Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista MVZ Córdoba
Print version ISSN 0122-0268On-line version ISSN 1909-0544
Rev.MVZ Cordoba vol.13 no.3 Córdoba Sept./Dec. 2008
ORIGINAL
ANÁLISIS DE DOS POBLACIONES DE CANINOS (Canis familiaris) DE LA RAZA CIMARRÓN URUGUAYO UTILIZANDO RAPDs COMO MARCADORES MOLECULARES
Silvia Llambí D,1* Ph.D, Rosa Gagliardi B, 1MV, Mónica Martínez T, 1MV, Jorge Estevez C,1MVM,
Ana Gorozurreta O, 1Br, Gabriela Costa De F, 1Br, Carolina Bianco Y, 1Lic,
Rody Artigas S, 1Br, María Victoria Arruga L, 2Ph.D.
1 University of Uruguay, UDELAR, Faculty of Veterinary, Laboratory of Genetics, Lasplaces 1550. CP 11600 Montevideo-Uruguay.
2 University of Zaragoza, Faculty of Veterinary, Laboratory of Cytogenetics and Molecular Genetics, Miguel Servet 177, CP 50013. Zaragoza. Spain. *Correspondencia: silvia.llambi@gmail.com
Recibido: Mayo 5 de 2008; Aceptado: Diciembre 10 de 2008
ABSTRACT
Objective. To analyze the genetic variability in two populations of the native canine breed "Uruguayan Cimarron" from different areas of the country. Materials and methods. DNA was isolated from 40 animals of the Cimarron breed. Using PCR, 11 RAPD (random amplified polymorphism of DNA) molecular markers were analyzed. Results. 14.9% of loci were polymorphic. We found an index of shared bands of 0.91, a genetic distance of 0.16 and a Nei's average diversity index of 0.08. Conclusions. Both populations present a high homogeneity and genetic identity for these markers suggesting that they belong to a common genetic nucleus.
Key words: Canis familiaris, genome, Uruguay.
RESUMEN
Objetivo. Analizar la variabilidad genética en dos poblaciones de caninos de la raza autóctona "Cimarrón uruguayo" provenientes de distintas zonas del País. Materiales y métodos. Se aisló ADN de 40 animales de la raza Cimarrón. Mediante la técnica de PCR se analizaron 11 marcadores moleculares RAPD (amplificación al azar de ADN polimórfico). Resultados. Se detectó un 14.9% de loci polimórficos. Se obtuvo un índice de bandas compartidas de 0.91, una distancia genética de 0.16 e índice promedio de diversidad de Nei's de 0.08. Conclusiones. Ambas poblaciones presentan una alta homogeneidad e identidad genética para estos marcadores y pertenecerían a un núcleo genético común.
Palabras clave: Canis familiaris, genoma, Uruguay.
INTRODUCTION
In Uruguay, the Cimarron is the unique native canine breed. In 2006, the International Cinologic Federation (ICF) grants the recognition to it like breed at the International level. During the second half of the XIX century, these animals undergo a persecution with important reduction of their number because, in wild state, they were attacking the cattle and causing economic losses (1). A reduced nucleus of animals that hid in indigenous mounts in the Cerro largo Department (northeast region of Uruguay) survived to that persecution. Then, descendants of those animals would be the genetic base for the preservation and officialization of this breed, allowing to make its standard in 1989 (2).To date, the breeding of these animals is carried out in the rural area (work dog with bovine, hunting) and in the city (surveillance, company) (1). Within a Preservation program of breeds it is important to know the zootechnic situation of a native genetic resource. Previous studies made in this breed provided data on the ethnologic characterization (morphometric and phaneroptical analyses) (3).
The Cimarron exhibits a molosoid type with concave profile, being longilineous of average size (corporal length of 64 cm and cross height of 57 cm) characteristic of prey and grab dogs. The hair is smooth, short and the coat color is varied not being accepted in the standard the black or solid white, existing preference in the breeding of animals with striped coats (2) (Figura 1).
The use of RAPD (random amplification of polymorphic DNA) markers allows to deep in the genetic characterization of breeds with conservation aims and breeding animal (4). The random amplification of polymorphic DNA fragments, known as RAPD markers, uses the PCR technique with short oligonucleotides of random sequence to detect genetic polymorphism. Among individuals, the differences in the pattern of the amplified bands (polymorphism) will be given by the bases sequences differences where the oligonucleotides ones are jointed. The amplification of equal amounts mixtures of DNA (pools of DNA), has been used in diverse species to quickly evaluate the ability of a great number of initiators to detect specific breeds or populations polymorphisms. The advantages of this technique, over others used for the detection of genetic variability and racial characterization, have favored its application in different mammal species (4, 5).
Rothuizen and Van Wolferen (5) did the first report of the application of this technique in analysis of genetic variability in dogs. From there, these markers have been used in diverse studies like phylogenetic analysis of the family Canidae and polymorphisms search in the canine genome (6). The RAPD markers have been used to analyze the variability and the genetic distance in Spanish native canine breeds like Galgo Español, Podenco, Perro de Agua Español y Alanos (7).
To know aspects about the intrarace genetic variability in the Uruguayan breed Cimarron, its molecular characterization was begun using molecular DNA markers (8). The purpose of this study was to analyze the genetic variability in two populations of the native canine breed "Uruguayan Cimarron" from different areas of the country.
MATERIALS AND METHODS
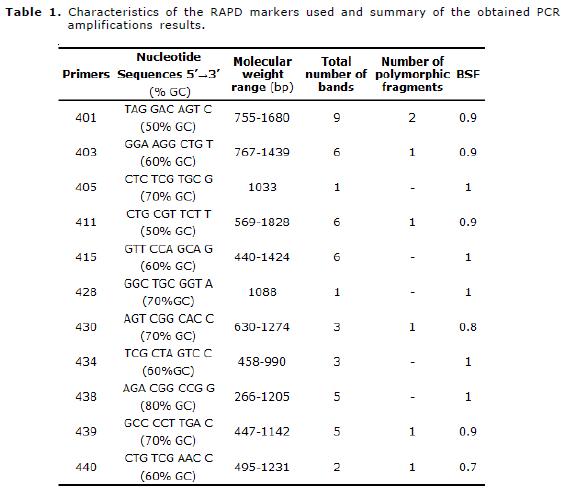
The DNA extraction was made from whole blood of 40 animals of the Uruguayan breed Cimarron (20 animals from the northeast region and 20 animals from the South region of the Country). The DNA purification was made according to the technique described by Cheung et al (9) and it was quantified by spectrophotometry. Two mixtures or pools were prepared adding equal amounts of DNA corresponding to the two populations (northeast population and South population of the Country). For the random DNA amplification, we used 11 oligonucleotides (10-mer with a GC richness between 50 and 80%) of the 5UBC series (University of British Columbia) (Tabla 1).
PCR reactions were carried out in a 25 ul-reaction volume containing 10 x PCR buffer (200 mM Tris.HCl, 500 mM KCl), 1.5mM MgCl2, 0.15 mM of each dNTP, 0.2mM of oligonucleotide and 0.75 U of Taq DNA polymerase. The PCR cycle profile used was: 94°C/2min (1 cycle for DNA denaturation); 94°C/2min, 36°C/1 min, 72°C/1min (35 cycles) and one final extension 72°C/5min. The products were separated by electrophoresis in ethidium bromide stained agarose gels (1.5%) and visualized under ultraviolet light. The Kodak Digital Science (1D, version 3.0) software for images analysis was used to study the amplified bands. To estimate the genetic variability in the populations the bandsharing frequency (BSF) was used, whose formula is: BS = 2Nxy/(Nx + Ny), where Nxy corresponds to the number of bands shared by the two populations (Nx and Ny to the bands that appears in the x and y populations, respectively) (10). The PopGene 32 program for dominant markers was used to calculate the Nei's gene diversity (H) and gene identity (I), the percentage of polymorphism (P) and the genetic distance (11).
RESULTS
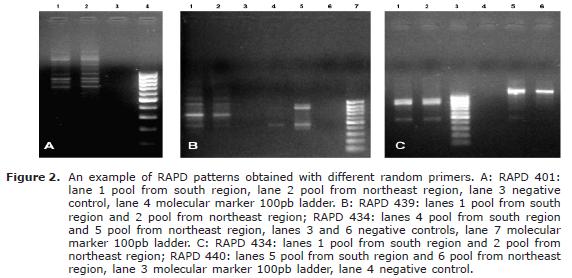
A total of 47 loci were obtained with the 11 oligonucleotides, being 7 of them (14.9%) polymorphic. The range of amplified loci was between 1 (405 and 428 markers) and 9 (401 marker). The size of the fragments varied between 266 and 1828 bp (Table 1, Figure 2) . The average of BSF was 0.91, Nei's average diversity index was H=0.08, genetic distance of 0.16 and a Nei's gene identity index of 0.85.
DISCUSSION
The utilization of RAPD with representative DNA pools of different populations has allowed to obtain a considerable number of marker fragments in a relative short time. In our work, when comparing the two populations, the average of BSF was of 0.91. The RAPDs 401, 403 and 434 showed more amplification bands in comparison with studies of intrapopulational variability carried out previously in the Cimarron dog (8). Nevertheless, in the present work the RAPDs 401 and 403 showed BSF (0,88 and 0,90) higher to that found by these authors (0.79 and 0.86) respectively.
Taking into account all the used markers, we found a high BSF (0.91), a low polymorphism (P=14.9%) and a Nei's average diversity index (H=0.08) indicating that both populations show a high homogeneity and gene identity for these markers. On the other hand, since both populations showed to be to a genetic distance of 0.16 and a high Nei's gene identity index of 0.85, we could infer that they belong to a common genetic nucleus. This could be due to the low original number of males used as reproducers when it began the rescue and standardization of the breed (2). In different Spanish native canine breeds, the genetic variability found with RAPD markers were smaller than it was detected using microsatellites markers (7,12). Believe that the used RAPDs series has been chosen without a previous knowledge of its value as detectors of polymorphism in the canine species. Given the low percentage of polymorphism obtained with this RAPDs series, is that we propose to continue the study of the genetic variability in the Uruguayan Cimarron dog and to compare it with other races using codominant microsatellites markers.
Acknowledgements
To Mrs. Iris C. Hernández for her technical assistance, Dr.M.V. Victor de Oliveira for his assistance in sample collection and Mr. Francisco Criserá for his assessment about the breed. This work was also support by grants from CSIC (University of Uruguay-UdelaR) and CIDEC (Faculty of veterinary-UdelaR).
REFERENCES
1. Fernández G. Situación de los recursos genéticos domésticos locales del Uruguay. Arch Zootec 2000; 49:333-340. [ Links ]
2. Silveira C, Fernández G, Barba C. Primeros datos de la caracterización etnológica del perro cimarrón. Arch Zootec 2002: 51:223-228. [ Links ]
3. Silveira C, Mernies B, Fernández G, Barba C. Estudio biométrico de una población canina de la raza cimarrón. Arch Zootec 1998; 47:529-532. [ Links ]
4. Olivier M, Meehl M, Lust G. Random amplified polymorphic DNA (RAPD) sequences as markers for canine genetic studies. J Hered 1999; 90:78-82. [ Links ]
5. Rothuizen J, Van Wolferen M. Randomly amplified DNA polymorphisms in dogs are reproducible and display Mendelian transmission. Anim Genet 1994; 25:13-18. [ Links ]
6. Stepniak E, Zagalska M, Switonski M. Use of RAPD technique in evolution studies of tour species in the family Canidae. J Appl Genet 2002; 43(4):489-499. [ Links ]
7. Morera Sanz L, Barba C, Garrido J. Detección de variabilidad en razas caninas autóctonas españolas mediante marcadores RAPD. Arch Zootec 2001; 50:379-382. [ Links ]
8. Llambí MS, Separovich MJ, Fernández G, Arruga MV. Primeros estudios moleculares en el perro Cimarrón del Uruguay. Veterinaria 2004; 39(155- 156):65-68. [ Links ]
9. Cheung W, Hubert N, Landry B. A simple and rapid DNA microextracción method for plant, animal, and insect suitable for RAPD and other PCR analyses. PCR Methods Appl 1993; 3:69-70. [ Links ]
10. Linch M. The similarity index and DNA fingerprinting. Mol Biol Evol 1990; 7:478-484. [ Links ]
11. Yeh F, Yang R, Boyle T, Ye Z, Mao J. PopGene. Alberta, Canada: University of Alberta and Center for International Forestry Research; 1967. [ Links ]
12. Morera Sanz L, de Andrés D, Barbancho J, Garrido J, Barba C. Detección de variabilidad genética por microsatélites en el alano español. Arch Zootec 1999; 48:72-77. [ Links ]














