Introduction
Vitamin D3 (VD) is a prohormone made principally in the skin from 7-dehydrocholesterol through conformational changes associated with ultraviolet B radiation, depending on the intensity of and length of exposure to these rays, and varies according to altitude and season 1-3. Vitamin D3 may also be obtained from the diet or from nutritional supplements, with these sources having less biological importance 4.
After VD is produced, it is transported in the bloodstream by vitamin D binding protein (DBP), which is capable of transporting all the VD-associated metabolites, with a greater affinity for the 25-hydroxyvitamin D [25(OH)D] form (2, 5, 6). The VD/DBP complex is transported to the liver, where the first hydroxylation occurs, mediated primarily by CYP2R1, producing 25(OH)D. This, in turn, is transported to the proximal convoluted tubule of the kidney where the second hydroxylation takes place by CYP27B1, thus synthesizing 1,25-dihydroxyvitamin D [1,25(OH)2D3], the biologically active form of the hormone 1,2,7.
The biological actions of 1,25(OH)2D3 have classically been linked to bone mineral metabolism. However, new functions have been described, including energy metabolism homeostasis, mediated by VD receptor stimulation, which directly modifies gene transcription and direct modulation of the cytoplasmic enzyme complex function in various types of cells 1,7-9.
There is no consensus regarding normal levels of 25(OH) D. However, most societies concur in defining a serum con centration of 30 ng/mL or more as sufficient, 20-29 ng/mL insufficient, and less than 20 ng/mL deficient 10-12. Close to one billion people worldwide are estimated to have VD in sufficiency or deficiency. In the United States, the prevalence is 50-80% of the general population, being more common in the elderly 10,13,14. In Colombia, a prevalence of insuf ficiency and deficiency of 55.1% and 16.6%, respectively, has been found in postmenopausal patients 15.
Low VD concentrations have been directly related to rickets and osteoporosis; colon, breast and prostate cancer; autoim mune diseases such as multiple sclerosis and type 1 diabetes mellitus; and cardiovascular diseases such as arterial hyper tension, type 2 diabetes mellitus and heart failure 13,16-19.
On the other hand, metabolic syndrome (MS) has been defined as a group of clinical and laboratory disorders, and is considered to be a primary event for the development of atherosclerotic disease 20,21. The global prevalence lies between 20 and 50%, but varies according to the population studied 20,22,23. Individuals with MS have five times the risk of type 2 diabetes mellitus, two to four times the risk of myocardial infarction and cerebrovascular accidents, two times the risk of cardiovascular disease and a greater risk of mortality compared to individuals without MS 21,24-27. Therefore, the search for new factors involved in the pathophysiology of MS, especially modifiable factors, has increased in recent years.
In addition, a relationship has been found between the serum concentration of VD and MS; however, the evidence is controversial in this regard. Thus, this study was proposed to characterize VD levels and correlate them with the presence of MS and its various components: waist circumference, glucose, arterial pressure, triglycerides and high-density lipoprotein cholesterol (HDL-C), as well as with the concentration of C-reactive protein (CRP).
Materials and methods
This was a cross-sectional study using convenience sam pling, including a total of 109 individuals. All participants were assessed by at least one physician and one nutritionist for the clinical evaluation and anthropometric measurements. In accordance with the current guidelines, obesity was defined as a BMI equal to or greater than 30 kg/ m228,29. All subjects with a diagnosis of diabetes mellitus, arterial hypertension, cardiovascular disease or other type of chronic disease, or who had taken medications on a regular basis during the previous year were excluded from the study.
One blood sample was taken to measure triglycerides (TGs), total cholesterol (TC), HDL-C, low-density lipoprotein cholesterol (LDL-C), glucose and insulin. The Homeostasis Model Assessment - Insulin Resistance index (HOMA-IR) was calculated using Matthews et al.’s method (30). The serum concentration of 25(OH)D was determined by electrochemi luminescence using a commercially available kit, according to the manufacturer’s protocol (LIAISON® 25 OH Vitamin D Total Assay); insufficiency and deficiency were defined as serum values between 20-29 ng/mL and less than 20 ng/mL, respectively 10-11.
The diagnosis of metabolic syndrome was established when three of the following criteria were met: waist cir cumference equal to or greater than 90 cm (cut-off point recommended for South American men), triglycerides equal to or greater than 150 mg/dL, HDL cholesterol less than 40 mg/dL, systolic arterial pressure equal to or greater than 130 mmHg or diastolic equal to or greater than 85 mmHg, and fasting glucose equal to or greater than 100 mg/dL 21,27.
The study was approved by the ethics committee of the Universidad Nacional de Colombia’s school of medicine. All participants received complete information regarding the study protocol, questions were answered, and then the consent form was signed.
Normality for the comparison groups (metabolic syn drome or no MS) was determined by the Shapiro-Wilk and Kolmogorov-Smirnov tests. Subsequently, measures of central tendency were obtained: mean and standard deviation for the normally distributed group and median with interquartile range for the non-normal group. A nonparametric Wilcoxon rank test was used to compare variables between groups in non-normal cases and Student’s t-test was used for normal cases; p < 0.05 was used to determine statistically significant differences.
Finally, the two groups were correlated using the Pearson correlation for normally distributed variables, and Spearman’s correlation for non-normal variables, with their respective p-values.
Results
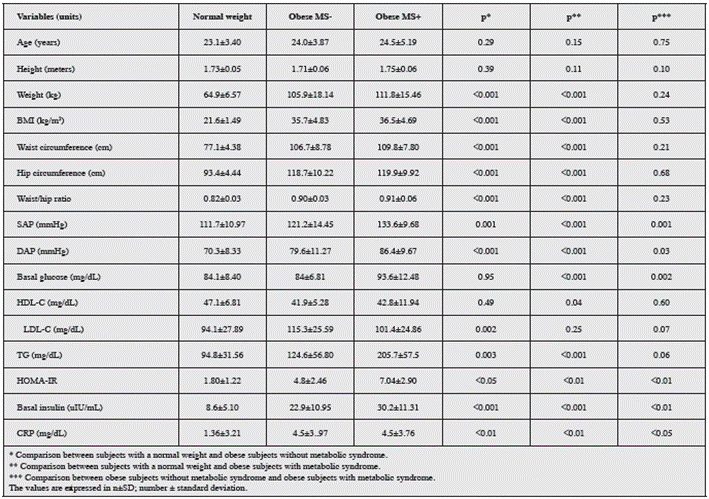
A total of 109 individuals were recruited for the study, with an average age of 23; of these, 62 had a weight within the normal range (normal weight) and the remaining 47 were in the obesity range. Of the obese individuals, 25 met at least three of the MS criteria (OMS+), while the other 22 met two or fewer criteria (OMS-). In the control group with normal weight individuals, none met the MS criteria.
Table 1 describes the baseline characteristics of the three groups; the anthropometric study found no significant differ ence in height, however, weight, BMI, waist circumference and hip circumference were significantly lower in the normal weight group than in both the obesity subgroups (p<0.01).
With regard to the parameters for defining MS, the mean waist circumference was 109.8±7.8 cm in the OSM+ group and 105.9±18.1 cm in the OSM- group, with no significant difference between the two (p=0.21); the same occurred with HDL-C levels (42.8±1.9 mg/dL vs 45.1±17.6 mg/dL p=0.6). The parameters which made a difference between obese indi viduals with and without MS were: systolic arterial pressure (133.6±9.7 mmHg vs 121.2±14.4 mmHg p=0.001), plasma glucose levels (93.6±12.5 mg/dL vs 84±6.8 mg/dL p=0.002) and diastolic arterial pressure (86.4±9.7 mmHg vs 79.6±11.3 mmHg p=0.03).
Furthermore, waist circumference, systolic and diastolic arterial pressure, LDL-C and triglyceride levels were sig nificantly elevated in obese subjects without MS criteria compared to normal weight subjects, with p<0.01 for all comparisons.
An assessment of insulin concentrations showed a sig nificant difference when all groups were compared, with the lowest values in normal weight subjects, intermediate values in OSM- and the highest values in OSM+, at 8.6±5.1 uIU/ mL, 2.9±10.9 uIU/mL and 30.2±11.3 uIU/mL, respectively, with the same phenomenon seen with the HOMA-IR (1.8±1.2, 4.8±2.5 and 7±2.9, respectively). The CRP levels were signifi cantly higher in OSM+ compared to OSM- (p<0.05), which, in turn, were greater than in normal weight individuals (p<0.01).
The mean serum level of 25-OHVD for the total popula tion was 30.6±8.3 ng/mL; 46.8% of the subjects were found to be sufficient, 45.9% insufficient and 7.3% deficient in this hormone, according to the previously described classification.
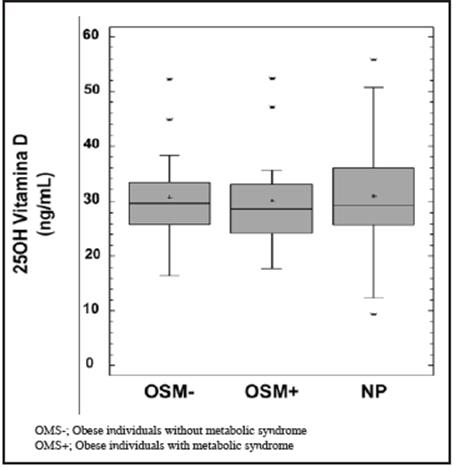
The mean plasma concentration of VD was 30.8±8.5 ng/ mL in normal weight subjects, 30.6±7.5 ng/mL in OSM- and 30.1±9.2 ng/mL in OSM+, with no significant difference between the subgroups studied (p=0.94) (Figure 1).
Lastly, a correlation matrix was constructed between the individual MS components and serum concentration of 25- OHVD for each group of obese individuals, with heterogenous results. The lowest levels of VD were related to higher systolic arterial pressure, waist circumference and serum concentra tions of triglycerides; however, the degree of correlation was weak, both in OSM- as well as OSM+ (Figures 2a, 2c and 2f). Serum glucose had a neutral correlation with VD concentra tion in both groups (Figure 2e), diastolic arterial pressure had a heterogenous correlation, being positive in OSM+ and negative in OSM- (Figure 2b).
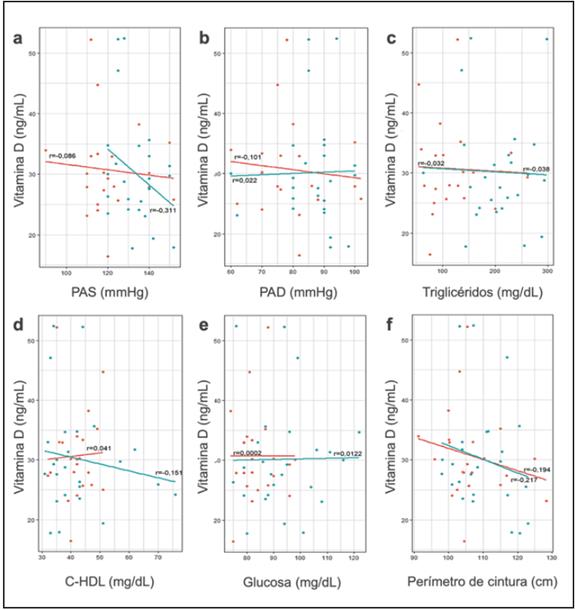
Figure 2 Diagram of the correlation between vitamin D concentration and the metabolic syndrome components in obese patients. Diagram of the correlation between the individual components of metabolic syndrome (a) systolic arterial pressure, (b) diastolic arterial pressure, (c) triglycerides, (d) HDL cholesterol, (e) basal glucose and (f) waist circumference and serum concentration of vitamin D. The red line corresponds to the obese patients without metabolic syndrome and the blue line to obese patients with metabolic syndrome. See text for more information.
Finally, the highest HDL cholesterol levels were related to higher vitamin D concentrations in obese individuals without MS, but to lower concentrations in obese individuals with MS (Figure 2d); despite the tendencies, the degree of correlation was weak for all the variables.
Discussion
The prevalence of MS within the study group was 21%, slightly lower than the 25-33% reported by the Asociación Latinoamericana de Diabetes [Latin American Association of Diabetes] 31, but similar to what has been described in Colombia, where highly variable data are reported, ranging from 13.2 to 72.7% depending on the criteria used for its definition 31-34. Central obesity, elevated systolic arterial pressure and high triglycerides were the most frequently af fected parameters, in accordance with what has been described in other studies 33.
There is little evidence in Latin America regarding the be havior of serum VD concentration in healthy adults. However, it is thought to be higher in tropical countries, given the greater sun exposure 35,36. Oliveri et al. reported a 25-OHVD deficit of 90% in healthy adults in Argentina, with a lower prevalence in the northern part of the country 37. A study of postmenopausal women with osteoporosis in Mexico, Chile and Brazil found inadequate levels of VD in 67, 50 and 42% of patients, respectively 35,38. In Colombia, reported data on the prevalence of hypovitaminosis D are scant. Molina et al. evaluated 205 postmenopausal women with osteoporosis, finding that 71.7% had VD concentrations under 30 ng/mL, similar to what was described by Devía et al. with a prevalence of 69.5% 39,40. In addition, Quintana et al. compared pa tients with rheumatoid arthritis and healthy controls, finding a prevalence of inadequate 25-OHVD levels of 98.5 and 72.8%, respectively 41. In our population, 53.2% of the subjects had VD concentrations below 30 ng/mL, lower than what was described in Colombia, Argentina and Mexico, partially explained by the lower age of the study patients together with the absence of comorbidities, this being one of the first studies to assess the status of serum vitamin D in a healthy young adult Colombian population.
A comparison of serum 25-OHVD levels in the MS and non-MS groups showed no significant difference, and there fore no relationship was found between VD insufficiency and deficiency and MS. There is still no consensus on the possible causal relationship between inadequate 25-OHVD levels and the risk of having MS and cardiovascular disease 42,43. However, some, mostly cross-sectional, studies have found an inverse relationship between these levels and MS, along with its isolated components, suggesting that pancreatic stimula tion of insulin secretion, inhibition of the renin-angiotensin-aldosterone system and stimulation of endothelial nitric oxide secretion, among others, could be some of the extra-skeletal effects of VD, which would explain the greater risk of car diovascular disease in these patients 1,43-46.
Moreover, as in the results of this study, several studies have found no significant relationship between serum con centration of 25-OHVD and metabolic state. The same is true for cardiovascular disease risk; a meta-analysis published by Sang et al. found no significant relationship in longitudinal studies, considering that the relationship described in the cross-sectional studies is probably not causal, and that many confounding factors may be influencing the results, with one of the most important factors being obesity 47.
Likewise, when the correlation studies between indepen dent MS variables and serum levels of VD were performed, there was no correlation with sufficient power to be considered relevant. Thus, there was considered to be no dependency between these variables, which could partially explain the negative results of intervention studies on the use of VD supplementation and the risk of developing MS, arterial hyper tension, diabetes mellitus and cardiovascular disease 48,49.
More recent studies have shown a relationship between low levels of vitamin D concentrations and the risk of cardiovascu lar morbidity and mortality, showing an increase of up to 7% in the risk of cardiovascular death with every 4 ng/mL lowering of VD plasma concentration, an thus being considered a non classical cardiovascular risk factor susceptible to intervention 49,50. However, recent clinical trials and meta-analyses have not been consistent in showing a clinically significant benefit for the strong outcomes in cardiovascular disease 51,52.
Among the study’s limitations, the convenience sampling is worth highlighting, as it does not allow a faithful represen tation of the population as a whole. Another limitation is the lack of data regarding sun exposure. Furthermore, the chang ing definitions of metabolic syndrome lead to a very variable epidemiology.
In conclusion, low concentrations of 25-hydroxyvitamin D were not associated with a greater probability of having MS, nor was there a strong correlation with individual MS variables in the study population. These results contribute information to the global body of knowledge and that which is being de veloped nationally regarding this controversy, supporting the relative independence of energy metabolism and the lack of benefits from 25-OHVD supplementation in the prevention of cardiovascular disease.











 text in
text in