XIOMARA ÚSUGA1, MARÍA TERESA RUGELES1
1Grupo InmunovirologíaBiogénesis, Universidad de Antioquia. Medellín, Colombia.
Presentado marzo 15 de 2006, aceptado junio 30 de 2006, correcciones agosto 10 de 2006.
RESUMEN
En la actualidad existe un gran interés por identificar proteínas o péptidos antimicrobianos que puedan ser herramientas terapéuticas que eviten el establecimiento o permitan el control de diferentes infecciones. Las ribonucleasas (RNasas), pertenecientes a la superfamilia Ribonucleasa A, son enzimas que participan en varios procesos fisiológicos, que van desde el procesamiento alternativo del RNA hasta la angiogénesis. Estas enzimas son expresadas por diferentes tejidos y exhiben especificidades variables contra diferentes sustratos de RNA. El potencial terapéutico de las RNasas se ha sugerido en procesos oncogénicos; adicionalmente, se ha descrito que tienen actividad antiviral directa y el potencial de activar células del sistema inmune innato induciendo su maduración y la producción de citoquinas proinflamatorias. Nuestro grupo de investigación ha realizado estudios que señalan la capacidad de cuatro RNasas recombinantes: EDN, 4EDN, RNasa A y angiogenina de inhibir la replicación del virus de la inmunodeficiencia humana tipo 1 en linfocitos T de sangre periférica activados. En este artículo se revisará la clasificación de las ribonucleasas que constituyen la superfamilia RNasa A y se describirá, en forma detallada, lo que se conoce de la función biológica, acción antiviral y mecanismo de acción de las RNasas a las que se les ha reportado actividad antiviral.
Palabras clave: actividad antiviral, RNasa 1, EDN, ECP, ONC.
ABSTRACTCurrently, there is a great interest to identify proteins or antimicrobial peptides to be included in the therapeutic arsenal for preventing different infectious diseases. Ribonucleases (RNases) that belong to the Ribonuclease A superfamily participate in several physiologic processes, from alternative splicing of RNA to organogenesis. These enzymes are expressed by various tissues and exhibit variable specificities against different RNA substrates. The therapeutic potential of RNases has been suggested for oncogenic processes; in addition, direct antiviral activity and the potential to activate cells from the innate immune system, inducing their maturation and release of proinflammatory cytokines have been also associated with these enzymes. Our research team, have carried out studies that indicate the ability of four recombinant RNases: EDN, 4EDN, RNase A and angiogenin to inhibit HIV1 replication in activated peripheral blood T lymphocytes. In this article we review the classification of RNases that belong to the Ribonucleases A superfamily; we describe in detail what is known regarding the biologic function, inhibitory activity and mechanism of action of the RNases recognized by their antiviral activity.
]]> Key words: antiviral activity, RNase 1, EDN, ECP, ONC. INTRODUCCIÓNLa población de RNA en las células es controlada postranscripcionalmente por enzimas denominadas ribonucleasas (RNasas) que poseen especificidades comunes o diferentes, exhiben un patrón de expresión variable y difieren en su actividad contra diferentes sustratos de RNA (Deshpande y Shankar, 2002). Una sola célula puede contener hasta 20 RNasas diferentes, las cuales pueden hacer parte de complejos supramoleculares y funcionar en conjunto con otras enzimas (Deutscher y Li, 2001). Las RNasas y sus homólogos estructurales son moléculas reguladoras que controlan procesos que van desde el procesamiento alternativo a la organogénesis (Beintema y Kleineidam, 1998). Diferentes estudios apoyan el uso de algunas RNasas para el tratamiento de enfermedades infecciosas y el cáncer, aunque su potencial terapéutico está limitado por su habilidad para penetrar las células (Costanzi et al., 2005).
DEFINICIÓN
Las RNasas son proteínas con actividad enzimática presentes en bacterias (Ilinskaya et al., 2001), hongos (Kao y Davies, 1999), plantas superiores (Roalson y McCubbin, 2003) y mamíferos (Breukelman et al., 2001), que participan en procesos fisiológicos diversos tales como: muerte celular (Lin et al.,1994), replicación del DNA (Deshpande y Shankar, 2002), transcripción, procesamiento y edición del RNA (Deshpande y Shankar, 2002), defensa del hospedero (Domachowske et al., 1998) y control del crecimiento tumoral (Griffiths et al., 1997).
CLASIFICACIÓN
Las ribonucleasas están divididas en tres grandes familias: la superfamilia RNasa A, la familia T1 y la familia T2. La superfamilia Ribonucleasa A está constituida por las RNasas de mamíferos y otros vertebrados como aves, reptiles y anfibios (Soochin et al., 2005). Este grupo de proteínas presenta altas tasas de duplicación génica y pérdida de genes de lo que ha resultado un número variable de genes en diferentes especies (Soochin et al., 2005). En el ser humano, los genes que las codifican están ubicados en el brazo largo del cromosoma 14. Sierakowska y Shugar (1977) agruparon las RNasas humanas en dos categorías: RNasas secretadas y no secretadas; posteriormente, el grupo de Weickmann et al. (1981) cambió el término de secretadas por el de RNasas de tipo pancreático y Sorrentino y Libonati (1994), introdujeron el término no pancreáticas en reemplazo de no secretadas. Las RNasas de tipo pancreático incluyen tanto ribonucleasas encontradas en el páncreas como en otros fluídos corporales con propiedades catalíticas y estructurales similares a las RNasas pancreáticas humanas y de bovino. El término de tipo no pancreático se utilizó para las RNasas que presentan propiedades catalíticas y tienen secuencia similar a la neurotoxina derivada del eosinófilo (EDN) o a la RNasa K2 de riñón de bovino. Otros miembros de la superfamilia RNasa A, tales como la RNasa 4 y la RNasa PL3 de hígado porcino constituyen una tercera familia denominadas RNasas de tipo pancreático/no pancreático, las cuales son estructuralmente más similares a las RNasas de tipo pancreático pero comparten algunas propiedades catalíticas con ambos tipos de ribonucleasas (Sorrentino y Libonati, 1997).
Actualmente algunos autores se refieren a las cinco primeras RNasas (Tabla 1) como RNasas pancreáticas según la clasificación anteriormente descrita. Debido a que dichas clasificaciones pueden crear confusión, ninguno de esos términos será usado en esta revisión.
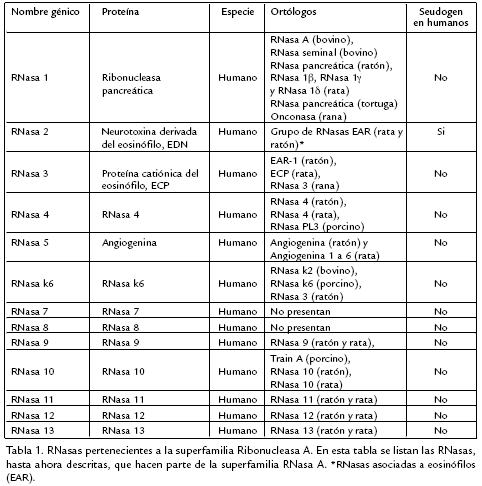
Figura 1. Representación esquemática de una RNasa humana. Las líneas continuas ( ) corresponden a los aminoácidos conservados entre las RNasas 1 a 8. Las líneas en puntos (….) representan cisteínas conservadas entre los miembros de la superfamilia RNasa A. Las líneas discontinuas () corresponden a los residuos del sitio activo de las RNasa 1 (H12, K41, H119) los cuales están presentes en todas las RNasas de la superfamilia RNasa A en posiciones similares (Soochin et al., 2005). Las RNasas 9 y 10 no poseen la tríada catalítica ni el péptido señal (Penttinen et al., 2003) y las RNasas 11, 12 y 13 presentan variabilidad en la región de la tríada catalítica (Soochin et al., 2005).
]]> Estas enzimas están compuestas de un péptido señal de aproximadamente 25 aminoácidos y un péptido maduro con alrededor de 130 aminoácidos. Las RNasas presentan varias características estructurales conservadas, entre las cuales son particularmente importantes tres residuos catalíticos (una lisina y dos histidinas) y de seis a ocho cisteínas que forman tres o cuatro puentes disulfuro (Soochin et al., 2005; Figs. 1 y 2). La homología entre las RNasas que hacen parte de la superfamilia RNasa A puede ser entre 20% hasta casi un 100% (Soochin et al., 2005). Las RNasas 9 y 10 no poseen la tríada catalítica ni el péptido señal (Penttinen et al., 2003) y las RNasas 11, 12 y 13 presentan variabilidad en la región de la tríada catalítica (Soochin et al., 2005), lo que sugiere que estas enzimas no poseen actividad ribonucleolítica. Todas estas enzimas exhiben una estructura tridimensional similar; las mayores diferencias se encuentran en otros residuos que forman el sitio activo ribonucleolítico el cual es responsable de las diferentes actividades específicas y preferencias de sustrato. A continuación se describirán, en mayor detalle, las RNasas humanas y la onconasa para las cuales la actividad antiviral ha sido demostrada in vitro.
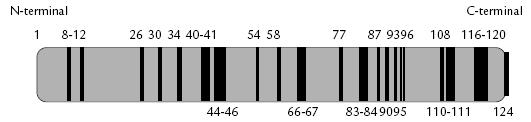
Figura 2. Representación esquemática de la RNasa A bovina. Las líneas continuas corresponden a los residuos conservados entre esta RNasa y la Onconasa.
RNASA PANCREÁTICA HUMANA/RNASA 1
Ésta es una glicoproteína con una masa molecular de 15 kDa sin glicosilación, secretada por un gran número de tejidos tales como: páncreas (Weickmann et al., 1981), riñón (Mizuta et al., 1990), hígado, cerebro, bazo (Futami et al., 1997) y endotelio (Landré et al., 2002); también es producida por células tumorales de adenocarcinoma pancreático (Peracaula et al., 2000). Adicionalmente, se encuentra en plasma seminal (De Prisco et al.,1984) y orina (Futami et al., 1997). Es una de las RNasas de mayor circulación en sangre con una concentración aproximada de 400 ng/mL (Weickmann et al., 1984). Esta enzima monomérica está constituida por 128 aminoácidos y según el tejido de origen puede presentar diferentes patrones de glicosilación, con tres puntos de glicosilación en el extremo amino terminal en los residuos asparagina34 (Asn34), Asn76 y Asn88. La enzima de origen urinario presenta los tres sitios glicosilados ; la proteína pancreática y parte de la RNasa 1 seminal sólo están glicosiladas en Asn34 mientras que la enzima del riñón no es glicosilada, al igual que el 50% de las enzima s presentes en plasma seminal y cerebro (Sorrentino y Libonati, 1997). Se ha encontrad o alteración en el patrón de glicosilación de la RNasa 1 producida por tejido de adenocarcinoma pancreático y líneas celulares de tumor pancreático. La RNasa 1 en condiciones normales presenta niveles más altos de fucosa y ausencia de ácido siálico en comparación a la RNasa 1 secretada por la línea celular tumoral humana pancreática (Dwek et al., 2003). Aunque la estructura primaria de esta proteína muestra un 70% de identidad con la RNasa bovina A (Soochin et al., 2005), a diferencia de esta proteína, la RNasa 1 humana posee una gran actividad contra el RNA de doble cadena, contiene mayor proporción de residuos básicos, su actividad es diferencialmente influenciada por la fuerza iónica y los iones divalentes y tiene una extensión en su porción carboxiloterminal de cuatro residuos de aminoácidos (Sorrentino y Libonati, 1997). Función biológica. Aunque la RNasa pancreática humana no ha sido asociada con alguna función biológica en especial (Sorrentino y Libonati, 1997), se demostró que cataliza eficientemente la degradación del RNA de doble cadena in vitro (Sorrentino et al., 2003), lo que sugiere su participación en la respuesta inmune innata. Además, se encontró que es capaz de estimular células dendríticas para la producción de varios factores solubles e inducir su maduración in vitro (De Yang et al., 2003). El uso de un conjugado de RNasa pancreática y albúmina de suero humano inyectado en ratones infectados con virus Influenza A e Influenza B, mostró alta actividad antiviral (Zelepuga et al., 2003). Estudios recientes nuestros señalan que la RNasa pancreática recombinante inhibe la replicación del virus de la inmunodeficiencia humana tipo 1 (VIH1) en un cultivo primario de linfocitos T activados (Bedoya et al., en prensa). Mecanismo de acción. La región amino terminal tiene un papel importante en el clivaje del RNA. Exhibe actividad catalítica específica por pirimidinas con una preferencia marcada por sustrato policitosina (poliC) sobre poliuracilo (poliU). Además, esta enzima es dos veces más activa que la RNasa A bovina, por poliadenina (poliA) debido a la presencia del residuo aspártico83 (Asp83). Su acción catalítica se potencia en altas concentraciones de cloruro de sodio (NaCl) y muestra actividad óptima a pH 8,0 (Boix et al., 1996). Inhibidores. Esta RNasa no es citotóxica gracias a la acción neutralizante de la proteína inhibidor de ribonucleasa (IR), glicoproteína citosólica de 50 kDa, cuya función es preservar la integridad del RNA celular (Sorrentino y Libonati, 1997). Gaur et al. (2001) demostraron que los residuos lisina7 (Lis7), glutamina11 (Gln11), Asn71 y glutámico111 (Glu111) en la RNasa pancreática humana son los aminoácidos que interactúan con el IR.
NEUROTOXINA DERIVADA DEL EOSINÓFILO EDN/RNASA 2
Esta proteína es producida y almacenada en eosinófilos (Durack et al., 1981), y se ha encontrado en hígado (Sorrentino et al., 1988), bazo (Yasuda et al., 1990), neutrófilos (Sur et al., 1998), placenta (Shapiro y Vallee, 1991), monocitos/macrófagos activados (De Yang et al., 2004) y en orina (Beintema et al., 1988). Su peso molecular es de 18,4 kDa; es una proteína termoestable (Motojima et al., 1989), catiónica y con una vida media en sangre muy corta (Boix et al., 1996). La estructura tridimensional de la EDN se caracteriza por la presencia de tres asas que la hacen diferente de las otras RNasas de la superfamilia RNasa A. Además, presenta una inserción de aminoácidos Asp115 a tirosina123 (Tyr123) que comparte con la proteína catiónica del eosinófilo y la RNasa k6 (Rosenberg, 1998). Posee cinco sitios potenciales para glicosilación en el aminoácido asparagina (Beintema y Kleineidam, 1998). La EDN presenta dos formas alternas resultado de un procesamiento alternativo o de una modificación postrasduccional; una de ellas contiene cuatro aminoácidos adicionales (SerinaLeucinaHistidinaValina) en la región amino terminal, considerada como parte de la secuencia señal ((4)EDN; Shapiro y Vallee, 1991). La EDN presenta una homología del 67% con la proteína catiónica del eosinófilo. Su actividad neurotóxica se demostró cuando al ser inyectada intratecalmente en conejos indujo un síndrome de rigidez muscular, ataxia y parálisis asociada con pérdida de las células de Purkinje; síndrome conocido como Fenómeno Gordon (Rosenberg, 1998). Sus niveles en sangre se han encontrado elevados en enfermedades asociadas con eosinofilia como por ejemplo en infección por helmintos (Durack et al., 1979), asma bronquial (Tischendorf et al., 1996) y dermatitis atópica (Dahl et al., 1978). Función biológica. Actúa como quimioquina para células dendríticas maduras e inmaduras mediante la activación de la proteína quinasa activada por mitógenos p42/44 (MAPK; Sugai et al., 1992). Adicionalmente, se ha demostrado su acción antiviral in vitro en líneas celulares crónicamente infectadas con el VIH1 y contra el virus respiratorio sincitial (Domachowske et al., 1998), y es responsable por la actividad antiVIH1 que exhibe el sobrenadante de reacciones alogénicas (Rugeles et al., 2003). Recientemente reportamos que la enzima EDN recombinante inhibe la replicación del VIH1 en linfocitos T activados (Bedoya et al., en prensa). Mecanismo de acción. Se ha demostrado su acción de ribonucleasa sobre RNA de cadena sencilla con preferencia sobre poli (U); esta actividad es inhibida en altas concentraciones de NaCl (De Yang et al., 2004) y muestra actividad ribonucleasa óptima sobre RNA de levadura a pH entre 6,57,0 (Sorrentino y Libonati, 1997). Es incapaz de catalizar la hidrólisis de nucleótidos cíclicos por la ausencia del aminoácido fenilalanina en la posición 120 (Sorrentino y Libonati, 1997) y de degradar RNA de doble cadena bajo algunas condiciones experimentales (Motojima et al., 1989). Inhibidores. La actividad de la EDN es neutralizada por el inhibidor de ribonucleasa placentaria humana (Sorrentino y Libonati, 1994); el efecto neutralizante es dependiente de la dosis (Iyer et al., 2005).
PROTEÍNA CATIÓNICA DEL EOSINÓFILO ECP/RNASA 3Esta RNasa constituye uno de los mayores componentes granulares del eosinófilo (Olsson y Venge, 1972). Adicionalmente, se ha encontrado en líneas celulares eosinofílicas (Olsson et al., 1977) y en neutrófilos (Sur et al., 1998). Es una proteína secretada, de carácter básico y su propiedad altamente catiónica es dependiente del número de residuos de arginina en la superficie molecular. En su secuencia presenta tres sitios de glicosilación, en la región amino terminal, con oligosacáridos complejos similares a los encontrados en EDN. ECP presenta un peso molecular de 15,6 kDa sin glicosilar, mientras que las formas glicosiladas pueden variar entre 16 y 22 kDa (Barker et al., 1989). La RNasa 3 presenta un 70% de homología con EDN; las diferencias entre estas dos RNasas radica en el número de residuos de arginina, que en EDN son nueve y en la ECP alcanza un total de 19 residuos. Su actividad ribonucleolítica es mucho menor que la de EDN. Función biológica. Varias actividades de la ECP han sido caracterizadas in vitro tales como disminución del tiempo de coagulación dependiente del factor XII de la cascada de coagulación (Venge et al., 1979), aumento de la fibrinolisis por activación del plasminógeno inducida por uroquinasa (Dahl y Venge, 1979) y regulación de los componentes de la vía clásica del complemento. Además, se demostró en varios estudios que la ECP tiene toxicidad hacia Schistosoma (Ackerman et al., 1985), Trypanosoma (Molina et al., 1988), Microphilariae (Hamann et al., 1990), Trichinella (Hamann et al., 1987) y Plasmodium (Waters et al., 1987). También se demostró su actividad contra bacterias Gramnegativas y Grampositivas independiente de su acción ribonucleolítica (Carreras et al., 2003). Es tóxica para células y tejidos de mamíferos (Maeda et al., 2002). La ECP humana recombinante ha demostrado actividad antiviral contra formas extracelulares del virus respiratorio sincitial (Domachowske et al., 1998). Mecanismo de acción. La ECP es considerada como una enzima con actividad específica por RNA de cadena sencilla, con preferencia por poli(U). In vitro, la ECP muestra actividad óptima a valores de pH entre 6,57,0 con RNA de levadura como sustrato y es incapaz de catalizar la hidrólisis de nucleótidos cíclicos (Sorrentino y Glitz, 1991). Debido a su carácter altamente catiónico, la ECP puede unirse a moléculas cargadas negativamente de las membranas celulares, siendo capaz de formar canales selectivos no iónicos o poros estables en la membrana (Young et al., 1986). Además de la formación del poro transmembrana, la ECP se internaliza y por medio de la interacción con la proteína carboxipeptidasa E, en células neuroendocrinas, escapa al ataque proteolítico. Si logra acumularse en exceso y sobrepasa la concentración de los inhibidores de RNasas, induce la degradación de moléculas de RNA citosólico inhibiendo el crecimiento celular (Wu et al., 2004). Inhibidores. Su acción citotóxica es completamente bloqueada por el inhibidor de RNasa citosólico expresado ubicuamente (Maeda et al., 2002).
ONCONASA/P30/RANPIRNASALa onconasa (ONC) es una RNasa aislada de oocitos y de embriones en estadío temprano de la Rana pipiens. Es un miembro de la superfamilia RNasa A que presenta un 30% de homología con la RNasa A bovina. A pesar del bajo grado de homología entre las estructuras primarias, la estructura tridimensional de la ONC muestra una topología muy similar a la RNasa A (Ardelt et al., 1994). Posee los principales residuos del sitio activo y tres de los cuatro puentes disulfuro característicos de las RNasas; sin embargo, la ONC parece tener un único mecanismo catalítico debido a un residuo piroglutamato en su región amino terminal (Boix et al., 1996). Es una proteína con alta estabilidad térmica gracias a su estructura terciaria compacta (Notomista et al., 2000). La ONC aislada de los oocitos de Rana pipiens ha sido evaluada en varios ensayos clínicos en humanos como terapia antitumoral (Costanzi et al., 2005).
]]> Función biológica. Se ha demostrado que esta RNasa es citotóxica in vitro para varias líneas celulares tumorales de mamífero (Darzynkiewicz et al., 1988) y se ha descrito también actividad antitumoral in vivo en modelos animales (Milkulski et al., 1990). Tiene una gran capacidad de inhibir la replicación del VIH1 en líneas crónicamente infectadas con este virus (Saxena et al., 1996). Mecanismo de acción. ONC muestra una actividad específica disminuída hacia sustratos específicos en comparación con la RNasa A; exhibe preferencia por poli(U) y por poliguanina(poliG). Su actividad óptima se alcanza a pH 5,5. Estudios previos en modelos animales indican que la citotoxicidad de ONC se debe a su capacidad de unirse a la superficie celular, entrar al citosol y degradar el RNA causando la muerte celular (Wu et al., 1993). Iordanov et al. (2000) reportó la especificidad de ONC por RNA de transferencia, sin afectar el RNA ribosomal y mensajero. Las células tratadas con ONC presentaron signos de apoptosis similares a los inducidos por caspasa3, que fueron independientes de la inhibición de la síntesis de proteínas. Inhibidores. Las RNasas de anfibios no son bloqueadas por el inhibidor de ribonucleasas placentarias de mamíferos (Beintema y Kleineidam, 1998), lo que puede explicar su alta citotoxicidad en células de mamíferos.PAPEL ANTIVIRAL DE LAS RIBONUCLEASAS
Los mamíferos están constantemente expuestos a una gran cantidad de microorganismos contra los cuales responden a través del sistema inmune innato y adaptativo. La inmunidad innata representa la primera línea de defensa que es rápidamente activada en respuesta a la invasión microbiana, de la cual hacen parte diferentes células y factores solubles. En los últimos años, varios de estos factores solubles con actividad microbicida, entre ellos las RNasas, han suscitado particular interés por el potencial terapéutico que tienen.
Se ha propuesto que algunas RNasas se unen a la superficie celular, gracias a su carácter altamente catiónico, entran al citosol y degradan el RNA induciendo la muerte celular. Esta acción catalítica per se de las RNasas hace parte del mecanismo antiviral de estas enzimas, tal como se ha sugerido para la ONC y la ECP, las cuales degradan el RNA ribosomal. La acción citotóxica sobre la célula blanco limitaría la replicación viral. Adicionalmente, se ha sugerido una acción directa de las RNasas, particularmente de la ONC sobre el RNA viral en cultivo de células H9 infectadas con el VIH1 (Saxena et al., 1996). En el caso de virus DNA, la acción catalítica de las RNasas podría darse después de la transcripción, una vez se hayan producido los RNAm. Otro mecanismo efector es la acción quimiotáctica y por ende proinflamatoria, como la actividad exhibida por EDN. Al reclutar células dendríticas se induce la producción de factores quimiotácticos para otras células del sistema inmune; de esta manera, EDN potencia la respuesta innata y promueve la inmunidad adaptativa que permitiría eliminar más fácilmente la infección viral (Shapiro y Vallee, 1991).
El potencial terapéutico de las RNasas fue inicialmente explorado por Glukhov et al. (1976) al usar la RNasa 1 para el tratamiento de pacientes infectados con el virus de la encefalitis Tickborne. Después del tratamiento, se observó una rápida resolución de los síntomas meníngeos y de la pleocitosis en el fluído cerebroespinal. Desde entonces, se han realizado varios estudios que han evaluado el uso terapéutico de estas enzimas en otros modelos, particularmente tumorales, tanto in vitro como in vivo. Utilizando EDN purificada, a partir de preparaciones comerciales de la hormona gonadotropina coriónica humana (hCG), se observó una disminución del crecimiento de una línea celular derivada de Sarcoma de Kaposi, KSIMM, la cual fue dependiente de la dosis. Igualmente, se analizó un péptido sintético de 16 residuos, similar a la secuencia activa de la enzima 4EDN, el cual exhibió un efecto citotóxico dependiente de la dosis, sobre la misma línea celular (Dricu et al., 2004). La ONC también ha sido evaluada como agente antitumoral en estudios de fase II en pacientes con mesotelioma maligno (Milkulski et al., 2002) y en estudios de fase I administrándola en forma intravenosa a pacientes con diferentes tipos de tumores, quienes habían presentado resistencia a otras terapias (Milkuski et al.,1993). Esta RNasa demostró una buena actividad antitumoral y un perfil de toxicidad tolerable. Actualmente se están realizando estudios de fase III con esta RNasa para el tratamiento del cáncer de pulmón, pancreático y mesotelioma maligno.
CONCLUSIONES
Basándose en estudios realizados in vitro y algunos in vivo, es evidente que las RNasas tienen actividad antimicrobiana potencial, y un papel importante en iniciar y amplificar la respuesta inmune innata y adaptativa contra la invasión microbiana. La actividad antiviral demostrada para estas RNasas abre un nuevo campo de estudio dirigido a la creación de una nueva clase de agentes antivirales usando estas proteínas o formas modificadas.
AGRADECIMIENTOS
Esta revisión hace parte del proyecto financiado por Colciencias y la Universidad de Antioquia, código 1150412948.
BIBLIOGRAFÍA
]]> ACKERMAN SJ, GLEICH GJ, LOEGERING DA, RICHARDSON BA, BUTTERWORTH AE. Comparative Toxicity of Purified Human Eosinophil Granule Cationic Proteins for Schistosomula of Schistosoma mansoni. Am J Trop Med Hyg. 1985;34:735–745.ARDELT W, LEE HS, RANDOLPH G, VIERA A, MIKULSKI, SM, SHOGEN K, Enzymatic Characterization of Onconase, a Novel Ribonuclease with Antitumor Activity. Protein Sci. 1994;3:S137.
[ Links ]BARKER RL, LOEGERING DA, TEN RM, HAMANN KJ, PEASE LR, GLEICH GJ. Eosinophil Cationic Protein cDNA Comparison with Other Toxic Cationic Proteins and Ribonucleases. J Immunol. 1989;143:952955.
[ Links ]BEDOYA V, BOASSO A, HARDY AW, RYBAK S, SHEARER GM, RUGELES MT. Ribonucleases in HIV1 Inhibition: Effect of Recombinant RNases on Infection of Primary T Cells and Immune ActivationInduced RNase Gene and Protein Expression. AIDS Res Hum Retroviruses. In press. 2006
[ Links ]BEINTEMA JJ, KLEINEIDAM RG. The Ribonuclease A Superfamily: General Discussion. Cell Mol Life Sci. 1998;54:825832.
[ Links ]BEINTEMA JJ, BLANK A, SCHIEVEN GL, DEKKER CA, SORRENTINO S, LIBONATI M. Differences In Glycosilation Pattern of Human Secretory Ribonucleases. Biochem J. 1988;255:501505.
[ Links ]BOIX E, WU Y, VASANDANI VM, SAXENA S, ARDELT W, LADNER J, YOULE RJ. Role of the N Terminus in RNase A Homologues: Differences in Catalytic Activity, Ribonuclease Inhibitor Interaction and Cytotoxicity. J Mol Biol. 1996;257:9921007.
[ Links ]BREUKELMAN HI, JEKEL PA, DUBOIS JY, MULDER PP, WARMELS HW, BEINTEMA JJ. Secretory Ribonucleases in the Primitive Ruminant chevrotain Tragulus javanicus. Eur J Biochem. 2001;268:38903897.
[ Links ]CARRERAS E, BOIX E, ROSENBERG JF, CUCHILLO CM, NOGUES MV. Both Aromatic and Cationic Residues Contribute to the Membranelytic and Bactericidal Activity of Eosinophil Cationic Protein. Biochemistry. 2003;42:66366644.
[ Links ]COSTANZI J, SIDRANSKY D, NAVON A, GOLDSWEIG H. Ribonucleases as a Novel proApoptotic Anticancer Strategy: Review of the Preclinical and Clinical Data for Ranpirnase. Cancer Invest. 2005;23:643650.
[ Links ]DAHL R, VENGE P. Enhancement of UrokinaseInduced Plasminogen Activation by the Cationic Protein of Human Eosinophil Granulocytes. Thromb Res. 1979;14:599–608.
DAHL R, VENGE P, OLSSON I. Variations of Blood Eosinophils and Eosinophil Cationic Proteins in Serum of Patients with Bronquial Asthma. Allergy. 1978;33:211215.
[ Links ]DARZYNKIEWICZ Z, CARTER SP, MIKULSKI SM, ARDELT WJ, SHOGEN K. Cytostatic and Cytotoxic Effects of Pannon (P30 Protein), a Novel Anticancer Agent. Cell Tissue Kinet. 1988;21:169182.
[ Links ]DE PRISCO R, SORRENTINO S, LEONE E, LIBONATI M. A Ribonuclease from Human Seminal Plasma Active on Double Stranded RNA. Biochim Biophys Acta. 1984;788:356363.
[ Links ]DE YANG QQ, ROSENBERG H, CHEN Q, DYER KD, KUROSAKA K, OPPENHEIM J. Eosinophil Derived Neurotoxin (EDN), an Antimicrobial Protein with Chemotactic Activities for Dendritic Cells. Blood. 2003;102:33963403.
[ Links ]DE YANG QQ, BIRAGYN A, HOOVER DM, LUBKOWSKI J, OPPENHEIM JJ. Multiple Roles of Antimicrobial Defensins, Cathelicidins and EosinophilDerived Neurotoxin in Host Defense. Annu Rev Immunol. 2004;22:181215.
[ Links ]DE YANG QQ, ROSENBERG HF, RYBAK SM, NEWTON DL, WANG ZY, FU Q, et al. Human Ribonuclease A Superfamily members EosinophilDerived Neurotoxin and Pancreatic Ribonuclease, induce Dendritic and Activation. J Immunol. 2004;173:61346142.
[ Links ]DESHPANDE RA, SHANKAR V. Ribonucleases from T2 Family. Crit Rev Microbiol. 2002;28:79122.
[ Links ]DEUTSCHER MP, LI Z. Exoribonucleases and Their Multiple Roles in RNA Metabolism. Prog Nucleic Acid Res Mol Biol. 2001;66:67105.
[ Links ]DOMACHOWSKE JB, BONVILLE CA, DYER KD, ROSENBERG HF. Evolution of Antiviral Activity in the Ribonuclease A Gene Superfamily: Evidence for a Specific Interaction Between EosinophilDerived Neurotoxin (EDN/RNase 2) and Respiratory Syncytial Virus. Nucleic Acids Res. 1998;26:53275332.
[ Links ]DURACK DT, SUMI SM, KLEBANOFF SJ. Neurotoxicity of Human Eosinophils. Proc Natl Acad Sci U S A. 1979;76:14431447.
[ Links ]DURACK DT, ACKERMAN SJ, LOEGERING DA, GLEICH GJ. Purification of Human EosinophilDerived Neurotoxin. Proc Natl Acad Sci U S A. 1981;78:51655169.
[ Links ]DRICU A, SERGIUBOGDAN C, BRISMAR K, BIBERFELD P, ANDERSSON LC. A Synthetic Peptide Derived from the Human EosinophilDerived Neurotoxin Induces Apoptosis in Kaposi’s Sarcoma Cells. Anticancer Res. 2004;24:14271432.
DWEK RA, RUDD PM, DE LLORENS R. Glycosylation of Human Pancreatic Ribonuclease: Differences Between Normal and Tumor States. Glycobiology. 2003;13:227244.
[ Links ]FUTAMI J, TSUSHIMA Y, MURATO Y, TADA H, SASAKI J, SENO M. Tissue Specific Expression of PancreaticType Rnases and RNase Inhibitor in Humans. DNA Cell Biol. 1997;16:413419.
[ Links ]GAUR D, SWAMINATHAN S, BATRA JK. Interaction of Human Pancreatic Ribonuclease with Human Ribonuclease Inhibitor. J Biol Chem. 2001;216:2497824984.
[ Links ]GLUKHOV BN, JERUSALIMSKY AP, CANTER VM, SALGANIK RI. Ribonuclease Treatment of TickBorne Encephalitis. Arch Neurol. 1976;33:598603.
[ Links ]GRIFFITHS SJ, ADAMS DJ, TALBOT SJ. Ribonuclease Inhibits Kaposi’s Sarcoma. Nature. 1997;390:568.
HAMANN KJ, BARKER RL, LOEGERING DA, GLEICH GJ. Comparative Toxicity of Purified Human Eosinophil Granule Proteins for Newborn Larvae of Trichinella spiralis. J Parasitol. 1987;73:523529.
[ Links ]HAMANN KJ, GLEICH GJ, CHECKEL JL, LOEGERING DA, McCALL JW, BARKER RL. In vitro Killing of Microfilariae of Brugia pahangi and Brugia malayi by Eosinophil Granule Proteins. J Immunol. 1990;144:31663173.
[ Links ]ILINSKAYA O, DECKER K, KOSCHINSKI A, DREYER F, REPP H. Bacillus Intermedius Ribonuclease as Inhibitor of Cell Proliferation and Membrane Current. Toxicology. 2001;156:101107.
[ Links ]IORDANOV MS, RYABININA OP, WONG J, DINH T, NEWTON DL, RYBAK SM, et al. Molecular Determinants of Apoptosis Induced by the Cytotoxic Ribonuclease Onconase: Evidence for Cytotoxic Mechanisms Different from Inhibition of Protein Synthesis. Cancer Res. 2000;1:19831994.
[ Links ]IYER S, HOLLOWAY DE, KUMA K, SHAPIRO R, ACHARYA KR. Molecular Recognition of Human Eosinophilderived Neurotoxin (RNase 2) by Placental Ribonuclease Inhibitor. J Mol Biol. 2005;347:637655.
[ Links ]KAO R, DAVIES J. Molecular Dissection of Mitolgillin Reveals that the Fungal Ribotoxins are a Family of Natural Genetically Engineered Ribonucleases. J Biol Chem. 1999; 274:1257612582.
[ Links ]LANDRÉ JBP, HEWETT PW, OLIVOT J, FRIEDL P, KO Y, SACHINIDIS A, et al. Human Endothelial Cells Selectively Express Large Amounts of PancreaticType Ribonuclease (Rnase 1). J Cell Biochem. 2002;86:540552.
[ Links ]LIN JJ, NEWTON DL, MIKULSKI SM, KUNG HF, YOULE RJ, RYBAK SM. Characterization of the Mechanism of Cellular and Cell Free Protein Synthesis Inhibition by an AntiTumor Ribonuclease. Biochem Biophys Res Commun. 1994;204:156162.
[ Links ]MAEDA T, KITAZOE M, TADA H, DE LLORENS R, SALOMON DS, UEDA M, et al. Growth Inhibition of Mammalian Cells by Eosinophil Cationic Protein. Eur J Biochem. 2002;269:307316.
[ Links ]MAEDA T, MAHARA K, KITAZOE M, FUTAMI J, TAKIDANI A, KOSAKA M, et al. RNase 3 (ECP) Is an Extraordinarily Stable Protein among Human PancreaticType Rnases. J Biochem (Tokyo). 2002;132:737742.
[ Links ]MIKULSKI SM, BERNSTEIN E, ARDELT W, SHOGEN K, MENDUKE K. Striking Increase in Survival of Mice Bearing M109 Madison Carcinoma Treated with a Novel Protein from Amphibian Embryos. J Natl Cancer Inst. 1990;82:151153.
[ Links ]MIKULSKI SM, GROSSMAN AM, CARTER PW. Phase I Human Clinical Trial of Onconase P30 protein) Administered Intravenously on a Weekly Schedule in Cancer Patients with Solid Tumors. Int J Oncol. 1993;3:5764.
[ Links ]MIKULSKI SM, COSTANZI JJ, VOLGELZANG J, MCCACHREN S,TAUB RN, CHUN H, MITTELMAN A, et al. Phase II Trial of a Single Weekly Intravenous Dose of Ranpirnase in Patients With Unresectable Malignant Mesothelioma. J Clin Oncol. 2002;20:274281.
[ Links ]MIZUTA K, AWAZU S, YASUDA T, KISHI K. Purification and Characterization of Three Ribonucleases from Human Kidney: Comparison with Urine Ribonucleases. Arch Biochem Biophys. 1990;281:144151.
[ Links ]MOLINA HA, KIERSZENBAUM F, HAMANN KJ, GLEICH GJ. Toxic Effects Produced or Mediated by Human Eosinophil Granule Components on Trypanosoma cruzi. Am J Trop Med Hyg. 1988;38:327334.
[ Links ]MOTOJIMA S, FRIGAS E, LOEGERING DA, GLEICH GJ. Toxicity of Eosinophil Cationic Proteins for Guinea Pig Tracheal Epithelium in vitro. Am Rev Respir Dis. 1989;139:801805.
[ Links ]NOTOMISTA E, CATANZANO F, GRAZIANO G, PIAZ FD, BARONE G, D’ALESSIO G, et al. Onconase: An Unusually Stable Protein. Biochemistry. 2000;39:87118718.
OLSSON I, VENGE P. Cationic Proteins of Human Granulocytes. Isolation of the Cationic Proteins from the Granules of Leukaemic Myeloid Cells. Scand J Haematol. 1972;9:204214.
[ Links ]OLSSON I, VENGE P, SPITZNAGEL JK, LEHRER RI. ArginineRich Cationic Proteins of Human Eosinophil Granules. Comparison of the Constituents of Eosinophilic and Neutrophilic Leukocytes. Lab Invest. 1977;36:493500.
[ Links ]PENTTINEN J, PUJIANTO DA, SIPILA P, HUHTANIEMI I, POUTANEN M. Discovery in Silico and Characterization in vitro of Novel Genes Exclusively Expressed in the Mouse Epididymis. Mol Endocrinol. 2003;17:21382151.
[ Links ]PERACAULA R, CLEARY KR, LORENZO J, DE LLORENS R, FRAZIER ML. Human Pancreatic Ribonuclease 1: Expression and Distribution in Pancreatic Adenocarcinoma. Cancer. 2000;89:12521258.
[ Links ]ROALSON EH, MCCUBBIN AG. SRNases and Sexual Incompatibility: Structure, Functions, and Evolutionary Perspectives. Mol Phylogenet Evol. 2003;29:490506.
[ Links ]ROSENBERG HF. The Eosinophil Ribonucleases. Cell Mol Life Sci. 1998;54:795803.
[ Links ]RUGELES MT, TRUBEY CM, BEDOYA VI, PINTO LA, OPPENHEIM JJ, RYBAK SM, et al. Ribonuclease is Partly Responsible for the HIV1 Inhibitory Effect Activated by HLA Alloantigen Recognition. AIDS. 2003;17:481486.
[ Links ]SAXENA SK, GRAVELL M, WU YN, MIKULSKI SM, SHOGEN K, ARDELT W, et al. Inhibition of HIV1 Production and Selective Degradation of Viral RNA by an Amphibian Ribonuclease. J Biol Chem. 1996;34:2078320788.
[ Links ]SHAPIRO R, VALLEE BL. Interaction of Human Placental Ribonuclease with Placental Ribonuclease Inhibitor. Biochemistry. 1991;30:22462225.
[ Links ]SIERAKOWSKA H, SHUGAR D. Mammalian Nucleolytic Enzymes. Prog Nucleic Acid Res Mol Biol. 1977;20:59130.
[ Links ]SOOCHIN C, JAAP J, BEINTEMA B, ZHANG J. The Ribonuclease A Superfamily of Mammals and Birds: Identifying New Members and Tracing Evolutionary Histories. Genomics. 2005;85:208220
[ Links ]SORRENTINO S, GLITZ DG. Ribonuclease Activity and Substrate Preference of Human Eosinophil Cationic Protein (ECP). FEBS Lett. 1991;288:2326.
[ Links ]SORRENTINO S, LIBONATI M. Human Pancreatic Type and NonPancreatic Type Ribonucleases: a Direct SidebySide Comparison of Their Catalytic Properties. Arch Biochem Biophys. 1994;312:340348.
[ Links ]SORRENTINO S, LIBONATI M. StructureFunction Relationships in Human Ribonucleases: Main Distinctive Features of the Major RNase types. FEBS Lett. 1997;404:15.
[ Links ]SORRENTINO S, TUCKER GK, GLITZ DG. Purification and Characterization of a Ribonuclease from Human Liver. J Biol Chem. 1988;263:1612516131.
[ Links ]SORRENTINO S, NADDEO M, RUSSO A, D’ALESSIO G. Degradation of DoubleStranded RNA by Human Pancreatic Ribonuclease: Crucial Role of Noncatalytic Basic Amino Acid Residues. Biochemistry. 2003;42:1018210190.
SUGAI T, SAKIYAMA Y, MATUMOTO S. Eosinophil Cationic Protein in Peripheral Blood of Pediatric Patients with Allergic Diseases. Clin Exp Allergy. 1992;22:275281.
[ Links ]SUR S, GLITZ DG, KITA H, KUJAWA SM, PETERSON EA, WEILER DA, et al. Localization of EosinophilDerived Neurotoxin and Eosinophil Cationic Protein in Neutrophilic Leukocytes. J Leukoc Biol. 1998;63:715722.
[ Links ]TISCHENDORF FW, BRATTIG NW, BÜTTNER DW, PIEPER A, LINTZEL M. Serum Levels of Eosinophil Cationic Protein, EosinophilDerived Neurotoxin and Myeloperoxidase in Infections with Filariae and Schistosomes. Acta Trop. 1996;62:171182.
[ Links ]VENGE P, DAHL R, HALLGREN R. Enhancement of F XIIDependent Reactions by Eosinophil Cationic Protein. Thromb Res. 1979;14:641649.
[ Links ]WATERS LS, TAVERNE J, TAI PC, SPRY CJ, TARGETT GA, LAYFAIR JH. Killing of Plasmodium falciparum by Eosinophil Secretory Products. Infect Immun. 1987;55:877881.
[ Links ]WEICKMANN JL, ELSON M, GLITZ DG. Purification and Characterization of Human Pancreatic Ribonuclease. Biochemistry. 1981;20:12721278.
[ Links ]WEICKMANN JL, OLSON EM, GLITZ DG. Immunological Assay of Pancreatic Ribonuclease in Serum as an Indicator of Pancreatic Cancer. Cancer Res. 1984;44:16821687.
[ Links ]WU Y, MIKULSKI SM, ARDELT W, RYBAK SM, YOULE RJ. A Cytotoxic Ribonuclease. Study of the Mechanism of Onconase Cytotoxicity. J Biol Chem. 1993;268:1068610693.
[ Links ]WU Ch, CHANG H, CHANG MD. MembraneBound Carboxypeptidase E Facilitates the Entry of Eosinophil Cationic Protein into Neuroendocrine Cells. J. Biochem. 2004;382,841848.
[ Links ]YASUDA T, MIZUTA K, SATO W, KISHI K. Purification and Characterization of a Ribonuclease from Human Spleen. Immunological and Enzymological Comparison with Nonsecretory Ribonuclease from Human Urine. Eur J Biochem. 1990;191:523529.
[ Links ]YOUNG JDE, PETERSON CGB, VENGE P, COHN ZA. Mechanism of Membrane Damage Mediated by Human Eosinophil Cationic Protein. Nature. 1986;321:613616.
[ Links ]ZELEPUGA EA, PONOMAREVA RB, KOLIKOV VM, PAUTOV VD, SHEVELEVA TV, MEDVEDEV ML, et al. Conjugates of Pancreatic Ribonuclease and LigandFree Human Serum Albumin. Biomed Khim. 2003;49:588596.
[ Links ] ]]>