1. INTRODUCTION
Polymer injection is one of the most promising techniques to increase oil recovery from light to heavy crude oil reservoirs [1]-[3]. This method has been implemented for about 40 years in fields all over the world [4,5]. By using this process, it is expected to obtain 50% of the last recovery, and even 15-20% incremental oil over water injection [6].
The polymer flooding process uses polymers as viscosifying agents to improve the mobility ratio and increasing oil recovery [7,8]. Polymers for EOR processes must be water-soluble, increase the viscosity of injection brines, and an expected decrease of the water/ oil mobility ratio [9,10]. The improvement in the mobility ratio increases the macroscopic displacement efficiency [11]. Additionally, the viscoelastic nature of HPAM polymers contributes to improve the macroscopic sweep efficiency [12].
Nevertheless, polymers are susceptible to thermal and chemical degradation, which means that the technology has technical limitations due to the polymer degradation mostly because of exposure to high temperature, high salinity/hardness, as well as mechanical efforts [13]. Similarly, high concentrations of a polymer are required to reach the viscosity target to change the mobility ratio [14]-[16]. Due to these limitations in many Colombian field conditions, it is not feasible to implement polymer injection processes regarding high operational costs.
In recent years, the research has focused on developing new polymers with improved performance at high temperatures and high salinity (HTHS) [17]. However, these alternatives imply higher production costs and technical limitations due to the molecular size obtained [18]. One of the alternatives to reduce this limitation is the use of nanoparticles that interact in polymeric water solution systems, which are substances showing great potential in EOR processes [19]. This hybrid nanomaterials and nanocomposites have improved properties based on organic-inorganic interactions that reduce or avoid chemical and thermal degradation of HPAM polymers [20]-[23].
This paper focuses on the study of the chemical and thermal degradation process of polymers, the alternatives for controlling or avoiding it, and the nanofluid formulations and synthesis routes of hybrid nanomaterials based on nanoparticles and synthetic polymer used for enhanced oil recovery. This review is organized as follows. First, it describes the most relevant characteristics of polymers for EOR, including thermal and chemical degradation mechanisms. Next, current methods to control the thermal and chemical degradation are reviewed. The use of nanoparticles stands out, mainly focused on the polymer-based synthesis of nanohybrid materials and the application in EOR processes evaluated at the laboratory.
2. CHEMICAL CHARACTERISTICS OF POLYMERS USED FOR EOR
The polymers used in EOR are composed of water-soluble molecules that increase the viscosity of brine solutions. They must maintain specific properties to ensure an improved oil displacement efficiency; the most relevant characteristics are summarized in Table 1.
Table 1 Main characteristics of the polymer solution used in EOR process.
| Characteristic | Description |
|---|---|
| Water solubility | Formation of hydrogen bonds between the lateral polymeric groups and the polar water molecules. |
| Molecular weight | Commercial polymers have a molecular weight that varies in the range of 1-25 million Daltons. The oil displacement ability of polymer can be reinforced by increasing the chain length |
| Distribution of the chain | Usually, mixtures of homologous polymers with different molecular weights are used. |
| Diversification of geometry | Polymers must have several molecular geometries: linear, branched, and corpus. |
| Viscoelasticity | In particular, a polymer with a longer chain exhibits stronger elastic properties, which enhances the foregoing pulling effect. |
| Viscosity performance | The polymers must be highly effective as to the accumulation of viscosity. This means that, even at a very low concentration, the polymer should quickly dissolve in water or brine solution and increase the viscosity up to the target, especially in high salinity brine. |
| Chemical and mechanical stability | The polymer will penetrate, migrate, and propagate in the porous media under reservoir conditions. It will eventually leave the formation with the water produced; therefore, the polymers must have excellent properties such as viscoelasticity, filtration, chemical, and mechanical stability. |
Source: Modified from [24]-[27].
According to the basic polymer requirements used for EOR applications, some functional groups, and chemical bonds in their structures are needed to ensure their performance. The main functional groups of EOR polymers are summarized in Table 2.
Table 2 Chemical groups in polymer molecular structures.
| Structure | Description | Examples of polymers |
|---|---|---|
| Carbon chain in the main column | Good thermal stability, degradation at temperatures higher than 80°C. | Polyvinyl, sodium polyacrylate, polyacrylamide, HPAM |
| -COO- in the hydrophilic group | Good viscosifier, less adsorption in sandstones due to the repulsion between anions components in the chain, solubility in water, but it precipitates with Ca2+ y Mg2+ and has less chemical stability. | Sodium alginate, sodium carboxymethyl cellulose, cellulose, HPAM, Xanthan gum |
| -OH or -CONH2 in the hydrophilic group | No precipitation occurs with Ca2+ y Mg2+, good chemical stability. However, there is no repulsion between chain bonds, thus obtaining less viscosifier power, high adsorption due to the hydrogen bond formed on the sandstones. | Polyvinyl, HEC, polyacrylamide, HPAM |
| Acrylamide tertiarybutyl sulfonic acid (ATBS) or AMPS in the hydrophobic group | High tolerance to brines containing divalent ions and prevented polymer hydrolysis at high temperatures. However, at temperatures above 100ºC, it presents high hydrolysis degree, which generates a significant loss of viscosity due to thermal degradation. | Sulfonate polymers |
On the other hand, polymers for EOR should be thermally stable at reservoir conditions for several months or even years. Polymer flooding in high temperature (HT) reservoirs can lead to a reduction in polymer solution viscosity, which has an impact on process efficiency. In addition to the viscosity reduction at HT, polymers can reduce the hydrodynamic radius or precipitate in high hardness conditions [30]. Suitable polymers for EOR processes should remain effective and stable in terms of viscosity for several months at reservoir conditions [31], [32], Hence, polymer stability will strongly depend on reservoir temperatures, brine composition, geochemistry, and well spacing in patterns, among others.
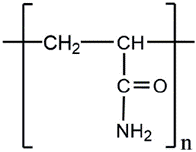
Most polymers currently used for Chemical Enhanced Oil Recovery (cEOR) applications are modified molecules of polyacrylamides (PAM). PAM structure is shown in Figure 1. Its nature is non-ionic in the non-hydrolyzed form [33].
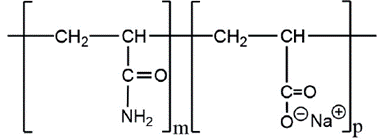
Although PAM does not hydrolyze at room temperature [34], the amide group present therein can hydrolyze at high temperature and/or pH [35]. The hydrolysis result is the formation of carboxylate groups that provides a negative charge in the polymerstructure. This mproves the viscosity due to intramolecular repulsions [36]. In this regard, the water-soluble polyacrylamide commercially available for cEOR applications is usually post-hydrolyzed. These polymers are produced by adding alkali or acrylamide (AM) and acrylic acid (AA) co-polymers or its salts. These cEOR polymers are known as HPAM (partially hydrolyzed polyacrylamide). Hydrolysis converts some of the amide groups (CONH2) into carboxyl groups (COO~) as shown in Figure 2.
Incorporating the acrylate group in the PAM structure makes the chain rigid due to repulsion of ions charges [37]. This makes the HPAM less sensitive to degradation by shear than PAM [38], and it is less adsorbed in the reservoir rock.
Due to these improvements, HPAM is the most widely used polymer in cEOR applications [39]-[44]. In this regard, a large number of publications have reported their evaluation under numerous laboratory and field conditions [45]-[56].
3. CHEMICAL AND THERMAL DEGRADATION
High temperatures cause loss of viscosity in polymer solutions due to the significant molecular thermal movement leading to the breakup of the molecular chain [57]-[61]. HPAM polymers have a substantial limitation at temperatures above 70°C [62, 63]. At high temperatures, the functional groups of molecules change particularly through hydrolysis, which allows degradation and viscosity reduction [64], [65]. This effect varies depending on the temperature and brine composition [47], [66]. However, HPAM can remain stable at high temperatures (<100°C) in the absence of divalent ions and very low content of oxygen or hydrogen sulfide (<20 ppb) [67].
The HPAM chemical structure is sensitive to salinity. It consists in long molecular chains with charges located along the chain; these charges interact with brines ions. Generally, salinity affects the polymer's viscosity as follows [68] [69]: At low salinities, the molecular chains expand due to the greater repulsion of charges causing an increase in viscosity. On the other hand, at high salinity and hardness, divalent ions could neutralize the charges in hydrolyzed acrylamide groups, causing a coiling effect and reducing viscosity.
At high salinities, the acrylate monomers in the HPAM are strongly associated with cations. Therefore, the solution approaches the polyacrylamide (PAM) viscosity. Multivalent cations have a more significant effect than monovalent cations, mainly in aerobic conditions. Zaitoun et al. [70] determined 33% as the degree of critical hydrolysis for precipitation in the presence of divalent cations at 80°C. However, if there is no precipitation, the strong interaction between the cations and the carboxylate group could reduce the viscosity of HPAM [42].
According to Müller's terminology (1981) [71], the term "chemical degradation" refers to the hydrolysis of the functional groups of the polymer, and "thermal degradation" is used when the free radical induce a breakdown of the acrylic bond, resulting in a reduction in molecular weight. Chemical degradation leads to a higher hydrolysis degree, and it could be prevented by the inclusion of chemically stable monomers.
The loss of viscosity during thermochemical degradation experiments is typically attributed to the reduction in molecular weight as a result of the breakage of the acrylic bond induced by the presence of free radicals [75]. Some chemical radicals can extract hydrogen from acrylic polymers, which cause a radical centered on the carbon in the polymer backbone, which can generate its division through the steps described by Grollman et al. in 1982 [76]. Further, for ionic polymers (such as HPAM), the solubility is very sensitive to the ionic strength of the aqueous phase. At high salt concentrations, the solubility of the ionic polymers decreases rapidly, and phase separation can occur [42],[70], [72]-[74].
At high temperatures, oxygen, even in traces, can lead to the polymer backbone breaking apart due to the reaction of free radicals in the chain [68], which in turn leads to a significant decrease in the molecular weight of the polymer and, therefore, the viscosity of the polymer solution.
In sum, the application of HPAM is generally limited to 70°C in the presence of divalent cations [75], [77]. However, in the presence of negligible amounts of divalent cations (<200 ppm), the application of HPAM can be extended up to 100°C [78] in anaerobic conditions. In this sense, it is clear that PAM and HPAM have deficiencies and require some modifications or changes for implementation in cEOR in high salinity and high-temperature (HSHT) reservoirs [79],[80].
4. ALTERNATIVES FOR THE DEGRADATION CONTROL
Numerous attempts have been made to improve the performance of EOR polymers for HTHS reservoirs conditions, mainly by modifying the chemical structure of PAM and HPAM. Generally, to extend the application of acrylamide (AM) polymer, copolymerization with monomers is used. This contributes to increase the strength and stiffness of the polymer chain [42][81]. The modified PAM is divided into three categories; the first one involves co-polymers or terpolymers synthesized by incorporating rigid or strong monomers. These monomers are more resistant to chemical degradation and capable of providing cationic protection. Additionally, this can sterically prevent the polymer chain from maintaining the hydrodynamic radius at high salinity [77], [82]. The second category includes polymers synthesized by the incorporation of hydrophobic monomers, which can improve the viscosity by intermolecular association. The third category is related to PAM based on thermo-viscosifying polymers having a thermosensitive monomer in the main hydrophilic chain [36].
In the first category of modified polymers, some AM monomers' replacement with other nonionic monomers that are more stable with electrostatic forces can also stabilize neighboring acrylamide fragments in the molecule [83]. These polymers withstand high temperatures and are known as copolymers or terpolymers depending on the components used. For example, terpolymers combined with acrylamide are obtained as ATBS (Acrylamide Tertbutyl Sulfonate) and NPV (N-Vinyl Pyrrolidone), which hold temperatures above 120°C. However, ATBS polymers are more expensive than conventional HPAM [16]. Other special polymers, resistant to high temperatures and other harsh conditions, are described in Table 3.
Table 3 Applications reference conditions of special polymers.
| Type of polymer | Salinity [ppm] | T [°C] | Time [days] | Comments |
|---|---|---|---|---|
| HPAM | 30,000 | 85 | 100 | Reference for other polymers |
| Sulfonic acid 2-acrylamido-2- methylpropane (AMPS) -AM | 33,560 | 93 | 100 | High Price |
| Na-AMPS | 33,560 | 121 | 100 | High Price |
| VP-AM | 33,756 | 121 | 100 | Low molecular weight, low viscosity |
| AM-AMPS-VN | 67,850 | 80 | 90 | Low molecular weight |
| AM-ATB | 68,917 | 105 | 50 | High price and high hydrolysis above 100°C |
The second group of polymers requires adding some hydrophobic monomers that allow intermolecular associations, improving the system's rheological behavior [87],[88], The difficulty of using some of these advanced polymers is related to flow properties in Low permeability and long-term thermal stability, which have not been extensively tested as the HPAM [89]-[91], Additionally, they have excellent rheological properties in high salinity. However, it also shows high adsorption in reservoir rocks, which affect performance (i.e., lower effective viscosity) and increase the polymer concentration required, which translates into increased project costs [92]-[94]. This type of polymers has been tested at a pilot stage for a heavy oil sandstone reservoir. Nevertheless, results have been unsuccessful due to its high molecular weight and the permeability reduction generated by the interaction between the hydrophobic chains [95],[96].
With respect to the third category, the thermo-thickening polymers contain some hydrophobic pendant groups, which are soluble in water at room temperature but start to self-aggregate at some critical temperature to minimize their exposure to water [97], The rheology is controlled by salt concentration, temperature, and pH and it has a thermo-thickening monomer in the main hydrophilic chain [98], Nonetheless, these products are not commercially available in high molecular weights.
h this regard, the co-terpolymers are more resistant than conventional HPAM to high salinities (70 g/l) and high hardness [99], However, they are expensive as high polymer concentration is necessary (2,000 - 3,000 ppm) to reach target viscosities between 10 to 20 cP (depending on the brine composition).
The use of nanotechnology is emerging as a possible solution to improve the properties of conventional and advanced HPAM polymers. Nanoparticles have been used to improve the thermal and chemical stability of HPAM aqueous solutions [100]. This is because nanoparticles offer interesting transport mechanisms (i.e., physical filtration, solution chemical stability, and adsorption on the rock/porous media surface) due to their dimensions (<100 nm), surfaces activity, and high surface area per unit volume [101].
The cost of chemicals is an important factor in cEOR projects. Generally, nanoparticles are used in very low concentrations, and they are cheaper than polymers used for HTHS reservoir applications as ATBS. Most of the nanoparticles considered for EOR applications are environmentally friendly materials compared to traditionally used chemicals. Such is the case of silica nanoparticles, which are the main component of sandstone reservoirs, and they have a better effective-cost ratio in most of the applications [102,103].
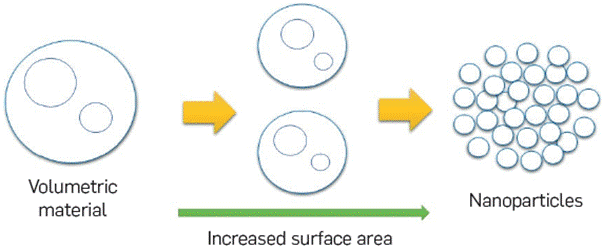
Nanoparticles have size ranges from 1nm to 100nm (Figure 3) [1023, Due to the nano size, nanoparticles have a very high surface to volume ratio, and the large surface area increases the number of atoms on the nanoparticle surface. Figure 3 shows the concept of the growing surface area with decreasing particle size; in each step, the sample has the same mass and volume, but a larger surface area with a very small size result [102].
5. NANOFLUIDS
The nanofluid concept was documented by Stephen U.S. Choi in 1995 [104], who described the nanofluid as a colloidal mixture in which the nanoparticles' synergy properties and the base fluid contribute to change the thermal and transport properties of the base fluid [105,106]. Nanofluids are characterized by the Brownian agitation exceeding any settlement movement due to gravity. Thus, a stable nanofluid is theoretically possible as long as the particles remain sufficiently small (usually 100 nm) [107],[108].
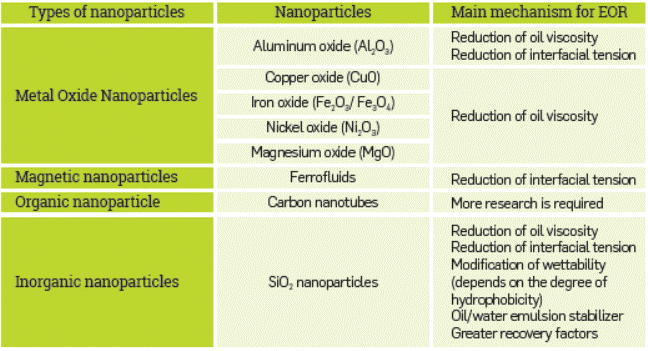
The development of nanofluids is still hampered by several factors such as few suitable models of theoretical properties, the Lack of matching between theoretical and experimental results, the poor characterization of suspensions, and a conceptual misunderstanding of the interaction mechanism [22], [109]-[112] However, many types of research are conducted on the properties of these compounds [113]. The evaluation of thermal properties, particle-based properties such as size, shape, surface area, colloidal properties such as suspension stability, Zeta potential, pH, and final properties based on lubrication like viscosity and shear stress [35] are to be highlighted. Table 4 shows the main mechanism of nanoparticles with EOR application potential. Most of them are based on new properties achieved by using nanoparticles in a base fluid.
According to the literature, the silicon oxide (silica) nanoparticles show the most useful and promising properties for EOR applications [115],[116], including high compatibility with the reservoir and less environmental impact [117]. Some parameters can also affect the displacement process in reservoir rocks, as temperature, base fluids (i.e., polymer), type of nanoparticles, nanoparticle size distribution, and injection time. These parameters have shown significant effects on the performance of SiO2 nanofluid injection. Hendraningrat et al. [118] studied the effects of nanoparticle size, rock permeability, initial rock wettability, and temperature in coreflood tests. They concluded that smaller nanoparticle size distribution and higher temperatures resulted in a high incremental oil recovery [118,119]. Additionally, due to its small size, the nanofluid injection has potential applications in a wide range of reservoir permeabilities [120,121] and Adelet al. [122].
Nonetheless, based on recent laboratory experiences reporting permeability losses in coreflood experiments [123],[124], additional efforts understanding possible formation damage (i.e., face plugging, nanoparticle bridging, deep-bed filtration) at different injection rates and modeling techniques considering filtration effects [125] are required to support the upscaling of this technology at a field scale. Table 5 summarizes some of the most critical studies of nanofluids for enhanced oil recovery.
Table 5 Experiments of silica-based nanofluids injection for EOR processes.
| Author | NPs/Base Fluid | Type of crude | Oil properties | Reservoir type | Increased oil recovery [%] | |
|---|---|---|---|---|---|---|
| Density [kg/m3] | Viscosity [cP] | |||||
| Alomair et al., 2001 [126] | Al2O3, SiO2, NiO, TiO2/brine | Heavy oil | 950 | 206,88 | Sandstone core | 16.94 - 23.73 |
| Ogolo et al., 2012 [127] | SiO2/ethanol | Medium oil | 911 | 53.28 | Sandpack | 13.324.1 |
| Torsater et al. , 2012 [128] | SiO2/brine | Paraffinic crude | 803 | 1,956 | Sandstone | Secondary (8) Tertiary (<2) |
| Maghzi et al., 2012 [129] | SiO2/brine | Heavy oil | 933 | 870 | Micromodel | 8.7 - 26 |
| Hendraningrat et al., 2013 [130] | SiO2/brine | Light oil | 826 | 5,1 | Sandstone | 5.93-14.29 |
| Li et al., 2013 [131] | SiO2/brine | Light oil | 826 | 5,1 | Sandstone | 4.26-5.32 |
| Hendraningrant et al., 2013 [121] | SiO2/brine | Light oil | 826 | 5,1 | Sandstone | 0-9.9 |
| Hendraningrant et al., 2014 [132] | SiO2, Al2O3, TiO2/brine | Light oil | 826 | 5,1 | Sandstone core | 7-11 |
| Adel et al., 2015 [133] | SiO2, Al2O3,/brine | Medium oil | 851 | 75 | Sandstone | SiO2 (8.74- 13.88) Al2O3 (8.12 - 4.65) |
| Tarek y El Banbi, 2015 [134] | Fe2O3 + Al2O3 + SiO2/brine | Mineral oil | 919 | 5,12 | Sandstone | 0.9 - 9.49 |
| Li et al., 2015 [135] | SiO2/brine | Light oil | 847 | 15,3 | Sandstone | 5-15 |
| Roustaei et al., 2015 [136] | SiO2/brine | Light oil | 857 | 11 | Carbonates | 9-12, 16-17 (24 h) |
| Mohebbifar et al., 2015 [137] | SiO2 - biomaterial/water | Heavy oil | 925 | 200 | Micromodel (Shale) | 28-40 |
| Kasemzadeh et al., 2015 [138] | SiO2, NiO, Fe3O4 /solution | Asphalt + toluene | N/A | N/A | Micromodel | SiO2 (22.6) NiO (14.6) Fe3O4 (8.1) |
| Adel et al., 2015 [122] | SiO2/brine | Light oil | 851 | 75 | Sandstone | 5-10 |
| El - Diasty, 2015 [139] | SiO2/brine | Mineral oil | 891 | N/A | Sandstone | 9-19 |
| Tarek, 2015 [140] | Fe2O3 + Al2O3 + SiO2/brine | Mineral oil | 919 | 5.12 | Sandstone | 8.99 - 20.42 |
As observed in Table 5, the silica-based nanofluid has excellent potential in EOR methods [141], obtaining up to 26% incremental oil recovery. The oil recovery increase is attributed to multiple mechanisms but not limited to IFT and oil viscosity reduction, wettability alteration, water/oil emulsion stabilizer [114]. However, most of these results were obtained at a laboratory scale, and few field cases have been documented in the literature except for specific well treatments or productivity enhancement. Some examples include the inhibition of formation damage, fine migration control, and mobility improvement of heavy and extra heavy oils implemented in Colombian oil reservoirs [142],[143]. Based on current and leading experiences in Colombia, several joint efforts among national agencies, universities, state and private companies are carrying out the evaluation of technical-economic feasibility for upscaling cEOR-assisted with nanotechnology.
Lastly, most of the nanotechnology applications to improve conventional processes, such as polymer injection, have been based on nanofluids (i.e., nanoparticles suspended in polymer solutions) [193[144]-[145]. This approach shows encouraging results, and the use of nanohybrid systems has been proposed to improve polymer's resistance. Nanohybrids, such as polymers chemically bonded on the surface of the nanoparticle, can enhance the thermochemical degradation resistance of polymers [146]-[150]. The use of nanohybrids will be addressed in the following section of this review.
4. NANOHYBRID SYSTEMS (POLYMERS -NANOPARTICLES)
In sum, there are two methods to incorporate nanoparticles in polymer solution for EOR processes [151]:
Regarding the first method based on the incorporation of nanoparticles at lab conditions, Zhu et al. in 2014 [154] studied the improvement of the rheological behaviour of the polymer/water fluid. This study considered the addition of silica nanoparticles using associative hydrophobic polymers of HPAM (HAHPAM). They obtained suspensions that show stable nanofluid and increased viscosity of the system due to the nanoparticles cross-linking properties, offering higher resistance and long-term thermal stability than HAHPAM (hydrophobic HPAM) in synthetic brine. This system also showed greater displacement efficiency (about 5% additional) when compared with the coreflood using only HAHPAM at the same concentration. Furthermore, the results show that the injection of silica nanoparticles can alter the wettability of the porous medium (to strongly water-wet) [155].
Yousefvanda and Jafaria [156] and Maghzi et al. [152] prepared silica nanoparticles (0.5% wt) and synthetic polymers (HPAM) formulations, obtaining increased fluid viscosity with respect to the polymer solution. Coreflood tests also reported higher oil recoveries (up to 10%) than the polymer solution in the absence of nanoparticles.
However, recent studies have reported that incorporating nanoparticles into the polymer structure by chemical reaction results in better chemical stability and greater displacement efficiencies due to the double interaction (organic-inorganic) on this nanohybrid system [157]. Table 6 summarizes some of the most prominent studies reporting the synthesis of nanohybrid systems based on EOR polymers and silica nanoparticles.
Table 6 State of the art for the synthesis of nanomaterials for Enhanced oil recovery.
| References | Nanomaterial (Route of synthesis) | Rheology behavior | Displacement Efficiency (%) | Comments |
|---|---|---|---|---|
| Ponnapati et al., 2011 [158] | Based on NanoSiO2 - Polyethylene (in-situ free radical polymerization) | Increase viscosity with high shearing rates | 7.2% OOIP in sandstone core | Viscosity reached very low |
| Nguyen et al., 2012 [153] | Nanocomposite of Surfactant/polymer/NanoSiO2 (encapsulation - polymerization-core - Shell) | Improves viscosity at a temperature of 92°C and 3.44% salinity | 6.2% incremental oil in offshore conditions | Reduction of interfacial tensionand improve viscosity atcritical concentration, highthermo |
| Pu et al., 2015 [162] | Hyperbranched polymer Core-Shell (HBPAMs) based on Nano-SiO2-PAM core of nano-silica, and hyperbranched polyamide (PAMAM) as subshell and linear hydrophilic chains in the outermost layer (in-situ free radical polymerization) | High resistance to shearing degradation, esistance to salt and temperature | 16% incremental oil compared to linear polymers | The hyperbranchedcontains hydrophilic linearchains |
| Lai et al., 2016 [163], [171],[172] | Hyperbranched polymer base on Acrylic acid/Acrylamide /FNS - functionalized nanoparticle (HPMS) (in-situ free radical polymerization) | Additional 20% viscosity retention to HPAM | 18.84% EOR for Low - Moderate permeability reservoirs | The complex improvesthermal stability, stressresistance, and viscoelasticity. |
| Liu et al. 2016 [164] | Core-shell polymer (HBAPAM) with functionalized nano-SiO2 as core multibranched associative polymeric chains as shell (in-situ free radical polymerization) | After 25d of aging, viscosity retention of 77% (47% viscosity retention rate additional of HPAM solution). Core-shell polymers exhibited the rheological property in multiscale, and only the sheared thinning for HPAM displayed in the higher shear rate region. | The highest EOR of 24% (12% additional over HPAM) and Et of 69% for HBAPAM with a polymer concentration of 1100 mg/l (0.3 PV of polymer slug) was recovered in moderate permeability conditions (356 mD) | The core-shell structure andmultibranched morphologyenhanced the supramolecularinteraction of the core-shellpolymer with functionalizednano-SiO2, which displayedthe desirable viscosifierefficiency and viscoelasticity,especially long-term stabilityin harsh temperature andsalinity conditions |
Ponnapati et al., 2011 [158] show an increased viscosity with high shearing rates due to a shear thickening property in hybrid solutions. This behavior is consistent with recent reports of the shear thickening observed in attractive colloidal systems, as well as nanoparticles stabilized with ionic moieties [159] - [161].
Also, Pu et al. 2015 [162] and Lai et al. 2016 [163] evaluated nanohybrid solutions and compared them with the HPAM solution, finding that nanohybrids had higher viscosity retention ratios under the same conditions. Therefore, the nanohybrids showed some resistance to shear, which depends on the degree of modification. This suggests that the inclusion of nanoparticles in a polymer matrix generated resistance to shear [164] - [165].
The inclusion of nanoparticles in the polymeric matrix generates materials that are more resistant to biodegradation during their life cycle. Furthermore, increasing nanomaterial amounts embedded in a polymeric nanocomposite reduce its biodegradability, while maintaining the nanocomposite structural and chemical integrity. It represents an advantage for applications that require biostability for long periods [166] - [168].
The degradation rate in nanocomposites in the presence of clay nanoparticles [169] shows the enhancement of polymers as barrier properties. The nanoparticles reduce biodegradation by increasing the path length and inducing a slower rate of degradation in comparison with the polymer precursor [170].
As shown in Table 6, nanohybrids synthesized by Ponnapati et al. 2011 [158] shows an oil recovery of 7.2% OOIP in sandstone core, and 6.2% incremental oil in offshore conditions as reported by Nguyen et al., 2012 [153]. These results are very similar to oil recovery with nanofluids documented by Yousefvanda and Jafaria [156] and Maghzi et al. [152] when compared with the coreflood using polymer at the same concentration in the absence of nanoparticles (up to 10%) [155].
However, the nanohybrids synthesized by Pu et al., 2015 [162] achieved 16% of incremental oil compared to linear polymers. Additionally, the nanohybrids developed by Lai et al., 2016 [163], [171] ,[172] reaches 18.84% oil recovery compared with 11.35% for the linear polymer (HPAM). These results showed that the nanohybrid had a more outstanding ability for EOR than the linear polymer under the same conditions, maybe due to its excellent shear resistance, rheology performance, and mobility control capability in a porous medium.
Also, nanohybrids prepared by Liu et al. 2016 [164] reached an oil recovery of 24%, which increase 12% the recovery obtained by HPAM on the same conditions. This particular application confirmed that nanohybrids has better mobility control capability. Thus, it might have a robust application with high-cost performance in the harsh formation water condition of reservoirs.
However, nanohybrids are usually more expensive than nanofluids due to the complex synthetic routes steps necessaries for generating a new compound. Nevertheless, the EOR application must be evaluated considering technical-economical aspects, including the possible nanoparticle loss from the nanofluid into the reservoir in high flow rate zones and highly adsorbed/retained in the porous media. Furthermore, nanohybrids can generate higher viscosity than nanofluids using the same polymer quantity, contributing to economic feasibility.
Table 6 shows that the most common synthesis route is based on the in-situ free radical polymerization strategy. Result reports improved resistance to thermal and chemical degradation, which maintain the viscosity and support the mobility control capability: This makes viable its application providing higher performance in harsh reservoir conditions. This technique allows obtaining denser polymers with a lower hydrodynamic radius than HPAM, but similar molecular weights. The latter results in higher viscosities with lower polymer concentrations.
The use of new nanohybrids based on polyacrylamide and silica nanoparticles was recently reported by Ruiz-Cañas et al. [173] [174]. Reported results suggest that these nanohybrids can potentially be applied in the oil and gas industry, including Improved and Enhanced Oil Recovery (IOR/EOR). Preliminary results show that the chemical bonding of Si02 nanoparticles and HPAM polymer (Nanohybrid) generate positive effects on the viscosity at low shear rates and temperatures for the same experimental conditions. It should be noticed that the nanohybrid evaluated (CSNH-BCTA2) at a shear rate of 10 s' 1 , has a viscosity 20% higher than the same HPAM at the same concentration (1,500 ppm) and temperature of 100°C. However, further details of this research are included in Quintero et al., [175]
CONCLUSIONS
The primary mechanism of thermal and chemical degradation of HPAM is related to changes in its chemical structure. In this regard, there are numerous studies about the improvement of the structure of HPAM polymers. However, the most significant advances are based on making the structure stiffer or protecting the monomer's chemical charges.
Nanotechnology could provide the solution to polymer degradation issues related to a new organic-inorganic interaction between HPAM and Si02 nanoparticles, which provides more resistance in harsh temperature and salinity conditions, in addition to other properties such as reducing interfacial tension or wettability alteration.
Recent research studies of nanofluids show promising results for cEOR methods. Nevertheless, only the applications for well treatments or productivity enhancement (i.e., formation damage inhibition, fine migration control, and mobility improvement of heavy and extra heavy oils) have been reported at field-scale.
Based on this review and on recent findings, great opportunities exist for implementing base polymer nanofluids in cEOR projects. This approach could be valuable to optimize the technical-economic feasibility of the projects by reducing the polymer concentration, using reasonable amounts of nanoparticles. However, more significant efforts are required to understand the impact of nanoparticle concentrations and injection rates to support the upscaling of this cEOR technology.
Nanohybrids based on polymers and silica nanoparticles for cEOR show promising results for improving viscosity and thermal stability as compared to the HPAM polymer precursor. Therefore, future research will be focused on seeking to extend its application to a pilot scale.