Introduction
One of the symbiotic associations with the greatest geographic and botanical distributions is the interaction between arbuscular mycorrhizal fungi (AMF), which covers more than 80% of plant species and is found in a great diversity of ecosystems (Brachmann and Parniske, 2006; Bonfante and Genre, 2008). A bi-directional exchange of nutrients is the basis of this association (Breuninger and Requena, 2004; Genre et al., 2005, 2008), which favors plant nutrition and plant tolerance to biotic or abiotic stress (Van der Heijden and Sanders, 2002; Smith and Read, 2008; Smith and Smith, 2011). To understand this complex symbiotic association, it is necessary to know the environment in which it is developed and the factors that affect the establishment and functioning of AMF communities. Many factors affect the dynamics of this symbiosis, such as geophysical factors (i.e. altitude) or the different stages of plant development that influence the composition of AMF communities (Husband et al., 2002a, b; Oehl et al., 2006; Senés et al., 2014). Senés et al. (2014) evaluated the composition of AMF communities in the Peruvian Andes in potato crops at four different altitudes from 2,658 to 4,075 m a.s.l., and they found a direct relationship between altitude and the community composition of AMF species. Some factors that affect the structure, diversity and distribution of AMF communities are soil microorganism populations (Garbaye, 1994; Gehring and Whitham, 2002), agricultural practices, such as logging, burning, use of fertilizer and tillage (Jansa et al., 2003), and indirectly microclimate and topography (Johnson, et al., 1992; Kernaghan, 2005).
Cape gooseberry (Physalis peruviana L.) belongs to the Solanaceae family and is distributed in the wild highlands of the South American Andes (Pérez, 1996; Trillos et al., 2008), its place of origin (Morton, 1987; Bartholomáus et al., 1990; Medina, 1991; Criollo and Ibarra, 1992; Chia et al., 1997). In Colombia, the optimal conditions for its cultivation include altitudes between 2300 and 2800 m a.s.l., temperatures between 13 and 17°C, relative humidity between 70 and 80%, and precipitation between 600 and 1100 mm/year (Fischer, 2000; Espinal et al., 2005). The interest in working with this plant species is based on the fact that the plant is native to the Andes and has a wide range of edapho-climatic adaptations (Fischer, 2000) that may be related to its ability to associate with AMF.
The objective of this study was to evaluate AMF diversity in Andean soils cultivated with cape gooseberry to determine if the composition of AMF communities is modulated by altitude. The possible effect of altitude on the establishment of AMF communities is fundamental for understanding symbioses and finding behavioral patterns in AMF communities that would allow a better management of agroecosystems.
Materials and methods
Soil sampling
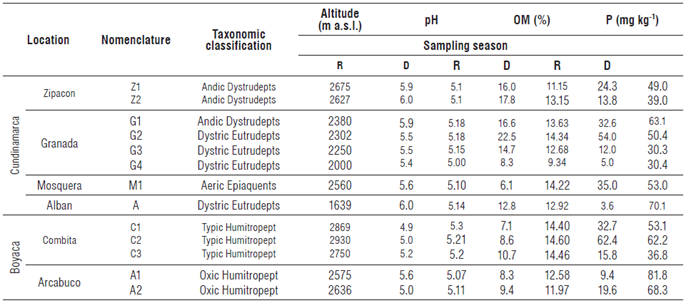
Sampling was performed on an altitudinal transect between 1500 and 3000 m a.s.l. Composite samples of cape gooseberry rhizospheric soils were collected at 13 sites. At each site 4 kg (15 subsamples) of soils were collected at a depth of 0-20 cm, in duplicate, for physicochemical and AMF analysis (Tab. 1). From each sample, 200 g were taken for analysis of the abundance and diversity of AMF spores, in duplicate. The remaining soil was stored to be used as inoculum or as a source of spores for a plant tramp assay. Two samplings were carried out: one in the rainy season (150-330 mm/month) and the other in the dry season (020 mm/month).
Isolation and identification of AMF spores
For each sample, the number of spores 10 g-1 of soil was determined according to the methodology described by Gerdermann and Nicholson (1963), with modifications. The percentage of AMF colonization was estimated using the Trypan Blue differential staining methodology by Phillips and Hayman (1970) and Giovannetti and Mosse (1980) with modifications. The taxonomic classification of the AMF was performed at the species level based on the morphology of the spores. The spores were isolated and arranged in sheets with polyvinyl lactic acid-glycerin (PVLG) (Koske and Tessier, 1983) and, in some cases, with a mixture (1:1 v/v) of PVLG with Melzer (Brundrett et al., 1994). The isolated and identified spores corresponded to the two sampling periods, dry season (<20 mm/month) and rainy season (150-350 mm/month). The classification codes of Schenck and Pérez (1990) and INVAM (International Culture Collection of Vesicular Arbuscular Mycorrhizal Fungi (http://invam.caf.wvu.edu/cultures/cultsearch.htm)) were used, along with scientific publications as support for the genus or species classification (Morton and Benny, 1990; Blazkowski, 1991; Morton and Redecker, 2001; Schüffler et al., 2001; Oehl and Sieverding, 2004; Walker and Schüffler, 2004; Blaskowski et al., 2006, 2008; Sieverding and Oehl, 2006; Palenzuela et al., 2008; Alves Da Silva et al., 2009; Oehl et al., 2008, 2010, 2011 a, b, c, d; Goto, et al., 2011; Redecker et al., 2013).
Diversity index
Density (DE), richness (R), relative abundance (RA), isolation frequency (IF), Shanon-Wiener diversity index (H'), uniformity index (E), Simpson dominance index (D) and Simpson-Gini diversity index (Y) were used to determine AMF diversity in each sample and between all 13 samples collected. Indices were applied at the species level (Franke-Snyder et al., 2001; Zhang et al., 2004; Rodríguez et al., 2005; Zhao and Zhao, 2007; Kwasna et al., 2008; Chifflot et al., 2009).
Results and discussion
AMF Communities
The presence of plant-AMF symbiotic associations was measured as percentages of colonization of cape gooseberry roots and the total number of spores present in the rhizosphere of the plant to verify the interaction of these AMF communities with the plant.
The results showed that the highest percentages of colonization occurred during the rainy season for most municipalities (Fig. 1), except for G1, A2 and A, with values between 7.4 and 68.5%. Municipalities G4, A1, C3 and G2 were noted for having a higher percentage of colonization, and lower values were seen in G1, A2 and Z2. In the dry season, the colonization range was between 2 and 22%. The higher values were recorded in G1, G4, A and A2, and the lowest were seen in G3, C1 and Z2 (Fig.1). In all samples evaluated, the presence of AMF associated with roots of cape gooseberry plants was registered, independent of colonization rates, demonstrating that it is a mycotrophic species.
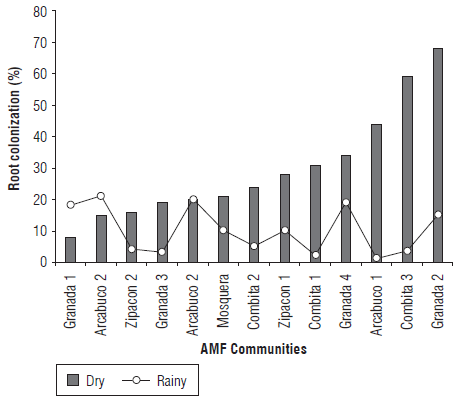
FIGURE 1 Natural colonization levels (%) of cape gooseberry roots by AMF at two sampling seasons, dry and rainy.
The number of spores varied between 20 and 120 spores 10 g-1 of soil during the rainy season. The higher spore number values were seen in municipalities A and C1, and the smallest amount was recorded in Z1. However, the dry season presented the highest spore values, between 170 and 1531 spores 10 g-1 of soil, in G3 and G4, respectively (Fig. 2).
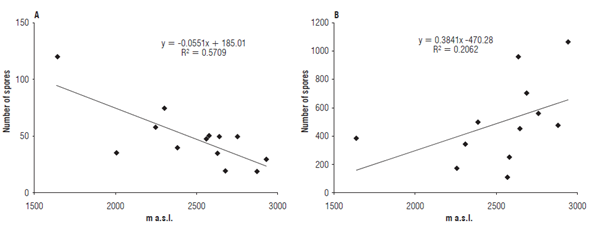
FIGURE 2 Relationship between altitude and number of AMF spores grown with cape gooseberry in the provinces of Cundinamarca and Boyaca. a) Rainy season and b) dry season.
During the rainy season, a negative correlation was observed in the number of spores, since the highest number of spores was recorded at the lowest altitudes. During the dry season, the correlation was positive, since a greater number of spores was observed at higher altitudes. These results agree with publications that show how, under water stress conditions, AMF sporulate by increasing the production of spores g-1 of soil (Caproni et al., 2003; Roveda et al., 2012; Pagano et al., 2013).
Taxonomic identification of AMF
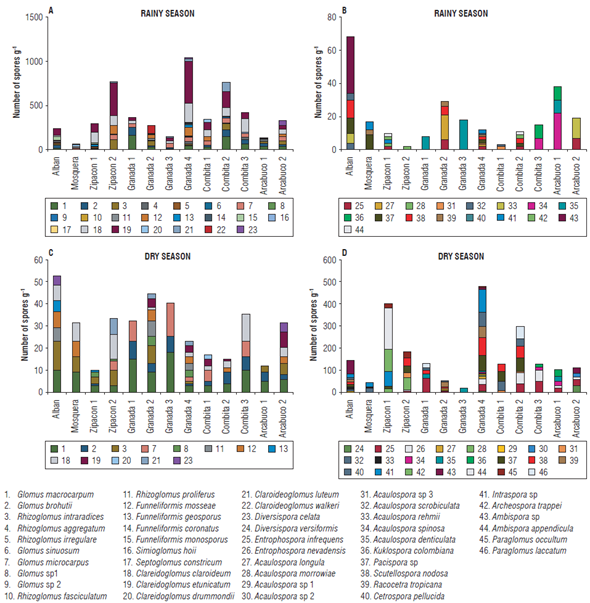
A total of 46 species, grouped in 16 genera, 11 families and 5 orders, were taxonomically identified, illustrating the great diversity of AMF found in the Colombian Andean soils. The distribution of species and genera of AMF identified in each of the evaluated locations during dry and rainy seasons can be seen in Table 2. The results show 23 species for Alban, 12 for Mosquera, 25 for Zipacon and 35 for Granada, the latter presenting the greatest diversity of AMF species. Three types of spores that had not previously been described were found: two of them corresponded to the genus Glomus and the other was found from the genus Acaulospora (Personal communication from F. Oehl and E. Sieverding), which were isolated from the soils of Granada, Zipacon and Combita. In the altitudinal transect (16362675 m a.s.l.) of the province of Cundinamarca, between 4 and 18 species were found in the soils during the rainy season and between 18 and 33 were found in the dry season in consolidated zones of cape gooseberry production (more than 20 years). For the altitudinal transect of the province of Boyaca (2572 to 2869 m a.s.l.), between 6 and 13 species were identified in the rainy season and between 11 and 17 in the dry season, in a zone that is considered as new for cape gooseberry cultivation (between 5 and 7 years).
TABLE 2 Distribution of AMF genus and species in soil samples cultivated with cape gooseberry in an altitudinal transect between 1636 and 2869 m a.s.l. (R: Rainy, D: Dry).
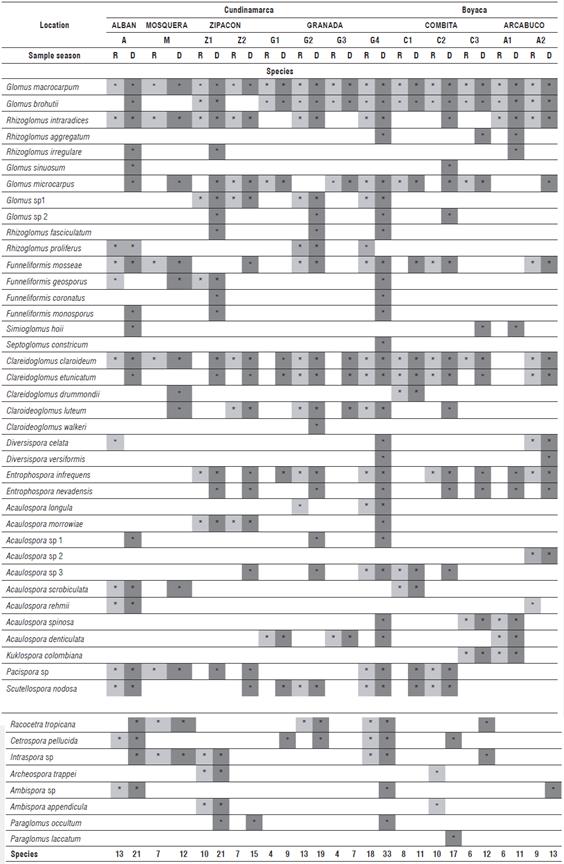
It is important to point out that five AMF species were not identified in soils of the consolidated zones of cape gooseberry production in Cundinamarca: Acaulospora sp2, A. scrobiculata, A. rehmii, A. colombiana and Paraglomus laccatum. In a recent crop production in the province of Boyaca, 13 AMF species were not identified: Glomus sp1 , Rhizoglomus fasciculatum, R. proliferum, Funneliformis geosporum, F. coronatus, F. monosporum, Septoglomus constrictum, Claroideoglomus walkerii, Acaulospora longula, A. morrowiae, Acaulospora sp1, Intraspora sp. and P. occultum. Mahdai et al. (2017) reported a higher density of AMF spores associated with a coffee crop (256 spores 100 g-1 soil) at higher altitudes (1400 m a.s.l.) as compared to lower altitudes (700 m a.s.l.) in the mountains of Saudi Arabia.
AMF species
The total number of species in the rainy season was 31, while in the dry season it was 46. Regardless of the sampling time, the highest number of species was observed in sample G4, followed by A, while the lowest values were observed in G1 and G3.
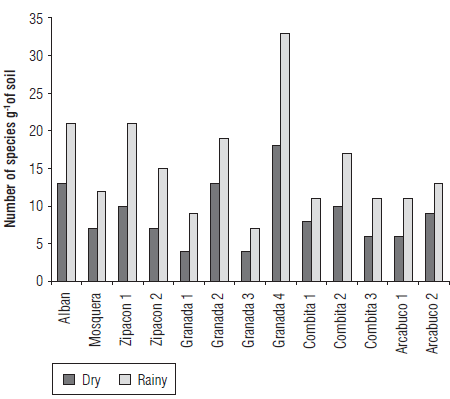
FIGURE 3 Number of AMF species (10 g-1 of soil) identified in the cape gooseberry crop in Cundinamarca and Boyaca at two sampling seasons, dry and rainy.
At the seasonal level, a high diversity in AMF communities was also observed, which was expressed as a higher number of spores, richness and relative abundance of species in the dry season compared to the wet season in which higher levels of root colonization were detected. Similar results have been obtained by several authors in the dry season (Pagano et al., 2013; Guadarrama et al., 2014; Rabelo et al., 2014) as well as in the wet season (Guadarrama and Álvarez-Sánchez, 1999). The seasonal variation of the communities was evident by the differences found in the number of species between the dry (46) and rainy (31) seasons, of which 32.6% of the species were not isolated in the rainy season. These results are in agreement with previous reports on seasonal variations of AMF communities (Courty et al., 2008; Davey et al., 2012; Bonfim et al., 2013; Guadarrama et al., 2014). Some authors have reported similar effects of the rainy season on root colonization to those obtained in the present study (Rabatin, 1979; Allen, 1983; Lodge, 1989; Miller, 2000; Miller and Sharitz, 2000), while other authors observed no effect (Bryla and Duniway, 1997; Ming and Hui, 1999). The presence of spores and the different levels of root colonization showed the existence of an active interaction between AMF and cape gooseberry plants in the Andean soils. The dry season increased the number of spores.
In the present study, a high number of spores was found in 10 g of soil, both in the rainy season (20-120 spores g-1) and dry season (170-1531 spores g-1). These values were higher than those reported by Jayachandran and Shetty (2003) for the wetlands of the Everglades (18-124 spores g-1) and by Lopes et al. (2013) in humid forests and pastures in Brazil (2.5 and 77.9 spores g-1), where the number of species varied between 31 and 46 for the rainy and dry seasons, respectively. These values are similar to those reported in the Chilean Andes (39 species) (Castillo, 2005; Castillo et al., 2005), in the Amazonian-pasture forest in Brazil (36 species) (Lopes et al., 2013), and in the forests of Mexico (37 species) (Violi et al., 2008). A lower diversity of species has been reported in the dunes in Brazil (25 species) (Stümer et al., 2013), forests (13-29 species) and pasture lands (18 species) of Mexico (Gavito et al., 2008; Fernandes et al., 2009; Guadarrama et al., 2014), tropical humid forests of Colombia (18 species) (Peña-Venegas et al., 2007), forests in Brazil (Aidar et al., 2004; Zandavalli et al., 2008; Moreira et al., 2009; Bonfim et al., 2013; Rabelo et al., 2014) and in general in various studies that have demonstrated a range of 12-26 species 10 g-1 of AMF (Wilson et al., 1992; Wang et al., 2008).
The high number of spores identified in the present study, associated with high species diversity from the ecological point of view, is a reflection of the history of the establishment of communities in a specific environment. It can be considered a reserve bank that may contains AMF adapted to various environmental conditions with the potential to associate with different hosts at a particular moment in time, with different growth strategies and adaptive mechanisms to the dynamic changes of the environment (Hijri et al., 2006; Oehl et al., 2006; Moebius-Clune et al., 2013). From the agronomic point of view, the high diversity represents the high potential presented by the Andean ecosystems for the establishment of symbiotic associations. Although this was an analysis of agroecosystems with semi-intensive use, AMF diversity was high, contrary to that reported by different authors on the reduction of AMF diversity in agricultural systems (Mason et al., 1992; Munyanziza et al., 1997; Cowden and Peterson, 2009).This study verified the presence of "generalist" species (according to Oehl et al., 2003). These are AMF species that can be isolated under different soil and climatic conditions, in contrast to "specialist" species that only occur under specific soil or climatic conditions. Generalist species can be isolated under different edaphic conditions and at different altitudes, showing their high tolerance for diverse soil and climatic conditions, including: G. macrocarpum; G. brouhtii; G. microcarpum, C. claroideum, C. luteum, E. infrequens and E. nevadensis. The existence of "generalist" species has been reported by different authors (Oehl et al., 2003, 2010; Castillo, 2005; Öpik et al, 2006; Guadarrama et al., 2007; Stümer and Siqueira, 2008). Glomus macrocarpum has been reported as a "generalist" species in several ecosystems and agroecosystems (Oehl et al., 2004; Castillo et al., 2005; Oliveira Freitas et al., 2014; Rabelo et al., 2014). Guadarrama et al. (2014) identified 10 species considered as generalist, while Rabelo et al. (2014) identified 4 generalist species among 40 identified species.
In the case of fungi of the genus Glomus, their predominance in diverse edaphoclimatic conditions was reported in several plant species (Gavito et al., 2008; Wang et al., 2008; Schnoor et al., 2011; Boonlue et al., 2012; Mahdhi et al., 2017). The high presence of species of the genus Glomus was probably associated with their high sporulation capacity favoring the colonization of roots in different environments, especially in environments with agronomic operations (Caprioni et al., 2003; Rabelo et al., 2014). This quality results in the species of this genus being more abundant in manmade systems, especially in agroecosystems like the one in the present study, where fungi of the order Glomerales and specifically species of the genus Glomus showed wide dispersion and high abundance in soils of the Colombian Andes.
Other species of fungal genera, such as Acaulospora and Glomus, predominate in the Chilean Andes (Castillo, 2005; Castillo, et al., 2005), showing similarity to those found in this study in Colombia, possibly because of the affinity of some of the edaphic characteristics, such as the presence of volcanic ash (allophane), soil acidity, low P contents in the soil and high organic matter (OM), although they differ in altitude and latitude. In contrast, in studies in the Peruvian Andes Sénes et al. (2014) found that Funneliformis mossseae was the most predominant species in both the soil and root of potato plants. This species is an early stage colonizer and seems to be adapted to frequent soil disturbances, such as contamination by hydrocarbons, fungicides, heavy metals, salinity, drought and cold climates (Abdel-Azeem et al., 2007; Huang et al., 2007; Zarei et al., 2010; Hassan et al., 2011; Krishnamoorthy et al, 2014).
The increment of the available phosphorus in the soil produced a reduction of AMF root colonization, which was previously reported by Jansa et al. (2009) and Selvam and Mahadevan (2002). Low levels of phosphorus in the soil favor and promote the establishment and development of symbiosis, and therefore, AMF multiplication. Phosphorus deficiency in the soil is one of the main activators of recognition signals between plants and HFMA (Ramírez and Rodriguez, 2010). It was found that 32% of AMF species, especially Glomerales, were favored by increasing the contents of this element in the dry season, while 17.4% were negatively affected in the wet season. This type of interaction has been previously reported (Jeffries et al., 1988; Sieverding, 1991; Oehl et al., 2003; Landis et al., 2004; Uhlmann, et al., 2004; Bashan et al., 2007), showing correlations between phosphorus contents and richness and abundance of AFM species. There are reports of the presence of HFMA in soils with high phosphorus contents (Davidson and Christensen, 1977; Allen and MacMahon, 1985), showing the great versatility of adaptation that AMFs have. The soils of the Andean region have high phosphorus fixation, so at relatively high phosphorus levels, but with low availability, the abundance of certain HFMA species can be favored.
The tolerance of some species to edaphoclimatic conditions is a desirable characteristic of species considered "generalist" since it allows species to be easily adapted to changes in the environment. This is a common situation in agro-ecosystems, where the edaphic environment is modified by cultural practices associated with crops. In addition, the identification and use of "generalist" species can facilitate the establishment of symbiosis under different conditions in cape gooseberry because of the high mobility of the crop as an escape mechanism for diseases. The high frequency of isolates along the altitudinal transect of some species showed the high adaptability of these species to conditions of biotic stress and strong changes in agroecosystems from practices such as fertilizer applications (Sturmer and Siqueira, 2008; Zangaro and Moreira, 2010). In this study, species such as G. macrocarpum were seen under rainfall conditions below 20 mm/month as well as with rainfall between 150 and 300 mm/month, while other species only occurred in the dry season.
In the case of AMF species considered as "specialists", because their presence is associated with specific conditions (climate, soil or both), it was found that R. agregatum, R. irregulare, G. sinuosum, two species of Glomus sp., F. coronatus, F. monosporum, S. hoi, S. constrictum, C. walkeri, E. nevadensis, Acaulospora sp, P. occultum and P. laccatum were exclusively associated with climate, while S. constrictum, C. walkierii, Acaulospora sp2 and P. laccatum were associated with the soil type, specifically soils cultivated with Physalis peruviana. These AMF characteristics, which present a "specialist" behavior for soil types, climate and moisture regimen, have been reported in AMF community analysis studies in tropical ecosystems, such as humid forests and semi-arid zones of Brazil and Africa, in tropical savannas and the Swiss Alps (Landis et al., 2004; Uhlmann et al., 2004; Lekberg et al., 2007; Oehl, et al., 2010). Although they are not ecologically similar to the Colombian Andes, they showed a trend of AMF behavior. Rabelo et al. (2014) identified 26 intermediate and 19 exclusive species or specialists in communities composed of 40 species. The identification of "specialist" species allows species to adapt to specific stresses, which can occur both in space and time because of anthropic intervention or global or local phenomena, such as climate change and variation.
Relative abundance and species richness of AMF
The relative abundance exhibited higher values in the dry season than in the rainy season, related to the greater number of species collected and identified at that time. A relative abundance of more than 71% was obtained for the sample from G4, whereas for that same sample in the rainy season, only 60% was reached. The samples from G1 and G3 had the lowest relative abundances. It is evident that in C1, A2 and G2, the relative abundance of the species was higher in the rainy season, while in the other samples it was always higher in the dry season (Fig. 4).
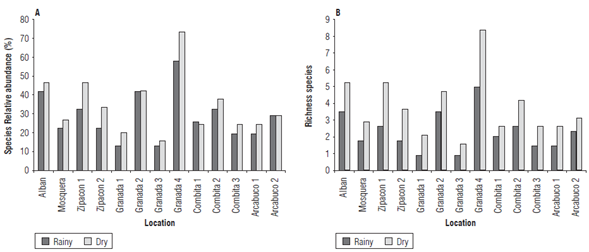
FIGURE 4 Relative abundance (a) and richness (b) of AMF species (g-1 of soil) in soils cultivated with cape gooseberry in Cundinamarca and Boyaca in rainy and dry seasons.
Species richness varied between 1.57 and 8.30 in the dry season and between 0.87 and 4.95 in the rainy season, reflecting the differences between the two sampling periods. The highest species richness values were observed in the samples from A and G4, and the lowest values in G1 and G3.
Frequency of species isolates in soil samples
In the rainy season, the frequency of isolation ranged between 7.6% from isolated species in a sample to 100% of isolated species in all samples. According to Oehl et al. (2003), this analysis allows the determination of "genera-list" species (15%), such as G. macrocarpum, G. brohutii, C. claroide, C. etunicatum, G. microcarpum, G. intraradices and E. infrequens, which were isolated from a high number of samples in rainy and dry seasons with a clear predominance of Glomus species in terms of isolation frequencies. The species that can be considered as "specialists" corresponded to 8.7% of the species: Septoglomus constrictum, Claroideoglomus walkierii, Acaulospora sp2 and Paraglomus laccatum, as they were isolated in a single sample and in one single season.
Of the total species 45.6% showed the highest frequency of isolation in the dry season (21), and only 4 showed higher frequency in the rainy season (Rhizoglomusproliferum, A. longula, A. rehmii and Archeospora tropeii). We observed that only 4 species (G. macrocarpum, Glomus sp2, Diversispora celata and Kuklospora colombiana) had the same frequency of isolation in the dry and rainy seasons, compared to the total species isolated in each season.
Only 32.6% of the species (15) were isolated in the dry season: Rhizoglomus agregatum, R. irregulare, G. sinuosum, two species of Glomus sp, F. coronatus, F. monosporum, Simioglomus hoi, S. constrictum, C. walkeri, E. nevadensis, Acaulospora sp, P. occultum and P. laccatum. These were "specialists" for the wet regime rather than for soil type or altitude. According to the scale proposed by Zhang et al. (2004), the dominant species in the rainy season were G. macrocarpum, G. brohutii, G. intraradices and C. claroi-deum, and in the dry season G. macrocarpum, G. brohutii, G. intraradices, G. microcarpus, F. mosseae, C. claraideum, C. etunicatum, E. infrequens, E.nevadensis, Pacispora sp. and Scutellospora sp. The rare species in the rainy season were C. drummondii and Ambispora sp., and for the dry season, they were S.constrictum, C. walkeri, A. longula, Acaulospora sp2, A.rehmi, Archeospora troppei, Ambispora appendicula and P. laccatum. Regardless of the season, G. macrocarpum, G. brohutii, G. intraradices and C. claroi-deum were dominant, and most of the rare species in the dry season were not isolated in the rainy season, except for A. longula, A. rehmi, Archeospora troppei and Ambispora appendicula.
Figure 5 shows the abundance of spores for each of the identified species in each community. We determined that although a species may be present in all evaluated communities, its abundance can vary widely.
Shannon-Wiener Index (H ')
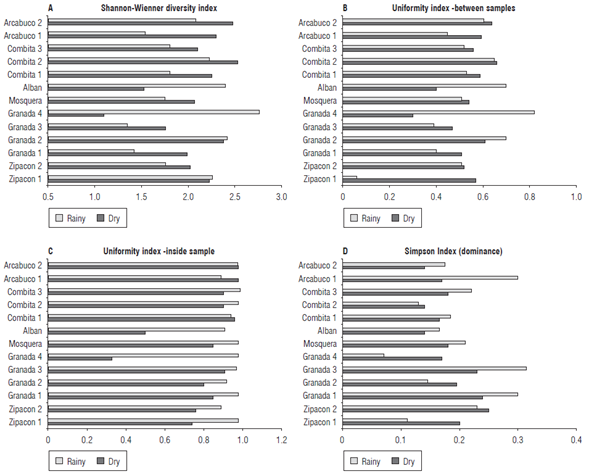
In the rainy season, Shannon-Wiener diversity indexes were found between 1.33 and 2.8, considered as mean values according to Gove et al. (1999). The locations G4, A, Z1, C2 and A2 had mean levels of diversity related to the number of species found in the samples (Fig. 6A). In the dry season, the values were similar, with variations between 1.12 and 2.5 with the highest levels of diversity in A2, A1, C2, C1, M, G2 and Z1. It is interesting to observe how in the same location but in a different sampling area the levels of diversity may vary widely either by soil type or agronomic management of the lots.
Uniformity index
This index had a range between 0 and 1, with 1 as the maximum value when all species are present in equal abundance and it decreases when the dominance extent of a species or morphotype occurs (Hurlbert, 1971).
Two measures of uniformity were considered: between samples and inside each sample. Results are presented in Figure 6 (B and C). In the first case, for the rainy season differences in uniformity between the samples were observed when the identified species had variations between 0.06 in Z1 and 0.82 in G4, representing values of low uniformity for Z1 and high uniformity for G4 in relation to the other sampling sites according to Hurlbert (1971). This indicates that Z1 had few species with high disparity with the other samples. In the dry season, there was greater homogeneity between the samples with values between 0.29 (G4) and 0.65 (A2), showing a more homogeneous distribution of the species. When measuring the uniformity per sample (ratio of number of spores to total spores in the sample), in the rainy season, there was a low number of spores the values were very close to 1 in all of the samples reflecting similar values of spores per species in each of the samples. In the dry season, the values were high but with greater variations, with a range between 0.32 in G4 and 0.91 in G3, showing wide variations in the number of spores of each species present in the sample.
Simpson's Dominance Index:
Simpson's Dominance Index shows the highest values for samples G1, G3 and A1 during the rainy season and for Z2, G1 and G3 in the dry season, signifying that these samples had dominant species (Bouza and Covarrubias, 2005). This is consistent with the results of the Uniformity Index since these were the same samples that showed lower uniformity values (Fig. 6D).
The estimated diversity indexes corroborated the hypothesis of high diversity of AMF in these systems, with low-average levels of uniformity between and within the analyzed samples and with species dominance in some of the analyzed communities, especially in those that had a low number of species. The Shannon-Wiener diversity index values recorded for cape gooseberry (1.1 to 2.8) showed that, while the host affects AMF diversity (Vandenkoornhuyse et al., 2002), edaphic conditions and altitude also play an important role. This is evident in the ranges of diversity found in the evaluated altitudinal transect where the variations were mainly environmental and not from the host. However, the host component can be evaluated by comparing the values of the present study with those obtained by Helgason et al. (1998) and Tanja et al. (2004), who reported ranges from 0.4 in agricultural soils to 2.3 in forests, with higher values in cape gooseberry. It is important to consider the characteristics of the cape gooseberry crop since it is a species that is cultivated in agricultural fields but is also in the process of domestication. Cape gooseberry originated from Andean ecosystems, where the diversity centers of the species are found and has been adapted to these ecosystems with restrictive soil-climatic conditions, possibly through co-evolution processes with AMF. Additionally, these results indicate that the tropical Andes of Colombia are a niche with broad AMF diversity.
The previous results confirm the existence of high AMF diversity in the ecosystems of the tropical Andes. Although only cape gooseberry soils were sampled, the number of AMF species was higher to that identified with a greater variety of hosts as mentioned previously. The identification of "generalist" or "specialist" species is very important for the establishment of the AMF-cape gooseberry association since this is a "nomadic" crop that changes with location, soil, climate and altitude. Due to this, AMF species with high adaptability to different climatic and altitude conditions may have a greater possibility of establishing symbiosis than those affected by edaphic changes. Knowing the factors that can affect the abundance of species allows the creation of practices that favor the presence of species of interest for an ecosystem or an agroecosystem.
Conclusions
This research contributed to our knowledge of AMF diversity in the cape gooseberry (Physalis peruviana L.) production system in the evaluated altitudinal transect (1500-3000 m a.s.l.). It also determined the relationship between diversity, abundance and composition of communities with the characteristics of the soils in which these communities of fungi associated with cape gooseberry plants are established. This is the first time this kind of research has been carried out in the Andes.
The presence of spores and different levels of root colonization showed the existence of an active interaction between AMF and cape gooseberry plants in Andean soils. This high diversity can be considered as a reserve bank of AMF species adapted to the conditions in the Colombian Andes, which will allow the establishment of symbiotic associations for sustainable and competitive agricultural systems.