el aprepitant y su alto costo limita su uso en nuestro medio, podría estar justificado en pacientes con alto riesgo de NVPO que tengan alergia a varios antieméticos profilácticos, y en aquellos con el antecedente de NVPO a pesar de recibir profilaxis multimodal con dexametasona, haloperidol y ondansetron. Ambas situaciones están mucho más indicadas en cirugías en las que es muy deseable evitar estos eventos. Además, cuando se tome conciencia de la NVPA y se inicie su registro, probablemente se empleará más el aprepitant como profilaxis.
En conclusión, se puede utilizar el aprepitant para la profilaxis del vómito a las 24 y 48 horas del postoperatorio en pacientes de alto riesgo, pero hacen falta más estudios para establecer su dosis efectiva 95 para la prevención de la NVPO y en poblaciones especiales como la pediátrica.
REFERENCIAS BIBLIOGRÁFICAS
1. Macario A, Weinger M, Carney S, Kim A. Which clinical anesthesia outcomes are important to avoid? The perspective of patients. Anesth Analg. 1999 Sep;89(3):652–8. [ Links ]
2. Watcha MF, White PF. Postoperative nausea and vomiting. Its etiology, treatment, and prevention. Anesthesiology. 1992 Jul;77(1):162–84. [ Links ]
]]>3. Gold BS, Kitz DS, Lecky JH, Neuhaus JM. Unanticipated admission to the hospital following ambulatory surgery. JAMA. 1989 Dec 1;262(21):3008–10. [ Links ]
4. Philip BK. Patients' assessment of ambulatory anesthesia and surgery. J Clin Anesth. 1992;4(5):355–8. [ Links ]
5. Gan T, Sloan F, Dear G de L, El-Moalem HE, Lubarsky DA. How much are patients willing to pay to avoid postoperative nausea and vomiting? Anesth Analg. 2001 Feb;92(2):393–400. [ Links ]
6. Apfel CC, Korttila K, Abdalla M, Kerger H, Turan A, Vedder I, et al. A factorial trial of six interventions for the prevention of postoperative nausea and vomiting. N Engl J Med. 2004 Jun 10;350(24):2441–51. [ Links ]
7. Apfel CC, Kranke P, Eberhart LHJ, Roos A, Roewer N. Comparison of predictive models for postoperative nausea and vomiting. Br J Anaesth. 2002 Feb;88(2):234–40. [ Links ]
]]>8. Apfel CC, Läärä E, Koivuranta M, Greim CA, Roewer N. A simplified risk score for predicting postoperative nausea and vomiting: conclusions from cross-validations between two centers. Anesthesiology. 1999 Sep;91(3):693–700. [ Links ]
9. Le TP, Gan TJ. Update on the management of postoperative nausea and vomiting and postdischarge nausea and vomiting in ambulatory surgery. Anesthesiol Clin. 2010 Jun;28(2):225–49. [ Links ]
10. Jokinen J, Smith AF, Roewer N, Eberhart LHJ, Kranke P. Management of postoperative nausea and vomiting: how to deal with refractory PONV. Anesthesiol Clin. 2012 Sep;30(3):481–93. [ Links ]
11. Watcha MF, White PF. New antiemetic drugs. Int Anesthesiol Clin. 1995 Jan;33(1):1–20. [ Links ]
12. Zhang GS, Mathura JR. Images in clinical medicine. Painless loss of vision after vomiting. N Engl J Med. 2005 Apr 28;352(17):e16. [ Links ]
]]>13. Nanji GM, Maltby JR. Vomiting and aspiration pneumonitis with the laryngeal mask airway. Can J Anaesth. 1992 Jan;39(1):69–70. [ Links ]
14. Rashiq S, Bray P. Relative value to surgical patients and anesthesia providers of selected anesthesia related outcomes. BMC BMC Med Inform Decis Mak informatics and decision making. 2003 Feb 13;3:3. [ Links ]
15. Carroll N V, Miederhoff P, Cox FM, Hirsch JD. Postoperative nausea and vomiting after discharge from outpatient surgery centers. Anesth Analg. 1995 May;80(5):903–9. [ Links ]
16. Aapro MS, Walko CM. Aprepitant: drug-drug interactions in perspective. Ann Oncol. 2010 Dec;21(12):2316–23. [ Links ]
17. Gray H. Aprepitant for postoperative nausea and vomiting. Anaesth Intensive Care. 2009 Jan;37(1):135–6. [ Links ]
]]>18. Dando TM, Perry CM. Aprepitant: a review of its use in the prevention of chemotherapy-induced nausea and vomiting. Drugs. 2004 Jan;64(7):777–94. [ Links ]
19. Carlisle JB, Stevenson CA. Drugs for preventing postoperative nausea and vomiting. Cochrane Database Syst Rev. 2006 Jan;(3):CD004125. [ Links ]
20. Moreno L, Rueda J, Rodríguez G, Rincón D. Prepitant para la prevención de la nausea y el vomito postoperatorio, una revisión sistemática de la literatura. Bogotá D.C.; 2012 p. 21. [ Links ]
21. Jung WS, Kim YB, Park HY, Choi WJ, Yang HS. Oral administration of aprepitant to prevent postoperative nausea in highly susceptible patients after gynecological laparoscopy. J Anesth. 2013 Jun;27(3):396–401. [ Links ]
22. Vallejo MC, Phelps AL, Ibinson JW, Barnes LR, Milord PJ, Romeo RC, et al. Aprepitant plus ondansetron compared with ondansetron alone in reducing postoperative nausea and vomiting in ambulatory patients undergoing plastic surgery. Plast Reconstr Surg. 2012 Feb;129(2):519–26. [ Links ]
]]>23. Lee S-J, Lee SM, Kim SI, Ok SY, Kim SH, Park S-Y, et al. The effect of aprepitant for the prevention of postoperative nausea and vomiting in patients undergoing gynecologic surgery with intravenous patient controlled analgesia using fentanyl: aprepitant plus ramosetron vs ramosetron alone. Korean J Anesthesiol. 2012 Sep;63(3):221–6. [ Links ]
24. Lim CS, Ko Y-K, Kim Y-H, Park S-I, Kim J-K, Kim M-J, et al. Efficacy of the oral neurokinin-1 receptor antagonist aprepitant administered with ondansetron for the prevention of postoperative nausea and vomiting. Korean J Anesthesiol. 2013 Mar;64(3):212–7. [ Links ]
25. Alonso-Damián ER, Anguiano-García MM. Aprepitant contra ondansetrón en la prevención de náusea y vómito postoperatorio en pacientes intervenidos de colecistectomía abierta. Revista Mexicana de Anestesiología. 2012;35(1):8–14. [ Links ]
26. Lai ML, Tang YK, Cheng C B, Kan K P, Fu YT, Ng YC, et al. Combining aprepitant with dexamethasone for the prevention of postoperative nausea and vomiting. Anaesth Intensive Care. 2008;36(4):597. [ Links ]
27. Gin T, Lai L, Tang Y, Cheng B, Chan M. Aprepitant for preventing postoperative nausea and vomiting: A dose finding study. Eur J Anaesthesiol. 2009;26(Sup 45):129. [ Links ]
]]>28. DiIorio TM, Sharkey PF, Hewitt AM, Parvizi J. Antiemesis after total joint arthroplasty: does a single preoperative dose of aprepitant reduce nausea and vomiting? Clin Orthop Relat Res. 2010 Sep;468(9):2405–9. [ Links ]
29. Hartrick CT, Tang Y-S, Hunstad D, Pappas J, Muir K, Pestano C, et al. Aprepitant vs. multimodal prophylaxis in the prevention of nausea and vomiting following extended-release epidural morphine. Pain Pract. 2010;10(3):245–8. [ Links ]
30. Uribe A, Puente E, Viloria A, Schultz A, Zisman G, Bergese S. Aprepitant vs ondansetron as triple-therapy for the prophylaxis of postoperative nausea and vomiting. Anesth Analg. 2011;112(S-277) [ Links ].
31. Mimms SE, Ibele A, McKenna D, Blythe L, Don S, Ditslear III J, et al. A retrospective study of the effect of emend (aprepitant) combined with corticosteroids on the Length of stay in patients undergoing laparoscopic Roux-en-y gastric bypass. Surg Endosc. 2011;25(S249) [ Links ].
32. Kakuta N, Tsutsumi YM, Horikawa YT, Kawano H, Kinoshita M, Tanaka K, et al. Neurokinin-1 receptor antagonism, aprepitant, effectively diminishes post-operative nausea and vomiting while increasing analgesic tolerance in laparoscopic gynecological procedures. J Med Invest. 2011 Aug;58(3-4):246–51. [ Links ]
]]>33. Green MS, Green P, Malayaman SN, Hepler M, Neubert LJ, Horrow JC. Randomized, double-blind comparison of oral aprepitant alone compared with aprepitant and transdermal scopolamine for prevention of postoperative nausea and vomiting. Br J Anaesth. 2012 Nov;109(5):716–22. [ Links ]
34. Moher D, Hopewell S, Schulz KF, Montori V, Gøtzsche PC, Devereaux PJ, et al. CONSORT 2010 Explanation and Elaboration: Updated guidelines for reporting parallel group randomised trials. J Clin Epidemiol. 2010 Aug;63(8):e1–37. [ Links ]
35. Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996 Feb;17(1):1–12. [ Links ]
36. Gan TJ, Apfel CC, Kovac A, Philip BK, Singla N, Minkowitz H, et al. A randomized, double-blind comparison of the NK1 antagonist, aprepitant, versus ondansetron for the prevention of postoperative nausea and vomiting. Anesth Analg. 2007 May;104(5):1082–9, [ Links ] tables of contents.
37. Diemunsch P, Gan TJ, Philip BK, Girao MJ, Eberhart L, Irwin MG, et al. Single-dose aprepitant vs ondansetron for the prevention of postoperative nausea and vomiting: a randomized, double-blind phase III trial in patients undergoing open abdominal surgery. Br J Anaesth. 2007 Aug;99(2):202–11. [ Links ]
]]>38. Habib AS, Keifer JC, Borel CO, White WD, Gan TJ. A comparison of the combination of aprepitant and dexamethasone versus the combination of ondansetron and dexamethasone for the prevention of postoperative nausea and vomiting in patients undergoing craniotomy. Anesth Analg. 2011 Apr;112(4):813–8. [ Links ]
39. Chandrakantan A, Glass PSA. Multimodal therapies for postoperative nausea and vomiting, and pain. Br J Anaesth. 2011 Dec;107 Suppl:i27–40. [ Links ]
40. Gan TJ, Meyer TA, Apfel CC, Chung F, Davis PJ, Habib AS, et al. Society for Ambulatory Anesthesia guidelines for the management of postoperative nausea and vomiting. Anesth Analg. 2007 Dec;105(6):1615–28, [ Links ] table of contents.
41. Roila F, Fatigoni S. New antiemetic drugs. Ann Oncol. 2006 Mar;17 Suppl 2:ii96–100. [ Links ]
42. Bajwa SS, Bajwa SK, Kaur J, Sharma V, Singh A, Singh A, et al. Palonosetron: A novel approach to control postoperative nausea and vomiting in day care surgery. Saudi J Anaesth. 2011 Jan;5(1):19–24. [ Links ]
]]>43. Laha B, Hazra A, Mallick S. Evaluation of antiemetic effect of intravenous palonosetron versus intravenous ondansetron in laparoscopic cholecystectomy: a randomized controlled trial. Indian J Pharmacol. 2013;45(1):24–9. [ Links ]
44. Moon YE, Joo J, Kim JE, Lee Y. Anti-emetic effect of ondansetron and palonosetron in thyroidectomy: a prospective, randomized, double-blind study. Br J Anaesth. 2012 Mar;108(3):417–22. [ Links ]
45. Gan TJ, Diemunsch P, Habib AS, Kovac A, Kranke P, Meyer TA, et al. Consensus guidelines for the management of postoperative nausea and vomiting. Anesth Analg. 2014 Jan;118(1):85–113. [ Links ]
]]>