Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Universitas Medica
Print version ISSN 0041-9095On-line version ISSN 2011-0839
Univ. Med. vol.60 no.4 Bogotá Oct./Dec. 2019
https://doi.org/10.11144/javeriana.umed60-4.sgar
Research articles
Acute Myocardial Infarction and Left Branch Bundle Block: Utility of the Sgarbossa Criteria
1Residente de Medicina Interna de la Universidad de la Sabana, Chía, Colombia
2Médico de la Universidad de La Sabana, Chía, Colombia.
3Médico de la Universidad de La Sabana, Chía, Colombia.
4Médico de la Universidad de La Sabana, Chía, Colombia.
5Médica de la Universidad de La Sabana, Chía, Colombia.
6Residente de Medicina Interna, Universidad Pontificia Bolivariana, Medellín, Colombia.
7Residente de Medicina Interna de la Universidad de la Sabana, Chía, Colombia.
8Médico internista, neumólogo, epidemiólogo, Clínica Universidad de La Sabana, Chía, Colombia.
Introduction:
Acute myocardial infarction (AMI) in the presence of left bundle branch block (LBBB) is a diagnostic challenge for the physician. The purpose of this article is to evaluate the usefulness of the Sgarbossa criteria for the diagnosis of AMI in patients with LBBB through a systematic review and meta-analysis.
Method:
Structured literature search in Medline, Lilacs, Ovid, and Embase from January 1996 to January 2018. Using the inclusion and exclusion criteria 3 reviewers selected the articles that answered the research question. The quality of the articles was evaluated using the QUADAS-2 tool.
Results:
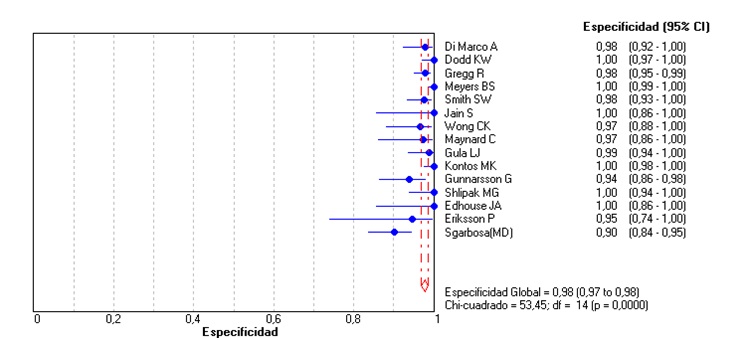
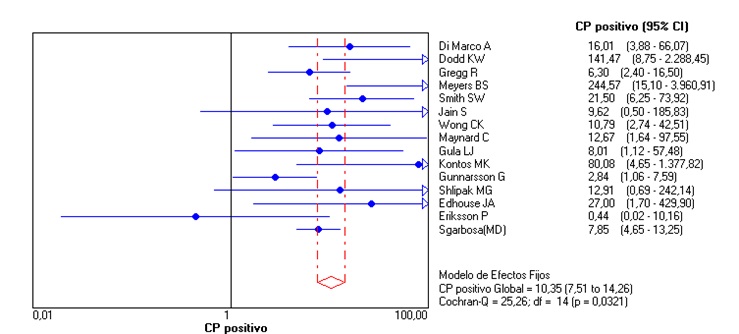
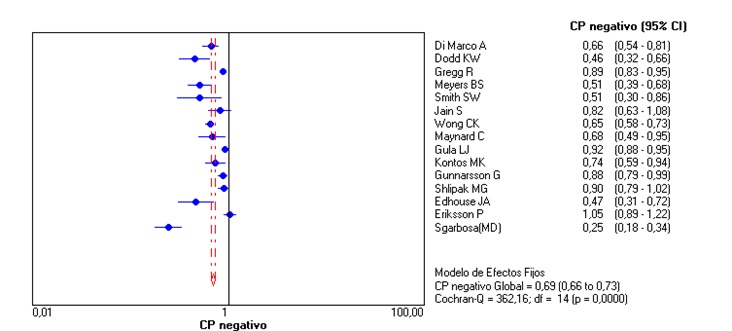
14 studies evaluated the Sgarbossa criteria using a score ≥ 3 points. With a total of 3689 patients, the sensitivity was 0.27 (95% CI: 0.24-0.29; p = 0.0000); specificity of 0.97 (95% CI: 0.96-0.98; p = 0.0001) LR(+) was 10.95 (6.28-19.11; p = 0.03), and LR(–) was 0.67 (0.56-0.81; p = 0.0000).
Conclusions:
The Sgarbossa criteria have a high specificity for the diagnosis of AMI with LBBB; however, the sensitivity and the LR(–) are low. It is necessary to evaluate new diagnostic algorithms and to validate the criteria for Colombia.
Keywords acute myocardial infarction; acute coronary syndrome; bundle branch block; diagnosis; Colombia
Introducción:
En presencia de bloqueo de la rama izquierda, el infarto agudo de miocardio (IAM) es un reto diagnóstico para el médico.
Objetivo:
Evaluar la utilidad de los criterios de Sgarbossa en el diagnóstico de IAM en pacientes con bloqueo de la rama izquierda mediante una revisión sistemática y metaanálisis.
Método:
Búsqueda estructurada de la literatura en Medline, Lilacs, Ovid y Embase, desde enero 1996 hasta enero de 2018. Basados en los criterios de inclusión y exclusión, tres revisores seleccionaron los artículos que respondieran la pregunta de investigación. La calidad de los artículos se evaluó con el instrumento QUADAS-2.
Resultados:
Catorce estudios evaluaron los criterios de Sgarbossa utilizando un puntaje mayor o igual a 3 como corte. En total, fueron 3689 pacientes. La sensibilidad fue de 0,27 (IC95 %: 0,24-0,29; p = 0,0000); especificidad de 0,97 (IC95 %: 0,96-0,98; p = 0,0001) LR(+) de 10,95 (6,28-19,11; p = 0,03) y LR(–) de 0,67 (0,56-0,81; p = 0,0000).
Conclusiones:
Los criterios de Sgarbossa tienen una alta especificidad para el diagnóstico de IAM con bloqueo de la rama izquierda; pero bajo LR(–) y sensibilidad. Se deben evaluar nuevos algoritmos diagnósticos y validar para la población colombiana.
Palabras clave infarto agudo de miocardio; síndrome coronario agudo; bloqueo de rama; diagnóstico; Colombia
Introduction
The concomitant presence of left bundle branch block (LBB) and suspicion of acute myocardial infarction (AMI) is a diagnostic and therapeutic challenge for the physician (1). For this reason, the Sgarbossa criteria facilitate the management of these patients (Table 1) (2). However, despite having a good specificity, in the original study published in 1996, the sensitivity barely reached 36% (2). This means that a large percentage of patients could be excluded from reperfusion therapies because they do not have an AMI diagnosis if these criteria are not interpreted adequately. We carried out a systematic review and meta-analysis, in order to determine if the Sgarbossa criteria perform well for the diagnosis of AMI in patients with LBBB.
Methodology
We carried out a systematic review in Pubmed, Embase, OVID and Lilacs databases. For the search strategy we used the terms: (acute myocardial infarction) OR (myocardial infarction) OR (coronary syndrome) AND (bundle branch block). Articles published from January 1, 1996 to January 1, 2018, were considered, including cases and controls and cohort studies in English, Spanish, Portuguese, French and Italian. We included adult patients who had been in the emergency room, hospitalized or in an intensive care unit, with suspected AMI and concomitant presence of LBBB, in whom a score ≥ 3 points in the Sgarbossa criteria would have been evaluated, compared to the gold standard. Regarding the latter, given the heterogeneity in the definition of the standard and the few existing articles, any of the following was taken into account: interobserver agreement, positive cardiac enzymes and positive cardiac catheterization concordant with arterial lesion responsible for AMI. We excluded studies in which each criterion was evaluated separately, without including the evaluation of a score ≥ 3 points. Pregnant women and the pediatric population were excluded from the review and meta-analysis. No topic reviews, case presentations or case series were considered.
Two members of the research group (DAMA and DVM) reviewed the different databases with all the titles and summaries of the initial search. They included and excluded the articles for evaluation and, later, the rest of the research group reviewed them. The data was transferred to an Excel® spreadsheet, where the characteristics of the studies and their specific estimates or summary measures were evaluated.
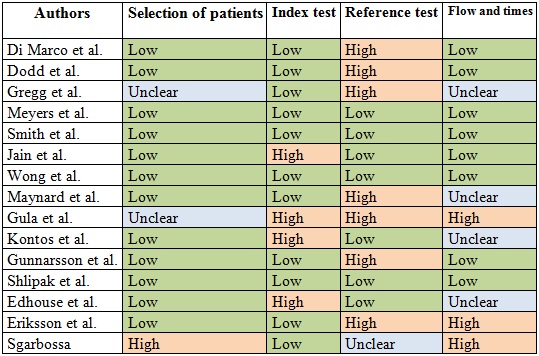
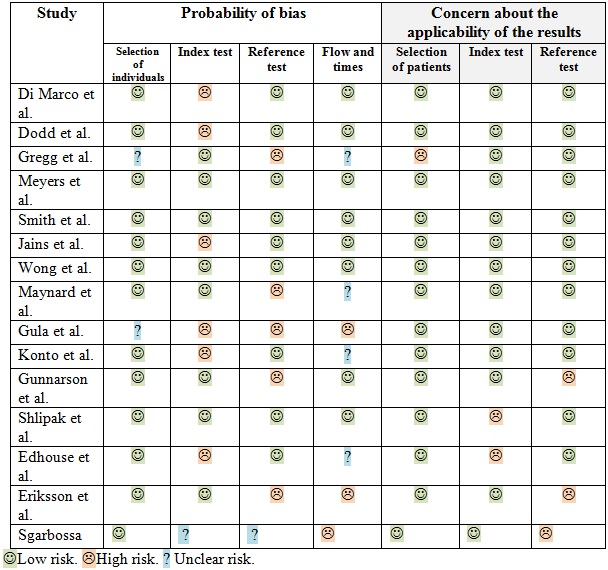
To evaluate the methodological quality and the quality of the evidence of the included studies, we used the QUADAS-2 instrument, which is the current recommended tool for assessing the risk of bias and the applicability of primary studies of diagnostic precision that are used in systematic reviews.
Through this tool, we evaluated two fundamental aspects: the proportion of concern generated by the applicability of the primary studies used for the systematic review, and the proportion of concern generated by the primary studies regarding the risk of bias; this by means of evaluating the flow and times, reference test, index test and patient selection.
Results
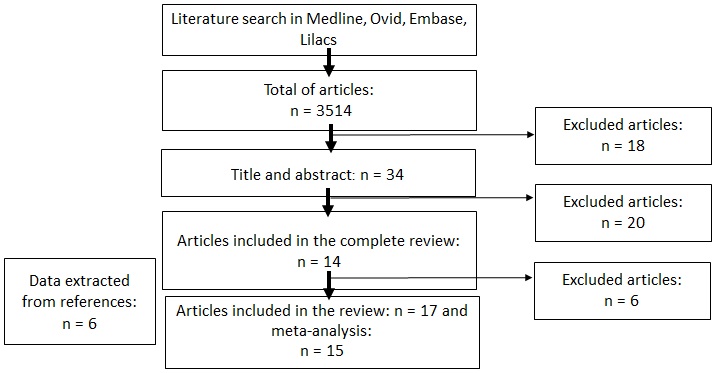
In the initial search, 3514 articles were identified, 17 of which were selected for the systematic review, 6 articles were chosen with the “snowball” strategy based on the references of different articles of the initial search; 12 corresponded to cases and controls and the others to cohort studies. Of the 17 reviewed, 15 were chosen to perform the meta-analysis. Two of them did not provide data to obtain the sensitivity and specificity of a score ≥ 3 in the Sgarbossa criteria (Figure 1).
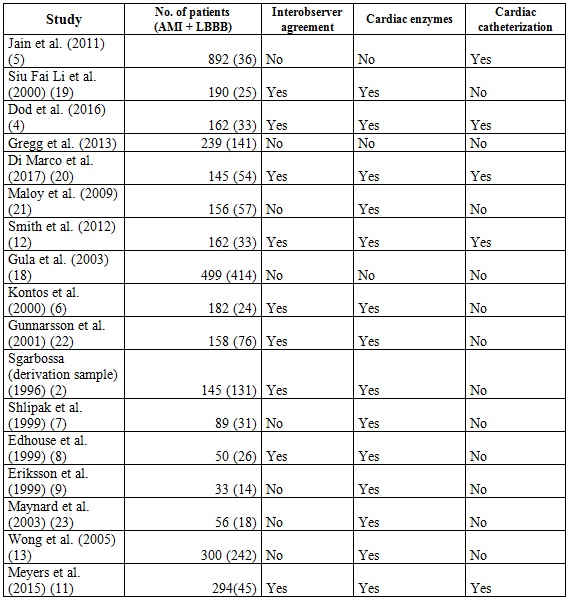
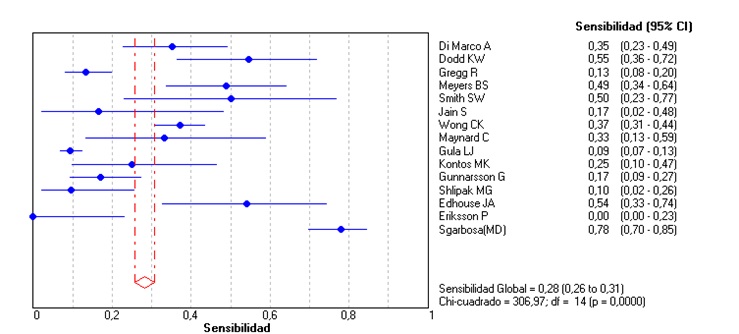
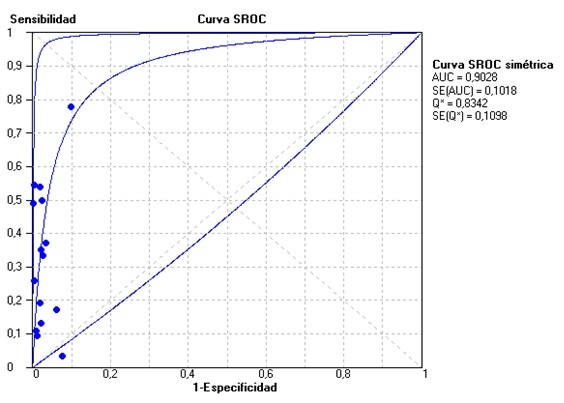
Unlike the meta-analysis published in 2008 by Tabas et al. (3), the original Sgarbossa article (2) was included, and the articles that used the patients of this study were not excluded. In the included studies, AMI was determined by cardiac enzymes in some or all of the patients, interobserver agreement and use of cardiac catheterization or the combination of these (Table 2). For a score ≥ 3 in the Sgarbossa criteria we obtained an overall sensitivity of 0.27 (95% CI: 0.24-0.29, p = 0.0000) (Figure 2); a global specificity of 0.97 (95% CI: 0.96-0.98, p = 0.0001) (Figure 3); an area under the curve of 0.90; an overall positive likelihood ratio (LR+) of 10.95 (6.28-19.11, p = 0.03) and an overall negative likelihood ratio (LR-) of 0.67 (0.56-0.81; p = 0.0000) (Figures 4, 5, 6), all of which shows great heterogeneity among the different studies.
In 6 articles (4,5,6,7,8,9) the LR+ could not be determined, given that its specificity was 100%; 50% of these articles did not exceed 100 patients, and only one exceeded 500 patients (5), and in the latter only 36 patients had AMI + LBBB. The small number of patients made our meta-analysis difficult. Despite this, the results are similar regardless of the sample size, as can be seen in Table 3, and in Figures 2 and 3, corresponding to sensitivity and specificity; a score ≥ 3 has a very low sensitivity and excellent specificity in all articles of the meta-analysis.
Table 3 Punctual estimated summaries: evaluation of the statistical parameters of the different evaluated works
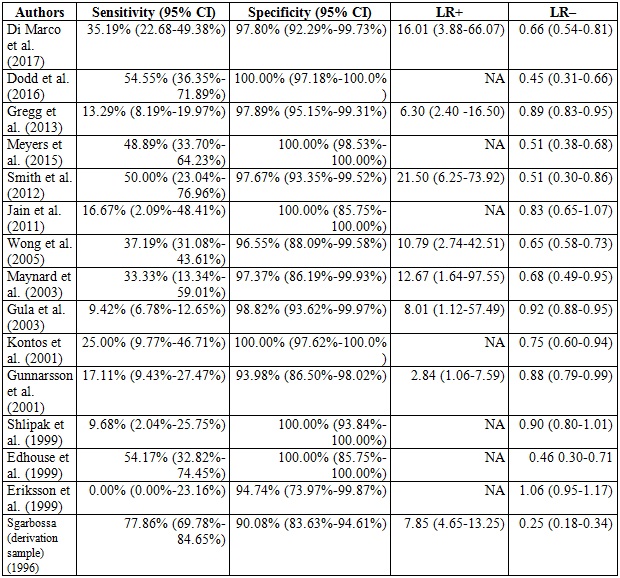
NA:not applicable.
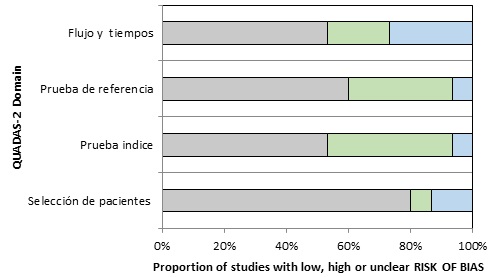
Regarding the definition of AMI, there is a wide heterogeneity regarding the reference pattern to define infarction: less than 10% of the patients underwent cardiac catheterization and only 5 studies (4,5,10,11,12) were evaluated with this diagnostic strategy that would correspond to the best method used in all articles. The quality of the evidence for each article was rated with the QUADAS-2 instrument (Tables 4 and 5; Figures 7 and 8).
Discussion
The diagnosis of AMI in patients with LBBB is a diagnostic challenge for the clinician. It is estimated that the prevalence of LBBB in AMI patients is between 2% and 9% (13,14). In the study Assessment of the Safety and Efficacy of a New Thrombolytic Regimen 2 and 3, a significant elevation of creatine phosphokinase-MB occurred in 62.5% of the 253 patients who had LBBB (14,15). The prevalence of LBBB in AMI patients varies considerably and even more in studies in which enzymes were used as a reference method to define AMI. In studies in which cardiac catheterization was used, the prevalence is more uniform and slightly higher compared to those that used cardiac enzymes, which is between 15% and 35% (5,14). For this reason, for more than a decade, different researchers have tried to develop diagnostic algorithms to increase detection or have a more adequate way to detect and identify patients with AMI who have LBBB. The Sgarbossa criteria (Table 1) are the best known. However, despite being frequently used in clinical practice, their sensitivity and specificity vary between studies, and their usefulness and application are unclear.
Based on this premise, our meta-analysis evaluated the efficacy of the Sgarbossa criteria. With the overall results of the performance of these criteria for the prediction of AMI associated with LBB with a score ≥ 3, we observed that, due to their low sensitivity, the presence of AMI cannot be ruled out when this score is not higher than indicated. On the contrary, given their high specificity (Figure 3), positive predictive value and LR+ (Figure 4), they allow us to diagnose with AMI those patients who meet the criteria. However, it should be emphasized that the clinician must always make the decision according to the clinical presentation of the patient. Most of these studies do not evaluate the different prognostic scales that involve physical examination, risk factors, cardiac enzymes and the electrocardiogram as a whole. This is how the 2017 European guidelines on AMI with ST elevation state that these diagnostic aids serve “to assist diagnosis” and the clinical suspicion of myocardial ischemia in conjunction with all these findings must be what defines the management (16).
According to the meta-analysis carried out by Tabas et al. (3) in 2008, with a Sgarbossa score ≥ 3, the sensitivity obtained was 20%, while the specificity was 98% and it had a LR+ of 7.9, which was clinically useful, because at that time it correlated with 7 of the 10 electrocardiographic criteria used to diagnose AMI in patients without branch block. They also measured the same variables when the score was ≥ 2 and they found that both sensitivity and specificity did not improve and, instead, they became heterogeneous, so it was not of clinical utility (3).
It should be noted that the original article by Sgarbossa was not included in that investigation. On the other hand, in the present study, based on scores ≥ 3, the overall sensitivity reached was 27%, which continues to be low despite the small increase (the specificity was 97%), which agrees with the results of the previous meta-analysis. However, despite this agreement with the studies and the previous meta-analysis, some limitations should be mentioned. Firstly, the sample of AMI + LBB patients remains small in order to obtain more adequate confidence intervals. On the other hand, the definition of AMI in the studies found is very variable and does not represent what is currently used. Most studies published before 2008 used creatine kinase as a strategy to diagnose AMI, while studies conducted since 2010 increased the use of troponins. However, although these (4,5,10,11,12,17) are closer to the current practice, differ in the use of cardiac catheterization and, sometimes, in the definition of the J point and different variables when interpreting the algorithms.
Some recent studies have proposed new criteria based on those of Sgarbossa, as is the case of the Smith algorithm, which includes the Sgarbossa criteria and scores, but with a modification in the third criterion, which makes the 1 mm discordant elevation of the ST segment more proportional. With this modification, an increase in the sensitivity of the Sgarbossa criteria was observed as an electrocardiogram-based diagnostic method to differentiate LBBB patients with or without acute coronary event, especially when they have occlusion of the anterior descending artery (10,12,18,19,20,21,22,23). However, more recent research has shown that the Sgarbossa criteria included in Smith rules continue to have a suboptimal sensitivity (24,25). Smith rules 1, 2 and 3 showed a sensitivity of 67%, 54% and 28%, respectively, while the specificity remained considerable, above 90%, in the three rules.
Conclusion
The Sgarbossa criteria have a high specificity for the diagnosis of AMI + LBBB. However, they have low sensitivity and very low negative likelihood ratios (LR). Based on the foregoing, these criteria can be used as a tool for diagnosis associated with the patient’s clinical condition, which should prevail in case of suspected acute coronary syndrome. These criteria should be validated and studied in the Colombian population, to have a concept of the performance of this strategy in our patients.
Referencias
1.Sgarbossa EB. Value of the ECG in suspected acute myocardial infarction with left bundle branch block. J Electrocardiol. 2000 Jan;33:87-92. [ Links ]
2.Sgarbossa EB, Pinski SL, Barbagelata A, Underwood DA, Gates KB, Topol EJ, et al. Electrocardiographic diagnosis of evolving acute myocardial infarction in the presence of left bundle-branch block. N Engl J Med. 1996 Feb 22;334(8):481-7. [ Links ]
3.Tabas JA, Rodríguez RM, Seligman HK, Goldschlager NF. Electrocardiographic criteria for detecting acute myocardial infarction in patients with left bundle branch block: A meta-analysis. Ann Emerg Med. 2008;52(4):329-36. https://doi.org/10,1016/j.annemergmed.2007,12.006 [ Links ]
4.Dodd KW, Elm KD, Smith SW. Comparison of the QRS complex, ST-segment, and T-wave among patients with left bundle branch block with and without acute myocardial infarction. J Emerg Med. 2016;51(1):1-8. [ Links ]
5.Jain S, Ting HT, Bell M, Bjerke CM, Lennon RJ, Gersh BJ, et al. Utility of left bundle branch block as a diagnostic criterion for acute myocardial infarction. Am J Cardiol. 2011 Apr 15;107(8):1111-6. [ Links ]
6.Kontos MC, McQueen RH, Jesse RL, Tatum JL, Ornato JP. Can myocardial infarction be rapidly identified in emergency department patients who have left bundle-branch block? Ann Emerg Med. 2001 May;37(5):431-8. [ Links ]
7.Shlipak MG, Lyons WL, Go AS, Chou TM, Evans GT, Browner WS. Should the electrocardiogram be used to guide therapy for patients with left bundle-branch block and suspected myocardial infarction? JAMA. 1999 Feb;281(8):714. [ Links ]
8.Edhouse JA, Sakr M, Angus J, Morris FP. Suspected myocardial infarction and left bundle branch block: electrocardiographic indicators of acute ischaemia. J Accid Emerg Med. 1999;16(5):331-5. [ Links ]
9.Eriksson P, Gunnarsson G, Dellborg M. Diagnosis of acute myocardial infarction in patients with chronic left bundle-branch block. Standard 12-lead ECG compared to dynamic vectorcardiography. Scand Cardiovasc J. 1999;33(1):17-22. [ Links ]
10.Di Marco A, Anguera I, Rodríguez M, Sionis A, Bayes-Genis A, Rodríguez J, et al. Assessment of smith algorithms for the diagnosis of acute myocardial infarction in the presence of left bundle branch block. Rev Esp Cardiol. 2017 Jul;70(7):559-66. [ Links ]
11.Meyers HP, Limkakeng AT, Jaffa EJ, Patel A, Theiling BJ, Rezaie SR, et al. Validation of the modified Sgarbossa criteria for acute coronary occlusion in the setting of left bundle branch block: A retrospective case-control study. Am Heart J. 2015 Dec;170(6):1255-64. [ Links ]
12.Smith SW, Dodd KW, Henry TD, Dvorak DM, Pearce LA. Diagnosis of ST-elevation myocardial infarction in the presence of left bundle branch block with the ST-elevation to S-wave ratio in a modified Sgarbossa rule. Ann Emerg Med. 2012 Dec;60(6):766-76. [ Links ]
13.Wong CK, French JK, Aylward PEG, Stewart RAH, Gao W, Armstrong PW, et al. Patients with prolonged ischemic chest pain and presumed-new left bundle branch block have heterogeneous outcomes depending on the presence of ST-segment changes. J Am Coll Cardiol. 2005;46(1):29-38. [ Links ]
14.Mehta N, Huang HD, Bandeali S, Wilson JM, Birnbaum Y. Prevalence of acute myocardial infarction in patients with presumably new left bundle-branch block. J Electrocardiol. 2012 Jul;45(4):361-7. [ Links ]
15.Al-Faleh H, Fu Y, Wagner G, Goodman S, Sgarbossa E, Granger C, et al. Unraveling the spectrum of left bundle branch block in acute myocardial infarction: Insights from the Assessment of the Safety and Efficacy of a New Thrombolytic (ASSENT 2 and 3) trials. Am Heart J. 2006 Jan;151(1):10-5. [ Links ]
16.Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, et al. 2017 ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2018 Jan;39(2):119-77. [ Links ]
17.Gregg RE, Helfenbein ED, Babaeizadeh S. New ST-segment elevation myocardial infarction criteria for left bundle branch block based on QRS area. J Electrocardiol. 2013;46(6):528-34. [ Links ]
18.Gula LJ, Dick A, Massel D. Diagnosing acute myocardial infarction in the setting of left bundle branch block: prevalence and observer variability from a large community study. Coron Artery Dis. 2003 Aug;14(5):387-93. [ Links ]
19.Li SF, Walden PL, Marcilla O, Gallagher EJ. Electrocardiographic diagnosis of myocardial infarction in patients with left bundle branch block. Ann Emerg Med. 2000;36(6):561-5. [ Links ]
20.Sionis A, Bayes-genis A, Marco A Di, Anguera I, Rodrı M, Ariza-sole A, et al. Assessment of smith algorithms for the diagnosis of acute myocardial infarction in the presence of left bundle branch block. Rev. Esp. Cardiol. 2016;70(7):559-66. [ Links ]
21.Maloy KR, Bhat R, Davis J, Reed K, Morrissey R. Sgarbossa criteria are highly specific for acute myocardial infarction with pacemakers. West J Emerg Med. 2010;11(4):354-7. [ Links ]
22.Gunnarsson G, Eriksson P, Dellborg M. ECG criteria in diagnosis of acute myocardial infarction in the presence of left bundle branch block. Int J Cardiol. 2001 Apr;78(2):167-74. [ Links ]
23.Maynard SJ, Menown IB, Manoharan G, Allen J, Mc CAJ, Adgey AA. Body surface mapping improves early diagnosis of acute myocardial infarction in patients with chest pain and left bundle branch block. Heart. 2003;89(9):998-1002. [ Links ]
Received: December 27, 2018; Accepted: April 12, 2019











 text in
text in